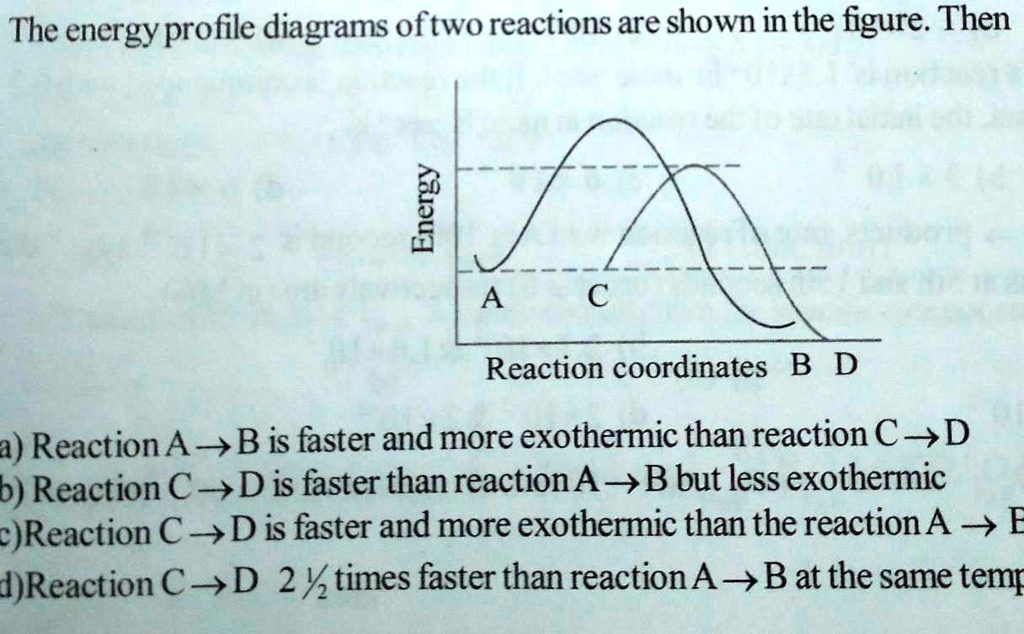
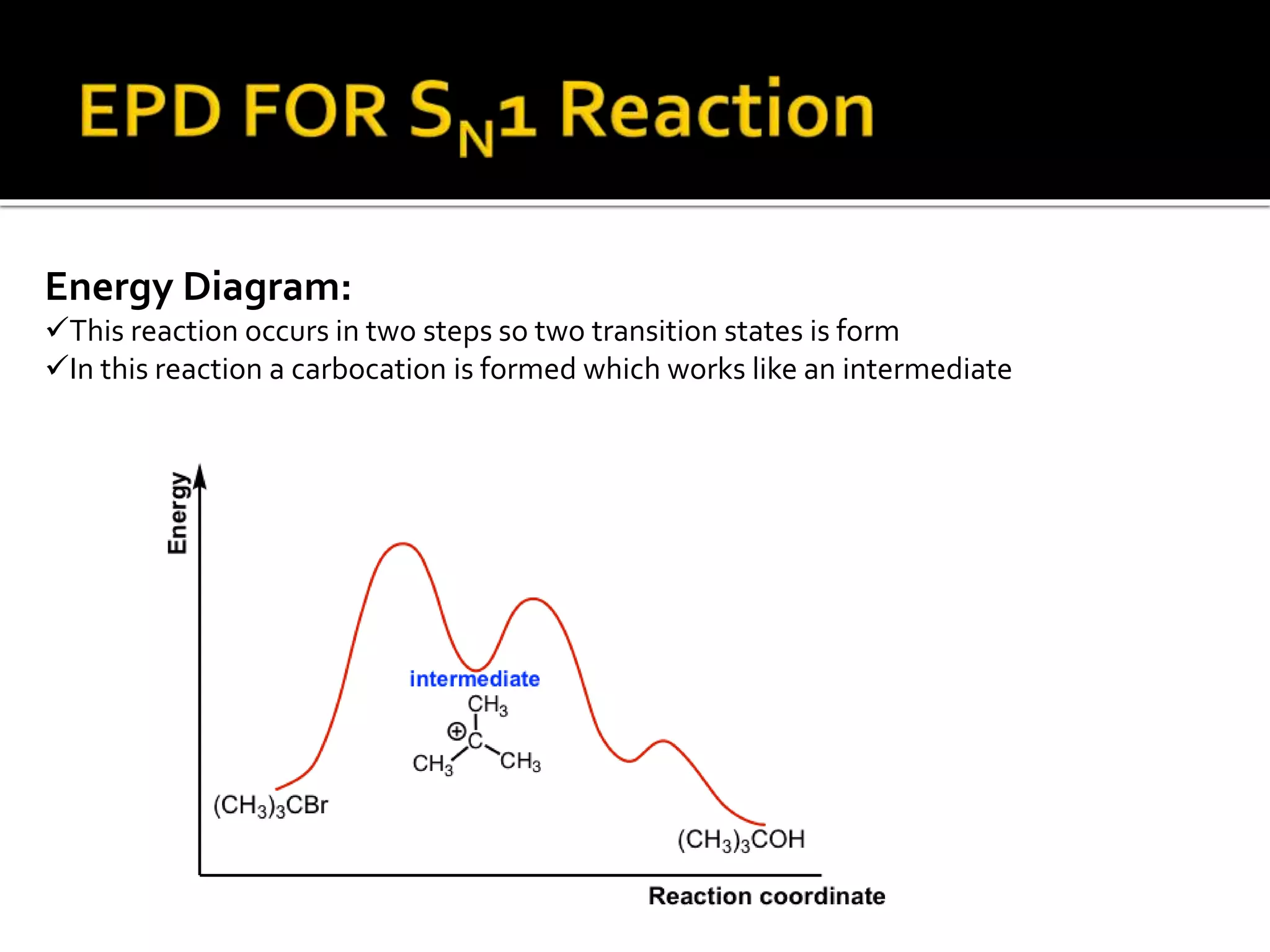
Energy Profile Diagram For Two Step Reaction
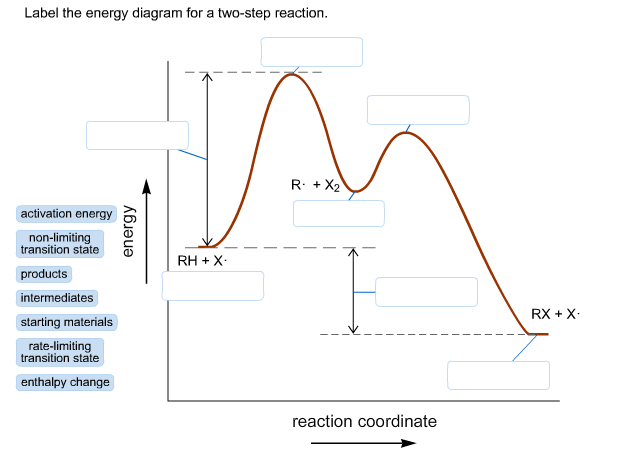
Ever feel like your life has a few distinct "acts"? Maybe you're lounging on the couch (Act 1: Relaxation Station) and then suddenly remember you need to do the laundry (Act 2: The Great Sock Expedition). Well, guess what? Chemistry has a similar, but way cooler, way of looking at how things happen: the Energy Profile Diagram for a two-step reaction! Sounds fancy, right? But trust me, it’s less about dusty textbooks and more about understanding the hidden energy rollercoaster that drives… well, pretty much everything!
So, what is this mysterious diagram? Imagine it as a visual storybook of a chemical reaction's energy journey. Think of it like climbing a hill. You start at a certain energy level, and to get to the top (where the magic happens!), you need to put in some effort, right? That effort is energy. In chemistry, we call this the activation energy. It's the "oomph" needed to get things going. For a two-step reaction, it's like tackling a mini-mountain range with a couple of peaks instead of just one.
Let's break it down. A two-step reaction isn't a single, smooth climb. Oh no, it’s a bit more dramatic! It involves intermediates. Think of an intermediate as a temporary resting spot, a little plateau between the two uphill battles. So, you climb the first hill, reach your little intermediate plateau, and then you gotta climb another hill to reach the final destination – the products of the reaction. Pretty neat, huh?
Must Read
The Peaks and Valleys of Transformation
Picture this: you're at the starting line, feeling a bit sluggish (that's your reactants). To get moving, you need to overcome the first energy hurdle – the first activation energy. This is the highest point of the first hill. Once you’ve cleared that, poof, you arrive at the intermediate. This intermediate is a bit like a quick pit stop. It’s not your final destination, but it’s a step along the way. It’s a brand new molecule formed during the reaction, and it’s usually pretty unstable. Like a juggling act, it doesn’t stick around for long!
