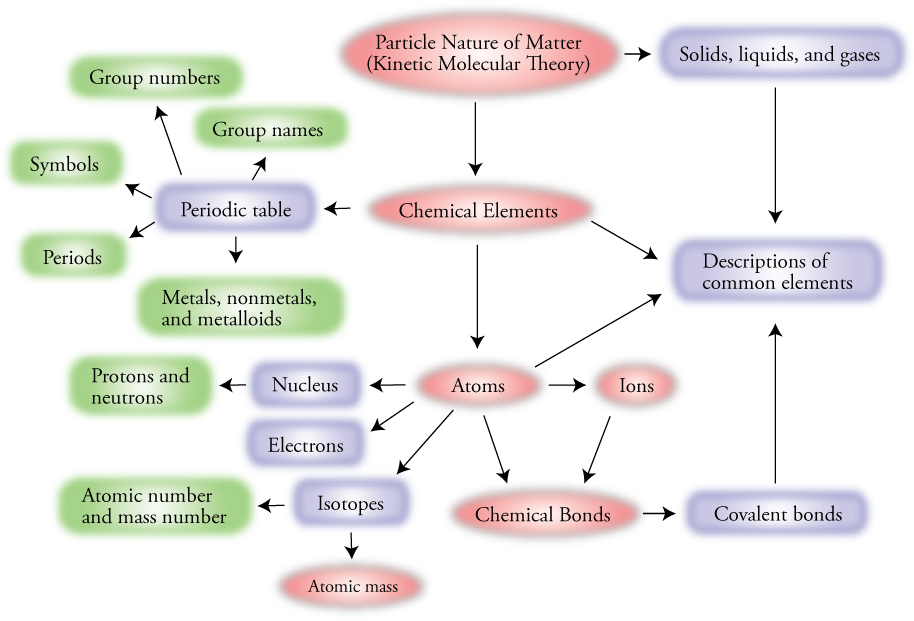
Atoms Have Subatomic Particles Concept Map
You know, sometimes I look at a giant, imposing skyscraper and think, "Wow, that thing is built out of something pretty hefty." Or I watch a strong weightlifter hoist a ridiculous amount of metal and marvel at their sheer solidity. We tend to think of things as solid, as ultimate building blocks. But here's a secret, a bit like finding out your favorite superhero has a secret identity involving knitting and collecting stamps: even the most solid-seeming stuff is actually made of even tinier, even more fundamental bits. And that, my friends, is where our journey into the wonderfully weird world of subatomic particles begins!
Think of it like a really, really fancy nesting doll. You've got your big doll, and then inside that, a slightly smaller one, and then another, and another, until you get to the itty-bitty, microscopic ones that make the whole thing possible. Atoms are kind of like those bigger dolls. They're the basic units of chemical elements, the LEGO bricks of the universe, if you will. You can't really break down gold into anything simpler and still have it be gold, right? So, the atom was for a long time considered the ultimate deal.
But then, scientists, bless their curious little hearts, started poking around. They were like those toddlers who just have to see what's inside the toy. And what they found was that atoms themselves weren't the end of the line. Oh no. They were just another layer in the grand cosmic nesting doll.
Must Read
The Atom: Not So Solitary After All
So, we've got these atoms. They're pretty cool. They do all sorts of things – they bond together to make molecules, which then make up everything from your morning coffee to your pet hamster. But inside this seemingly indivisible atom, there's a whole miniature solar system going on. And the planets in this solar system are our subatomic particles.
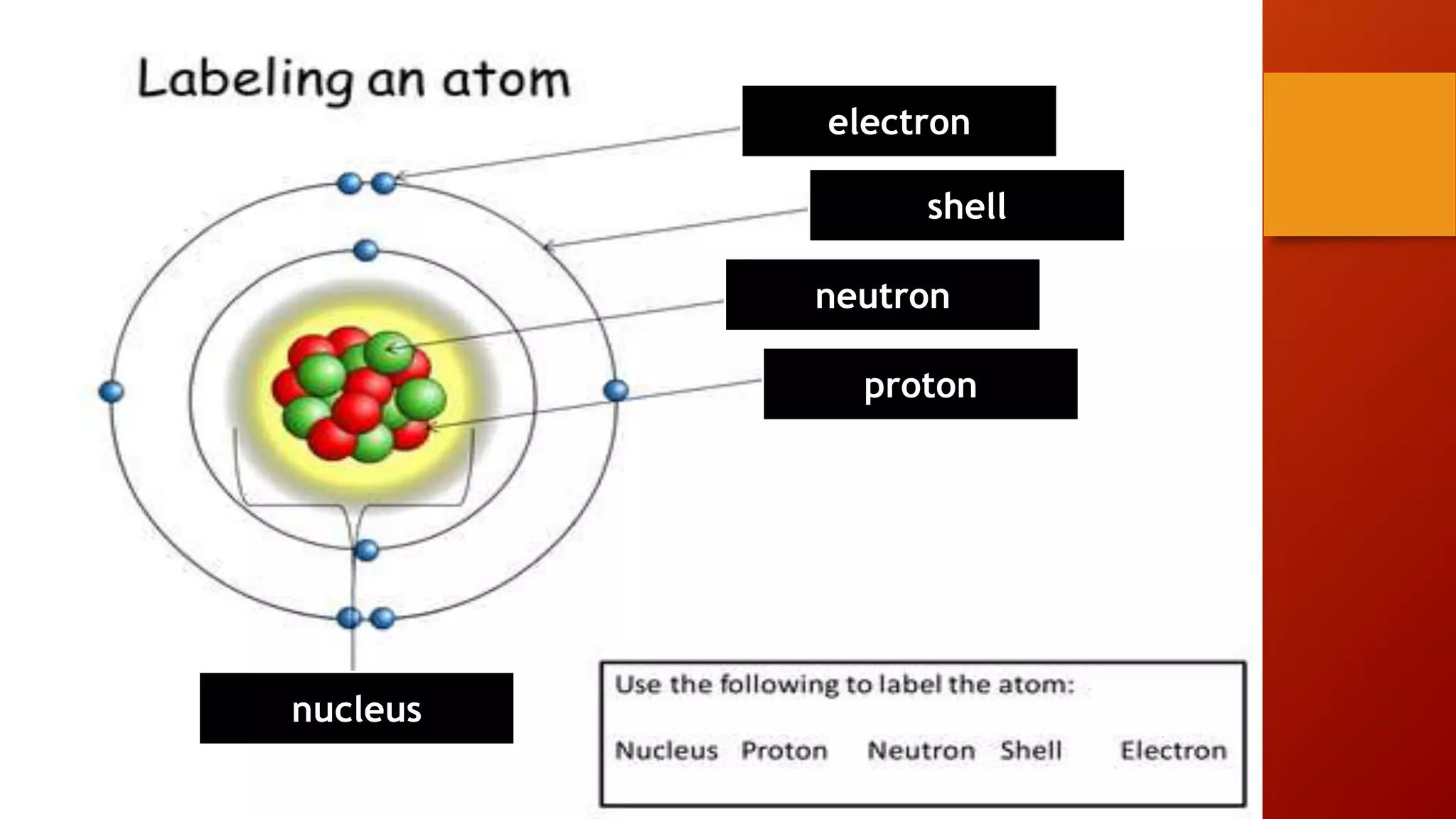
Imagine the atom as a tiny city. You've got the bustling downtown area, the outskirts, and all the transportation systems. The most famous residents of this atomic city are the protons and neutrons, who hang out together in the very center, like the mayor and the chief of police having a meeting in city hall. This central hub is called the nucleus. It's dense, it's important, and it's where most of the atom's weight is concentrated. Think of it as the all-you-can-eat buffet of the atom – heavy on the carbs and protein.
