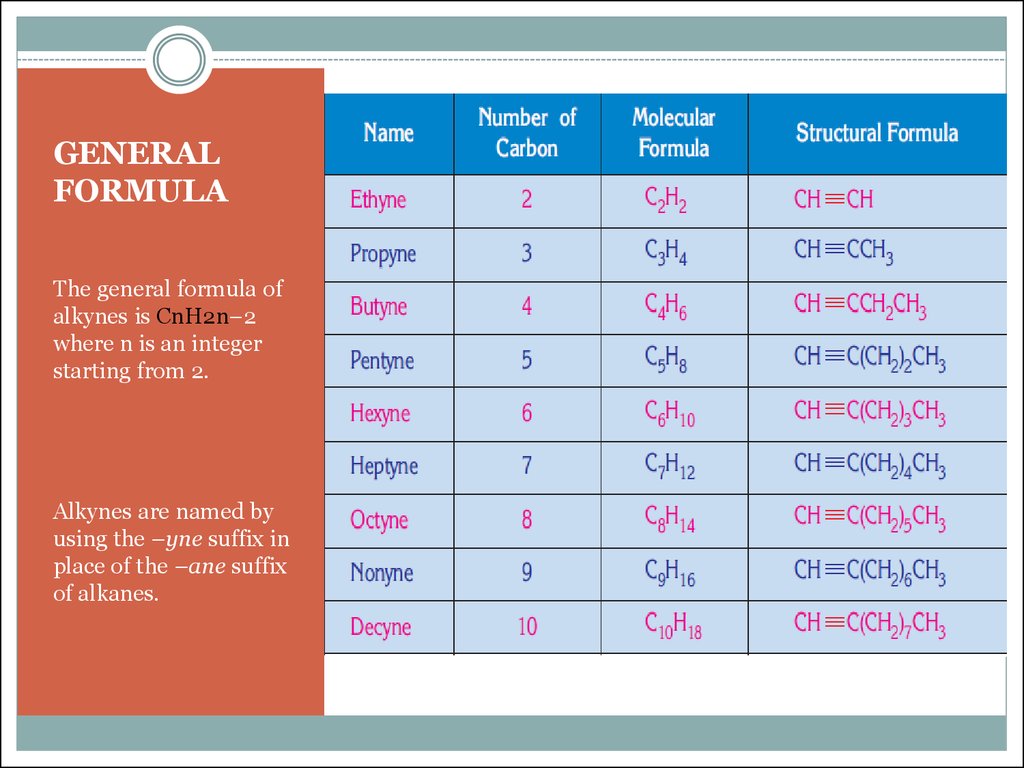
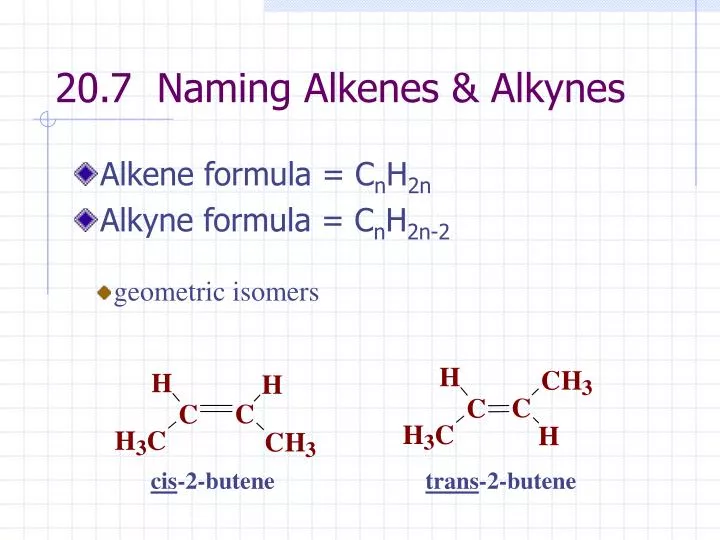
The General Formula For The Alkyne Series Is

Hey there, budding chemists and curious minds! Ever wondered about the secret handshake of alkynes? You know, those super cool hydrocarbons with a triple bond? They’ve got their own special way of being put together, like a recipe that always yields the same kind of delicious molecular dish. Today, we’re going to spill the beans on the general formula for the alkyne series. Don't worry, it's not some scary, brain-melting equation. Think of it more like a cheat code for understanding these awesome molecules.
So, let's dive in! We’re talking about alkynes, right? Remember those guys? They’re the ones rocking a triple bond between two carbon atoms. It’s like a super-strong handshake that makes them a bit more reactive than their single-bonded cousins, alkanes, or their double-bonded siblings, alkenes. They're the rebellious teenagers of the hydrocarbon world, always looking for a bit of excitement. And that triple bond? It’s the source of all their swagger.
Now, every family of organic compounds has its own way of being counted. Like, if you have a family of ducks, you count them as, well, ducks! Similarly, hydrocarbons have their own counting system based on how many hydrogens and carbons they've got. For alkynes, there’s a very neat and tidy way to figure this out. It's like a secret code that tells you exactly how many of each atom you’ll find in any given alkyne, as long as you know how many carbons there are.
Must Read
Drumroll, please! The general formula for the alkyne series is... CnH2n-2! Ta-da! See? Not so scary, is it? Let’s break this down, because understanding what this means is where the real magic happens. It’s your golden ticket to predicting the composition of any alkyne.
Here, 'n' is the star of the show. It represents the number of carbon atoms in the alkyne molecule. So, whatever number you pick for 'n', it dictates how many hydrogens you’re going to have, and not just any old number of hydrogens, but precisely twice that number minus two.

