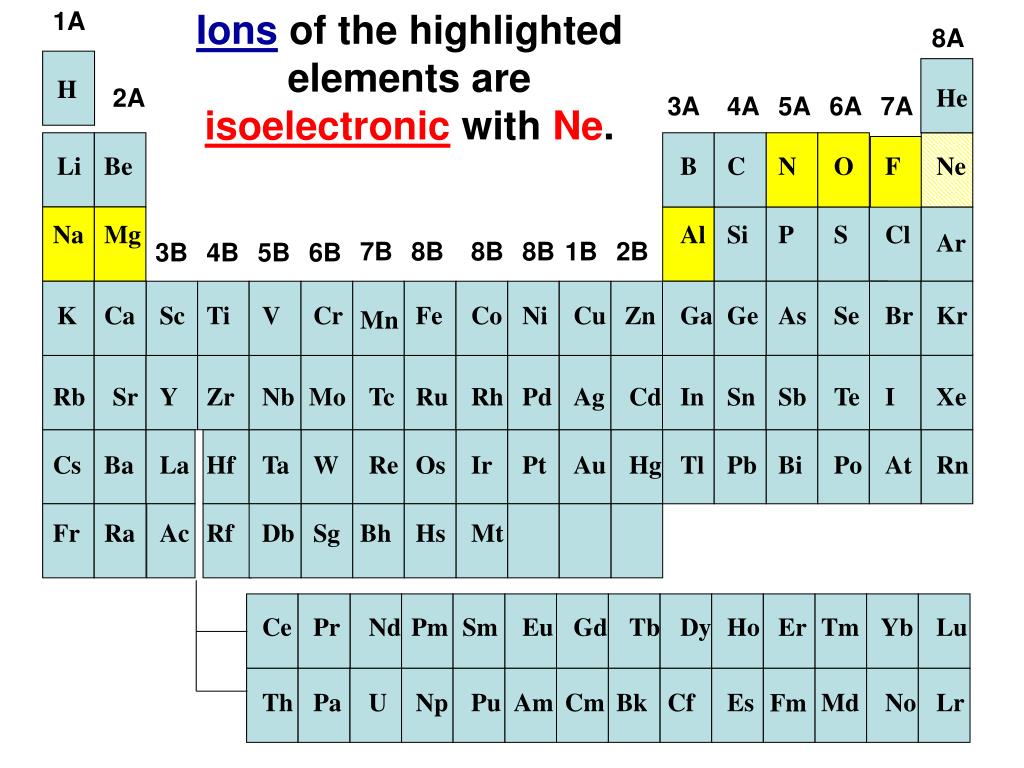
Which Neutral Atom Is Isoelectronic With N-3
Hey there, science pals! Ever feel like atoms are just, like, these tiny, boring little specks? Nah! They're actually super dramatic and love playing dress-up. Today, we're diving into a seriously fun game of "Who's Who?" in the atomic world. Get ready to meet our star: N-3. Sounds like a secret agent, right?
So, what's the deal with N-3? That little minus sign and the "3" are like N-3's party trick. It means Nitrogen has gone and snagged three extra electrons. Think of it like finding three super cool accessories you didn't expect to get with your outfit. Now, Nitrogen is feeling extra charged up, literally!
But here's the juicy gossip: N-3 has a secret identity. It's looking for a neutral atom buddy who's got the exact same electron count. This is where the super cool science term "isoelectronic" comes in. It's just a fancy way of saying "same number of electrons." Like twins, but with electron numbers!
Must Read
Why is this fun? Because it's a cosmic scavenger hunt! We're hunting for another atom, a neutral one (meaning no charges, no extra electrons or missing ones), that has pulled off the same electron trick as N-3. It's like finding a doppelganger in the universe of matter.
The Nitrogen Lowdown
First, let's get to know our Nitrogen. In its natural, chill, neutral state, Nitrogen (symbol N) is hanging out with 7 protons. And because it's neutral, it also has 7 electrons. Easy peasy. It’s just chilling, minding its own business, having 7 electrons like a normal atom.
But remember our N-3? It’s not chill. It's grabbed 3 extra electrons. So, take those original 7 electrons and add 3 more. That’s 10 electrons total for N-3! This is our magic number. We're on the lookout for a neutral atom that also boasts a grand total of 10 electrons.
The Electron Count Challenge!
So, our mission, should we choose to accept it (and we totally should, it's fun!), is to find a neutral atom with 10 electrons. How do we find that? Well, in a neutral atom, the number of electrons is always equal to the number of protons. That's the golden rule of neutrality!

