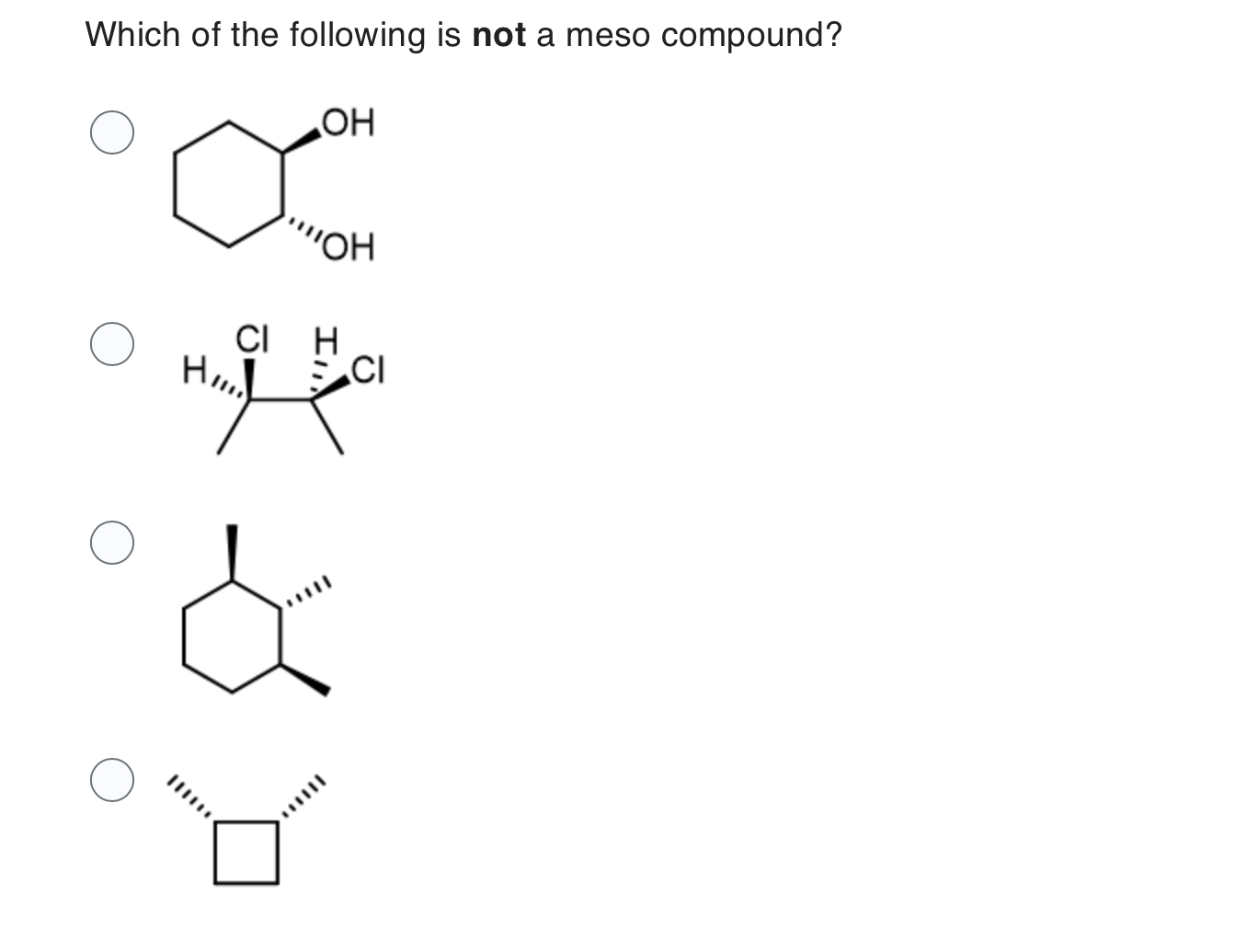
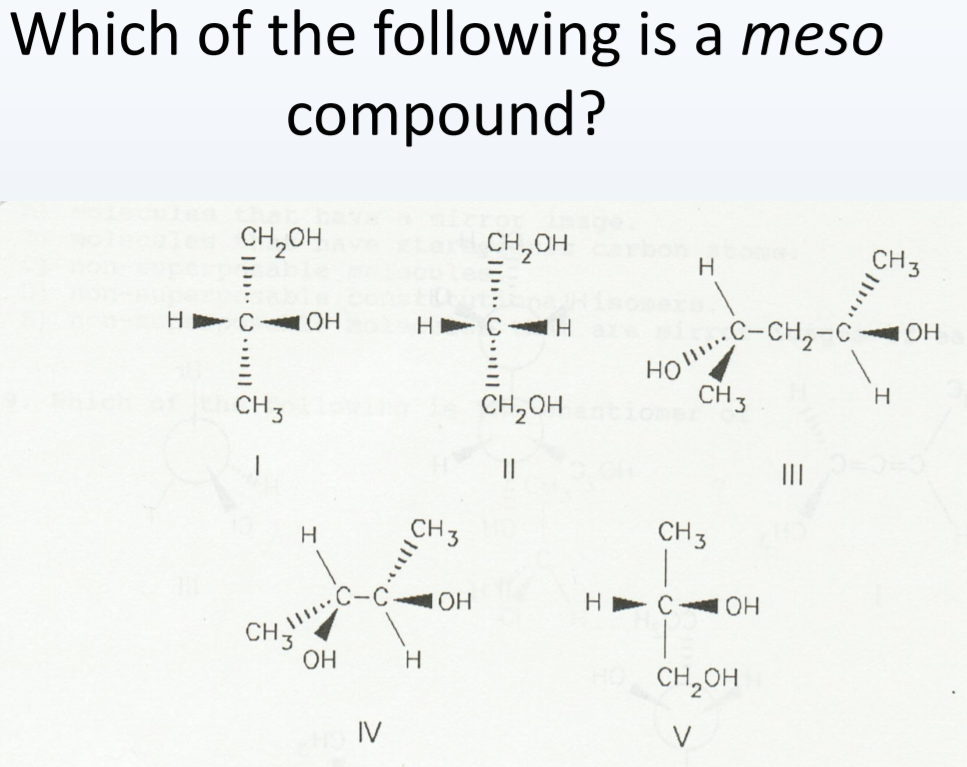
Which Of The Following Is A Meso Compound
You know, I was staring at a jar of pickles the other day – don't ask why, it was late and my brain does weird things. And it struck me, how something so simple, so ordinary, can have this hidden complexity. Like, one pickle might look just like another, right? But then you start thinking about how they're made, the way they twist and turn, and suddenly, it’s not so straightforward anymore. It’s like that moment when you’re trying to explain something obvious to someone, and they just… don't get it. And you're left thinking, "Seriously? Is it just me?"
Well, that’s kind of how I feel when I’m trying to wrap my head around the concept of meso compounds in chemistry. They're these molecules that, on the surface, seem to have all the ingredients for being "chiral" – you know, like a left hand and a right hand. They’ve got these stereocenters, these little points of asymmetry that should make them non-superimposable on their mirror images. But then, poof, they’re not! They're achiral. It’s like finding a pickle that’s its own mirror image. Wild, right?
So, let’s dive into this fascinating little puzzle. We're going to talk about which of the following is a meso compound. Now, if you’re already scratching your head, don't worry. That’s the exact right place to be. Chemistry, especially organic chemistry, is full of these delightful little twists that make you question everything you thought you knew. And meso compounds are the poster children for that kind of delightful confusion.
Must Read
First off, what even is a stereocenter? Think of it as a carbon atom that's bonded to four different groups. These are the building blocks of chirality. If a molecule has one stereocenter, it's pretty much guaranteed to be chiral. If it has two, things get a little more interesting, but it still can be chiral. But what if it has two or more stereocenters, and still manages to be achiral? Welcome to the club, meso compounds!