Let’s test this out, shall we? It’s always more fun to see it in action. Imagine you have an alkyne with just two carbon atoms. What would 'n' be? Yep, you guessed it: n = 2. Now, let’s plug that into our trusty formula: C2H(22 - 2). That simplifies to C2H(4 - 2), which gives us C2H2. Anyone know what C2H2 is? It's acetylene! The simplest alkyne. The OG of alkynes, if you will. It’s the founding father of this triple-bonded clan.
Let’s try another one. What if we have an alkyne with three carbon atoms? So, n = 3. Our formula becomes C3H(23 - 2). That's C3H(6 - 2), which gives us C3H4. This molecule is called propyne. See how the formula works its magic? It’s like a molecular fortune teller, predicting the hydrogen count with uncanny accuracy.
How about four carbons? n = 4. Plugging it in: C4H(24 - 2). That’s C4H(8 - 2), resulting in C4H6. This one goes by the name of butyne. You might find it existing as two different compounds, depending on where that triple bond decides to hang out, but the general formula still holds true for both! It’s like a versatile ticket that gets you into any alkyne party.

It’s super important to remember that this formula *only applies to alkynes, meaning compounds that have one carbon-carbon triple bond. If a molecule has more than one triple bond, or a mix of triple and double bonds, it’s a whole different ballgame and needs a different formula. This formula is like a VIP pass for simple alkynes, and it doesn't extend to the more complex party animals of the hydrocarbon world.
Why is this formula so useful? Well, it’s a fantastic tool for identification and prediction. If someone tells you they have an alkyne with 10 carbon atoms, you can instantly calculate the number of hydrogen atoms without breaking a sweat. Just plug in n=10: C10H(210 - 2) = C10H18. Easy peasy, lemon squeezy! You’ve just unlocked the secret identity of this alkyne.

It also helps us understand the degree of unsaturation in a molecule. That '-2' at the end of the formula is a big clue! For every 'pair of hydrogens' you're 'missing' compared to a saturated alkane (which has the formula CnH2n+2), you generally have a ring or a pi bond. In the case of alkynes, the triple bond has *two pi bonds. So, compared to an alkane with the same number of carbons (CnH2n+2), an alkyne (CnH2n-2) has 4 fewer hydrogens. That difference of four hydrogens points directly to the presence of those two pi bonds in the triple bond. It's like a forensic scientist for molecules, telling you about the structure just by looking at the numbers.
Think about it: an alkane with 'n' carbons has 2n+2 hydrogens. An alkene with 'n' carbons has 2n hydrogens (one less pi bond than an alkyne, hence two fewer hydrogens than an alkane). And our alkyne friend, with its fancy triple bond, has 2n-2 hydrogens (two pi bonds, hence four fewer hydrogens than an alkane). It’s a progression, a neat little sequence that shows how adding multiple bonds changes the hydrogen count.
So, the next time you’re faced with a hydrocarbon and you suspect it’s an alkyne, just remember that little magic phrase: CnH2n-2. It’s your secret weapon. It’s the key that unlocks the structure and properties of this fascinating class of compounds. It’s the bedrock upon which their chemical identity is built.

The beauty of this general formula is its simplicity and its universality within the alkyne family. It’s a constant, a reliable anchor in the vast ocean of organic chemistry. It allows us to predict, to classify, and to understand without needing to memorize every single alkyne out there. It's like having a universal translator for the language of alkynes.
And honestly, isn't that just fantastic? That a simple mathematical relationship can describe such diverse and important molecules? It’s a testament to the elegant order that governs the universe, even at the atomic level. It’s a reminder that even the most complex structures can be understood through fundamental principles. It's like finding a beautiful pattern in a chaotic mess, bringing clarity and order to the molecular world.
So, go forth, my friends! Armed with the knowledge of CnH2n-2, you can now confidently identify, predict, and even imagine new alkyne molecules. You’ve unlocked a fundamental secret of organic chemistry, and that’s pretty darn cool. Keep exploring, keep questioning, and remember that the world of chemistry is full of these amazing, elegant formulas just waiting to be discovered. Every time you encounter an alkyne, give a little nod to its general formula – it’s the unsung hero that holds it all together, and it’s here to help you shine!