Therefore, our target neutral atom must have 10 protons. Every element has a unique number of protons, called its atomic number. So, we're basically asking: "Which element has an atomic number of 10?" It's like a cosmic census!
Drumroll, Please...
Let's scan the periodic table. We're looking for that element with the atomic number 10. What pops up? Drumroll please... it's Neon! Yep, good old Neon (symbol Ne).
Neon, the stuff that makes those cool, glowing signs? That's our winner! In its natural, neutral state, Neon has 10 protons. And because it's neutral, it also has exactly 10 electrons.
The Isoelectronic Squad!
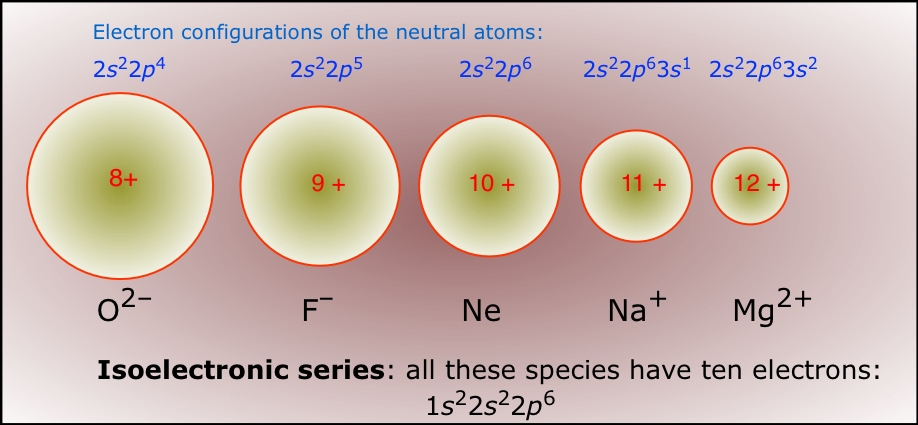
So, there you have it! Neon (Ne), the neutral atom, is isoelectronic with Nitride ion (N-3). They both have 10 electrons! They're like secret twins, both rocking that same electron configuration. How cool is that?
Think about it: N-3 is a charged particle, an ion. It's got that negative vibe going on. But Neon? It's a neutral atom. It's all balanced and proper. Yet, electron-wise, they're playing on the same team. It's a neat little quirk of the atomic world.

Why is this so cool?
Because it shows us how atoms can "cheat" a little. Nitrogen is normally a bit reactive, always looking to share or grab electrons. But when it grabs three, it becomes like Neon, which is super stable and unreactive. It's like Nitrogen is saying, "You know what? Being like Neon is pretty sweet!"
Neon, on the other hand, is famous for being a noble gas. This means it's super content and doesn't like to react with much. It's got a full outer shell of electrons, making it very happy and stable. It’s the ultimate chill atom, basically. Like the queen bee of stability.
So, when Nitrogen becomes N-3, it achieves that same super-stable, full outer electron shell that Neon enjoys. It's like Nitrogen is temporarily borrowing Neon's incredibly popular fashion sense and its guaranteed spot at the "cool kids' table" of atoms.
Quirky Facts & Fun Details
Did you know Neon gets its name from the Greek word "neos," meaning "new"? It was discovered in 1898. Imagine finding a whole new element that glows! That's pretty neat.

And those iconic Neon signs? They work because when electricity passes through Neon gas, it excites the electrons. When those excited electrons calm down, they release energy in the form of light. And in Neon's case, that light is usually a bright, vibrant red-orange. Other gases make different colors, but pure Neon is all about that fiery glow!
So, the next time you see a bright Neon sign, you can think, "Hey! That's the atom that's like N-3 in disguise!" It's a little piece of hidden atomic chemistry right there in plain sight.
This whole isoelectronic idea is a big deal in chemistry. It helps us understand how different atoms and ions behave. If two species have the same electron configuration, they're likely to have similar chemical properties in certain situations. It's like recognizing that even though two people are from different families, they might have the same favorite ice cream flavor. They're still individuals, but they share something in common.
It's not just Nitrogen. Lots of ions and neutral atoms can be isoelectronic. For instance, O-2 (Oxygen with 2 extra electrons) is also isoelectronic with Neon! Oxygen has 8 protons, so O-2 has 8+2=10 electrons. Another friend for Neon in the electron club!
And get this: F-1 (Fluorine with 1 extra electron) also has 10 electrons! Fluorine has 9 protons, so F-1 has 9+1=10 electrons. Wow, Neon is super popular! It's like the celebrity that everyone wants to be like.

The element before Neon, Fluorine (atomic number 9), when it gains one electron, becomes like Neon. The element after Neon, Sodium (atomic number 11), when it loses one electron (becomes Na+1), also becomes like Neon! See the pattern? It’s all about achieving that stable, 10-electron shell.
So, Is It Fun?
Absolutely! This is like a fun puzzle. You're given a clue (N-3 with its 10 electrons) and you have to find the matching piece (a neutral atom with 10 electrons). It's a little detective work, a little bit of number crunching, and a whole lot of "aha!" moments.
It’s a reminder that even though atoms can look simple on the surface, they have this incredible complexity and these fascinating relationships. They’re not just static building blocks; they’re dynamic entities playing a constant game of electron exchange and stability-seeking.
So next time someone mentions an ion and you’re curious about its neutral buddies, just remember the magic number of electrons. And always keep an eye out for those noble gases – they’re the ultimate trendsetters in the atomic world!
Keep exploring, keep wondering, and keep having fun with science!