The magic trick that makes a meso compound achiral, despite having stereocenters, is something called a plane of symmetry. Imagine you can slice that molecule right down the middle, and both halves are perfect mirror images of each other. Not just sort of mirror images, but exact mirror images. This internal mirror plane cancels out any potential chirality. It’s like having two opposite forces that perfectly neutralize each other. So, even though it looks like it should be able to exist as a pair of enantiomers (those non-superimposable mirror images), it doesn't. It’s its own mirror image. Mind. Blown.
Now, let's get to the nitty-gritty. You might be presented with a list of molecules and asked to identify the meso one. This is where your detective skills need to kick in. You need to be able to:
1. Identify Stereocenters
This is your first step. Go through the molecule, carbon by carbon. Look for those carbons that are attached to four different atoms or groups. Don't be fooled by carbons that are part of double bonds or are only attached to two or three other things. We're looking for saturated carbons with four unique partners.
For example, if you see a carbon bonded to a hydrogen, a methyl group (CH3), an ethyl group (CH2CH3), and a chlorine atom (Cl), bingo! That carbon is a stereocenter. It has four different things attached to it. Easy peasy… well, maybe not easy peasy, but you get the idea. You’re basically playing a game of "spot the difference" for each carbon atom.
2. Consider the Possibility of Enantiomers
Once you've identified your stereocenters, you need to think about whether the molecule could exist as different stereoisomers. If there's only one stereocenter, it's chiral. If there are two or more, you have to do more work.