And then, whizzing around this nucleus, like hyperactive teenagers on skateboards or maybe like those drones delivering your pizza way too fast, are the electrons. These guys are way smaller, way lighter, and they've got a bit of a reputation. They're always on the go, always zipping around, and they carry a negative electric charge. Protons, on the other hand, are like the calm, collected elders of the nucleus – they've got a positive charge. Neutrons, bless their neutral hearts, are like the quiet observers; they have no charge at all. They're the folks who just show up to the party and don't really say much, but they're there.
The Proton: The Positive Guy
Let's talk about protons for a sec. These guys are the backbone of the element's identity. If you have six protons, you're carbon. If you have one proton, you're hydrogen. It's like their social security number, but way more important for the universe. They're positively charged, and this positive vibe is what attracts those zippy electrons. It's a cosmic dance, really. The positive nucleus is like a popular cafe, and the negative electrons are the customers lining up to get in. It’s a constant give and take, a fundamental attraction.

Think of it this way: you know how sometimes you meet someone, and they just have this magnetic personality? They draw people in? Protons are kind of like that, but with a scientific twist. Their positive charge is their inherent charm, their irresistible pull. Without protons, you wouldn't have the elements as we know them. No carbon means no diamonds, no trees, and definitely no chocolate. You'd be staring at a very boring, very empty universe. So, props to the protons for keeping things interesting.
The Neutron: The Unassuming Mediator
Now, the neutrons. They're the quiet ones in the nucleus. They don't have a charge, which makes them kind of neutral, obviously. But don't let their lack of electric personality fool you. They're super important for keeping the protons from getting too riled up and flying apart. Imagine a crowded party where everyone's got strong opinions (those are the protons, with their positive charges pushing each other away). The neutrons are like the friendly bouncers, or maybe the chill DJs, keeping the peace and making sure the party doesn't get too chaotic. They add mass to the nucleus, making it more stable, like adding extra ballast to a ship so it doesn't capsize in a storm.
Sometimes, atoms can have different numbers of neutrons but still be the same element. These are called isotopes. It's like having twins who look almost identical but one has a slightly different haircut. They're still fundamentally the same person, but with a little variation. For example, carbon-12 is the common one, but carbon-14, with an extra neutron, is famous for radioactive dating. It's proof that even a tiny difference can have a big impact. So, while neutrons might seem like the background characters, they play a crucial role in the atomic drama.