After your brief intermediate breather, it's time for the second act. You've got to tackle the second activation energy. This is the second hill, and it might be taller or shorter than the first, depending on the specific reaction. Reaching the top of this second hill means you've finally conquered the entire process, and you’ve arrived at your products – the new things that have been created! It’s like crossing the finish line after a challenging race.

The beauty of the energy profile diagram is that it shows you all of this visually. You see the starting point (reactants), the dips and rises in energy, the little intermediate plateau, and the final landing spot (products). It’s a map of the energy landscape of a chemical change. And guess what? Understanding this map can actually make you feel smarter about the world around you. Seriously!
Why This Stuff is Actually Fun! (Yes, Really!)
Okay, I know what you might be thinking. "Energy profile diagrams? Fun? You're pulling my leg!" But bear with me. Think about your favorite recipe. Some ingredients mix easily, and others need a bit of a nudge – maybe some heat, some vigorous stirring. That "nudge" is exactly what activation energy is about! A two-step reaction is just a recipe with a slightly more involved cooking process, with a temporary concoction (the intermediate) being created before the final dish is ready.
Knowing about these energy steps can help you understand why some reactions happen super fast, while others take ages. It’s all about the height of those energy hills! If the activation energy is low, the reaction is a breeze. If it’s high, it's like trying to push a boulder uphill. Sometimes, scientists can even add catalysts. These are like super-helpful friends who come along and offer to carry part of the boulder for you. Catalysts lower the activation energy, making the reaction happen much more easily and quickly. Isn’t that fascinating? It's like having a secret shortcut!
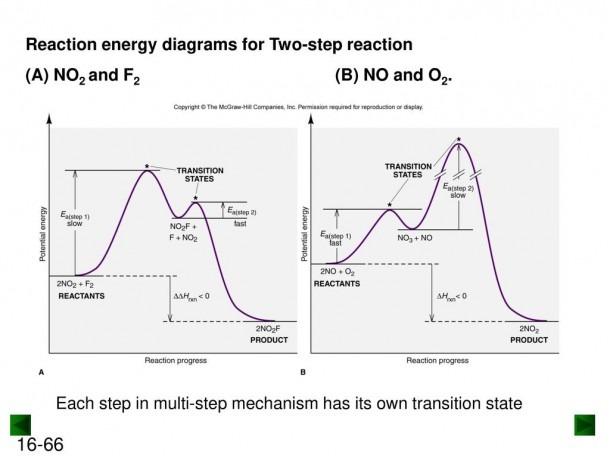
This concept isn't just for fancy labs. It’s happening in your body right now! Digestion, for instance, involves countless chemical reactions, many of them likely multi-step. Enzymes in your body are nature’s amazing catalysts, helping these reactions along without you even noticing. So, that delicious meal you enjoyed? It's all thanks to these tiny energy climbs and helpful shortcuts!
Understanding energy profiles can also spark your creativity. Imagine designing a new process for making something useful, like a new material or a cleaner fuel. By understanding the energy landscape, you can figure out how to make the process more efficient, use less energy, and be more environmentally friendly. You become a bit of an energy architect!

It's like learning a new language, but instead of words, you're learning about energy, transformation, and the hidden forces that shape our world. And once you start seeing these energy diagrams, you’ll start noticing them everywhere. They’re the unsung heroes behind everything from baking a cake to powering a car (eventually!).
So, the next time you hear about a chemical reaction, don’t just think of bubbling beakers. Think of the energetic journey, the mini-mountains, the temporary stops, and the final triumph of creating something new. The energy profile diagram for a two-step reaction is more than just a scientific tool; it's a window into the dynamic, ever-changing nature of matter. It shows us that progress, even in chemistry, often involves a series of steps, and with a little bit of energy and a smart approach, we can overcome challenges and reach our goals.
So, why not dive a little deeper? Explore these diagrams, learn about activation energy, and discover the magic of intermediates. You might just find that understanding these fundamental concepts opens up a whole new, exciting way of looking at the universe. The world is full of energy transformations just waiting to be understood, and you’ve got the power to be curious and learn more!