Here's a little trick for figuring out the maximum number of stereoisomers. For a molecule with n stereocenters, there can be a maximum of 2^n stereoisomers. So, if you have two stereocenters, you could have up to 2^2 = 4 stereoisomers. If you have three, it's up to 2^3 = 8. But this is just the maximum. Sometimes, due to symmetry, you end up with fewer. And that's where our meso friends come in.
3. Look for Internal Symmetry (The Key!)
This is the defining characteristic of a meso compound. After you’ve identified stereocenters, you need to check if there’s a plane of symmetry that cuts through the molecule in such a way that it makes one half the mirror image of the other. This plane of symmetry must pass through the molecule in a way that relates the stereocenters to each other.
Think of it like this: if you have two stereocenters, and the molecule has a plane of symmetry that bisects the molecule and swaps the two stereocenters as mirror images of each other, then it's likely a meso compound. This symmetry essentially "hides" the chirality that you'd expect from having those stereocenters.
So, imagine a molecule with two carbons, each with four different groups attached. If the groups attached to one carbon are, in a mirrored fashion, the same as the groups attached to the other carbon, and there’s a plane of symmetry between them, then it’s meso. It’s like a molecule that’s perfectly balanced on a seesaw, but the seesaw is made of mirrors.
Let's look at some classic examples, because examples are, like, the best way to understand this stuff. We're going to consider some common molecules you might encounter when this question pops up.