The Electron: The Speedy Social Butterfly
And then we have the electrons. Oh, the electrons! These little dynamos are the real movers and shakers when it comes to how atoms interact. They're negatively charged and orbit the nucleus. They're so light, they're practically ghosts. If the nucleus is a bowling ball, the electrons are like tiny gnats buzzing around it from a mile away. Their speed is incredible, and they occupy this fuzzy region around the nucleus called an electron cloud. It's not like they're orbiting in neat little circles like planets; it's more like they're everywhere and nowhere at once, a probability dance.

The number and arrangement of electrons are what dictate how an atom will behave chemically. They're the ones who get handed around during chemical reactions. When two atoms decide to team up and form a molecule, it's usually their electrons doing the heavy lifting. They either share electrons, or one atom might totally steal an electron from another. It’s like a cosmic dating app, with electrons being the most sought-after partners. They determine if you're going to have a strong bond, like superglue, or a weaker one, like sticky notes.
Think about how some people are super outgoing and love to interact with everyone, and others are a bit more reserved. Electrons are kind of like that. Some atoms have a full outer shell of electrons, making them content and unreactive, like someone who's already had two plates of Thanksgiving dinner and is absolutely done. Other atoms have just one or two electrons in their outer shell, and they're desperate to share or give them away to achieve a more stable configuration. They're the life of the chemical party, always looking for a dance partner.
Beyond the Big Three: A Deeper Dive
Now, here’s where things get really interesting, and you might need to put on your thinking caps, or maybe just a really cool hat. Scientists, in their never-ending quest for knowledge (and probably a few Nobel Prizes), discovered that protons and neutrons aren't the ultimate basic particles either! Nope, they're made of even tinier things called quarks.
Quarks are like the sprinkles on the atomic donut. You have different flavors: up quarks and down quarks are the most common ones found in protons and neutrons. A proton is made of two up quarks and one down quark. A neutron is made of one up quark and two down quarks. It's like a secret recipe that makes up the building blocks of the building blocks. And these quarks are held together by something called gluons. The name is perfect, right? They literally glue the quarks together, with an immense amount of force.
It's kind of mind-blowing to think that the solid desk you're probably leaning on, or the phone you're holding, is fundamentally made of these incredibly small, vibrating entities called quarks, held together by gluons. It's like realizing your favorite sturdy chair is actually a carefully orchestrated ballet of tiny dancers held by invisible ribbons. It makes you appreciate the sheer complexity and elegance of the universe, even if it does give you a slight headache trying to picture it all.
The Concept Map: A Visual Brainstorm
So, how do we wrap our heads around all of this? This is where a concept map comes in handy. Imagine it as a visual roadmap for your brain. You start with a central idea, like "Atoms," and then you branch out to related concepts. You connect them with lines and words that explain the relationship.
In our case, the central concept is Atoms. From there, we can branch out to their main components: Nucleus and Electrons. Then, within the Nucleus, we have Protons and Neutrons. And here’s the kicker: from Protons and Neutrons, we can branch out again to Quarks, and from Quarks, we can connect to Gluons. And for electrons, while they're considered fundamental (meaning we don't think they're made of smaller things yet), their interaction is key to Chemical Bonding and forming Molecules.
You can even add more branches. For example, Protons have a positive charge. Neutrons have no charge. Electrons have a negative charge. Quarks have fractional charges. Gluons bind quarks. Electrons orbit nucleus and are involved in chemical reactions. It's like building a super-detailed family tree, but for the fundamental pieces of reality. It helps you see how everything fits together, from the biggest atom to the smallest quark.

It’s the scientific equivalent of a really good whiteboard session. You throw all your ideas out there, connect them, see the patterns, and suddenly, something that seemed incredibly complex becomes a little more manageable, a little more understandable. It's a way to organize the chaos and find the elegant order that underlies it all.
Why Does This Even Matter?
You might be asking, "Okay, this is cool and all, but why should I care about quarks and gluons?" Well, my friend, this isn't just abstract science talk. Understanding these subatomic particles is the bedrock of so much of modern life. From the lasers in your barcode scanner to the microchips in your phone, from the medical imaging techniques that save lives to the nuclear power that lights up cities – all of it is a direct result of understanding the behavior of these tiny, tiny particles.
It's like understanding how individual ingredients work together to make a delicious cake. You can't just throw flour and eggs together and expect a masterpiece. You need to know how they interact, what temperature to bake them at, and the right proportions. Similarly, scientists use their knowledge of subatomic particles to engineer new materials, develop new technologies, and solve some of the world's biggest challenges. It’s the ultimate scientific engineering, built on the smallest of scales.
So, the next time you look at something solid, remember that it's not quite as simple as it appears. It's a bustling, energetic world of subatomic particles, a cosmic dance of protons, neutrons, electrons, and even the even smaller quarks and gluons. It’s a testament to the fact that there’s always more to discover, always more to understand, and that the universe, even at its most fundamental level, is a place of incredible wonder and complexity. And that, my friends, is something to smile about. Now, if you'll excuse me, I think I need a snack. All this talk of building blocks has made me hungry!