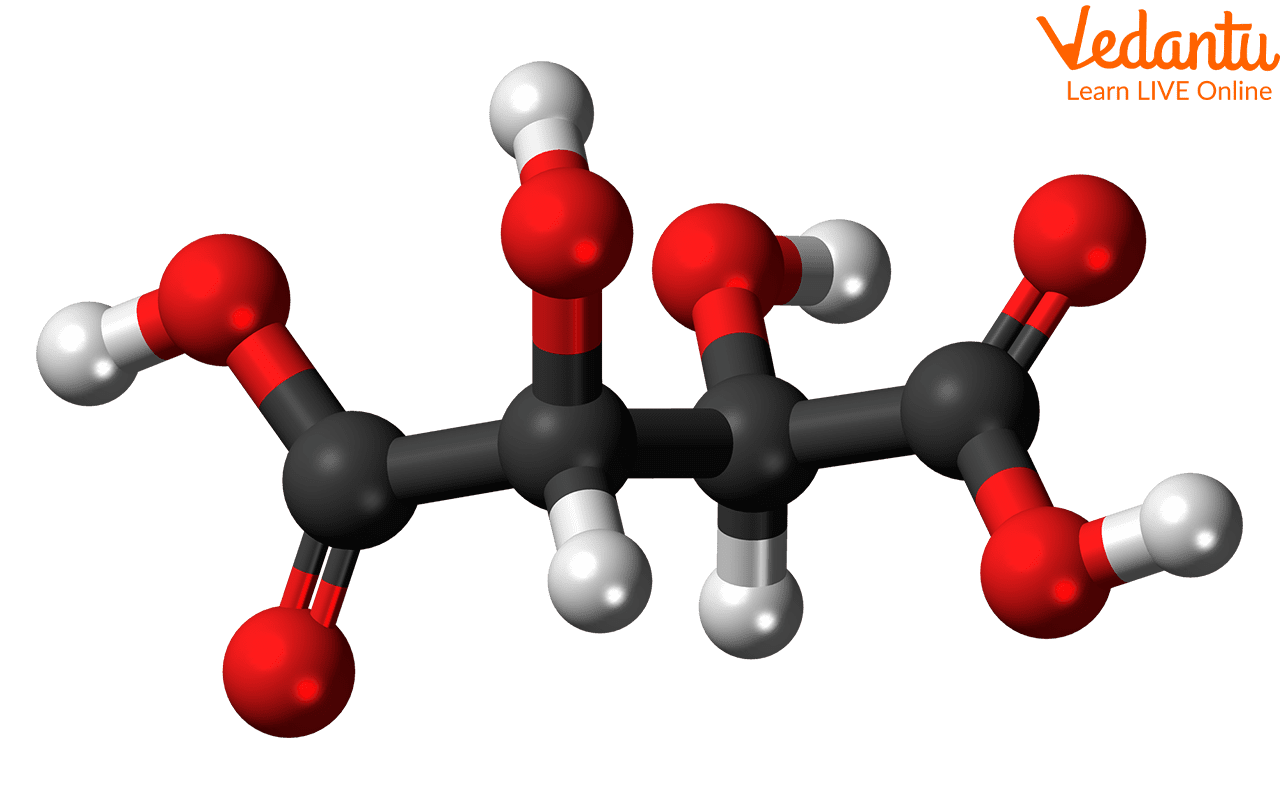
Example 1: Tartaric Acid
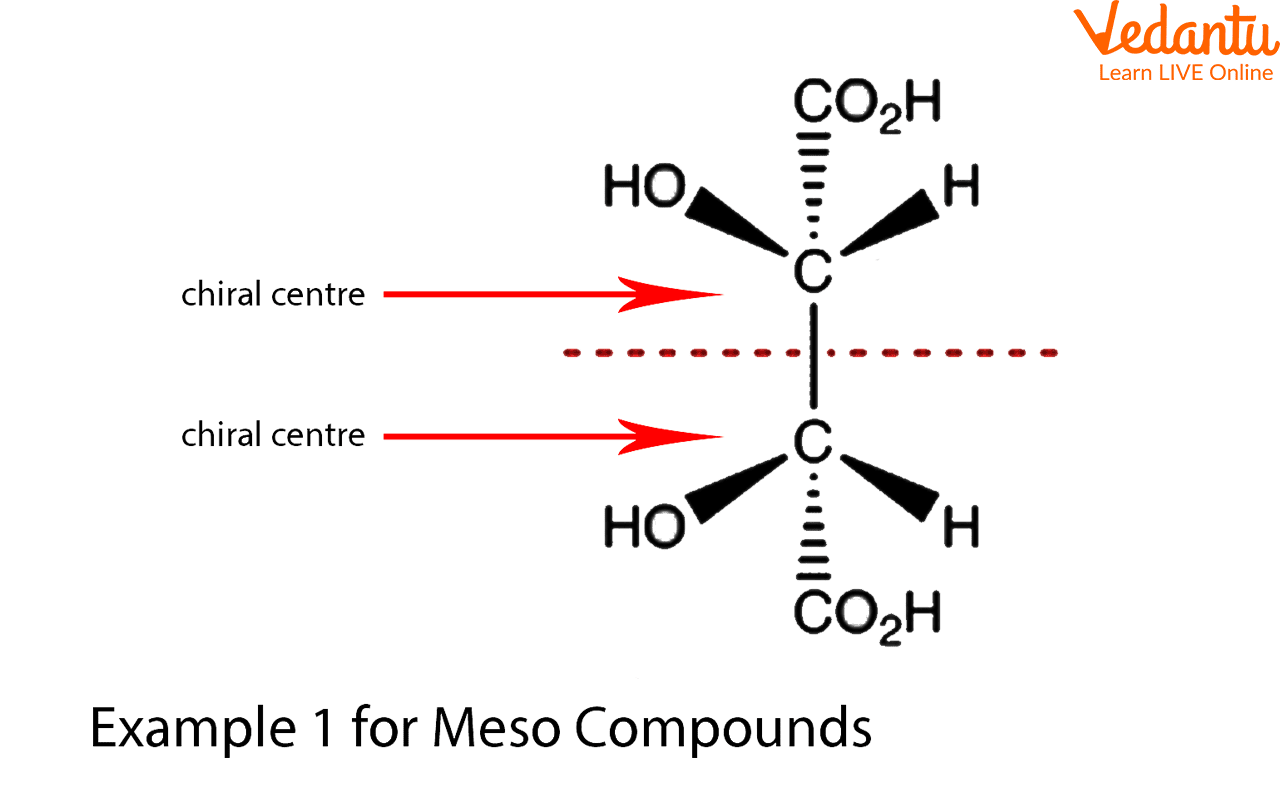
Tartaric acid is a textbook example. It has two stereocenters. You can draw its enantiomers (those left-hand and right-hand versions that are mirror images) and they will be different compounds. BUT, there's also a form of tartaric acid that is achiral. How? Because it has an internal plane of symmetry. Imagine drawing it out, and you can literally slice it in half, and each half is the mirror image of the other. This specific form is the meso tartaric acid. So, if tartaric acid is on your list, and you're looking for a meso compound, this is a strong contender!
It's kind of like finding a perfectly symmetrical snowflake. Each arm might have a unique pattern, but the overall structure is symmetrical. With meso compounds, the "arms" are the stereocenters, and the overall symmetry cancels out the chirality.
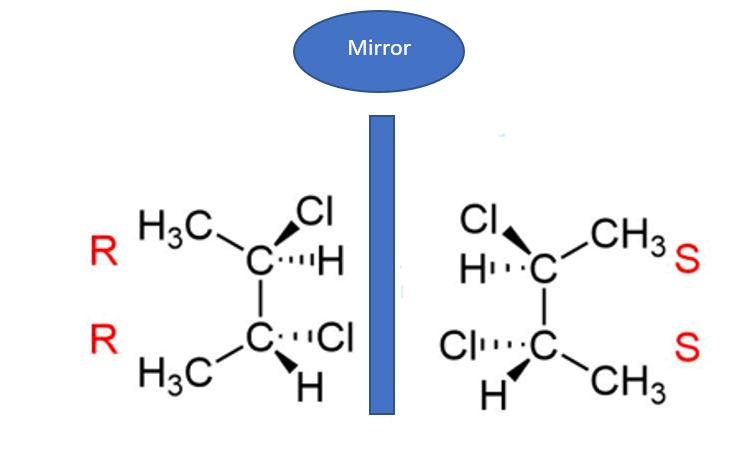
Example 2: 2,3-Dibromobutane
Let's consider 2,3-dibromobutane. This molecule has two stereocenters: the carbon at position 2 and the carbon at position 3. Each of these carbons is attached to a bromine atom, a hydrogen atom, a methyl group, and the rest of the butane chain. So, we've got our stereocenters identified.
Now, can we draw this molecule such that it has a plane of symmetry? Yes! If you draw the molecule in a specific conformation (think of it as a 3D arrangement), you can see a plane of symmetry that bisects the molecule between C2 and C3. This plane of symmetry makes the two stereocenters mirror images of each other, effectively making the molecule achiral.
So, 2,3-dibromobutane can exist as enantiomers, but it also exists as a meso compound. If this were on a test, and the other options weren't clearly meso, this would be your answer. It’s all about that internal reflection!
Example 3: 1,2-Dichloropropane
Now, let's switch gears slightly. How about 1,2-dichloropropane? This molecule has two stereocenters: C1 and C2. C1 is bonded to two chlorine atoms, a hydrogen, and the rest of the chain. Wait a minute… C1 is bonded to two chlorine atoms. That means C1 is not a stereocenter! It's bonded to three different types of atoms/groups (Cl, H, CH(Cl)CH3), but since two of them are the same (Cl), it can't be a stereocenter. My apologies, I misspoke! Chemistry is all about precision, right? You catch my drift though, right? We need four different things!
Let's correct that. The stereocenters in 1,2-dichloropropane are C2 (bonded to H, Cl, CH3, CH2Cl) and C1. No, C1 is still not a stereocenter because it's bonded to two Cl atoms. My apologies again! The stereocenter is C2. It is bonded to H, Cl, CH3, and CH2Cl. That's four different groups! So, 1,2-dichloropropane has one stereocenter at C2. A molecule with only one stereocenter is always chiral. Therefore, 1,2-dichloropropane cannot be a meso compound. Phew! Glad we cleared that up. Always double-check those groups!
This highlights a crucial point: a meso compound must have at least two stereocenters. If a molecule only has one stereocenter, it's chiral, period. No amount of symmetry can save it from being chiral. It’s like trying to find a symmetrical pickle when you only have one half of a pickle to begin with. Doesn't work!
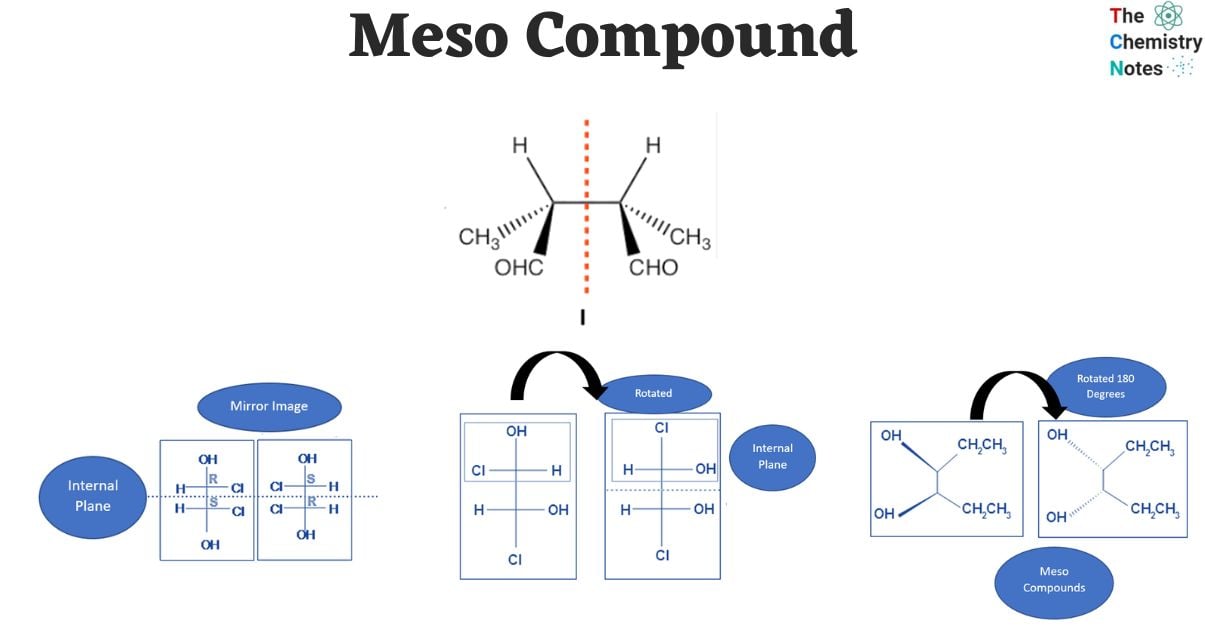
Example 4: 2,3-Dichlorobutane (The "Meso" King!)
Let's go back to a more reliable example: 2,3-dichlorobutane. This is very similar to 2,3-dibromobutane. It has two stereocenters at C2 and C3. Both C2 and C3 are bonded to a chlorine, a hydrogen, a methyl group, and the rest of the chain (which includes the other stereocenter). Again, we identify these as stereocenters.
Now, the question is, does 2,3-dichlorobutane have a plane of symmetry? Yes, it does! Just like with 2,3-dibromobutane, there's a conformation where a plane of symmetry can be drawn through the molecule, bisecting it between C2 and C3. This symmetry makes the molecule achiral, even though it possesses stereocenters. This is the definitive meso compound!
So, when you're presented with a list, and 2,3-dichlorobutane (or dibromo, or disubstituted similar structure) is among the options, you can be pretty darn sure that's your meso compound.
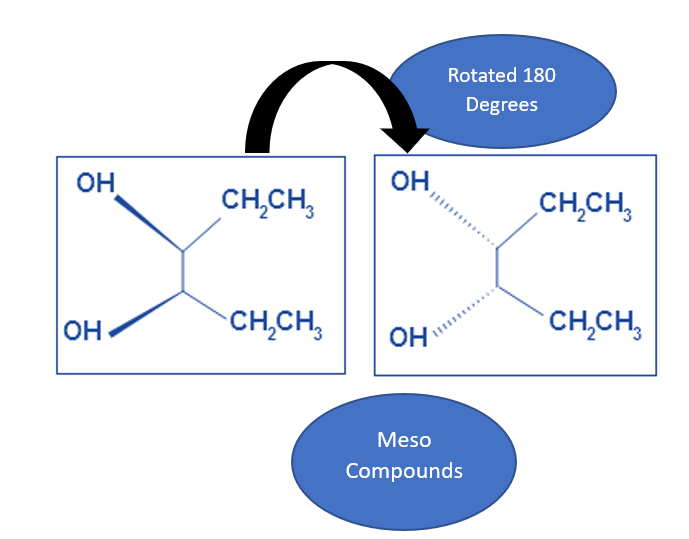
What if the molecule is symmetrical, but doesn't have stereocenters?
This is another good point to clarify. A molecule can be perfectly symmetrical and achiral without being a meso compound. For example, methane (CH4) is perfectly symmetrical, but it doesn't have any stereocenters. It’s just… methane. Meso compounds are specifically about molecules that have the potential for chirality due to stereocenters, but then have internal symmetry that cancels it out.
It's like a perfectly balanced dance troupe. Each dancer might be incredibly skilled and capable of a solo performance (chirality), but when they perform together, their movements are so perfectly synchronized and opposite that the whole troupe acts as a single, unified, symmetrical entity (achiral meso compound).
The Key Takeaway: Look for the Internal Mirror Plane!
The ultimate test for a meso compound is the presence of an internal plane of symmetry. If a molecule has two or more stereocenters, and you can draw a plane through it that divides it into two mirror-image halves, then it's a meso compound. This is the defining characteristic. Without that internal symmetry, even with multiple stereocenters, it will be chiral and exist as a pair of enantiomers.
Sometimes, these questions can be a bit of a trick. They might give you a molecule with stereocenters but no plane of symmetry, or a molecule with a plane of symmetry but no stereocenters. You have to satisfy both conditions: at least two stereocenters AND an internal plane of symmetry that relates them.
So, to recap our detective work:
- Find the stereocenters: carbons with four different groups.
- Count them: you need at least two for a meso compound.
- Look for the symmetry: can you slice it in half to get mirror images?
If you tick all those boxes, congratulations, you've found yourself a meso compound! It's a molecule that plays by its own rules, defying expectations and keeping organic chemists on their toes. And honestly, isn't that what makes chemistry so darn interesting? It’s full of these unexpected surprises, these little puzzles that make you think a little harder, and a lot more about those darn pickles.