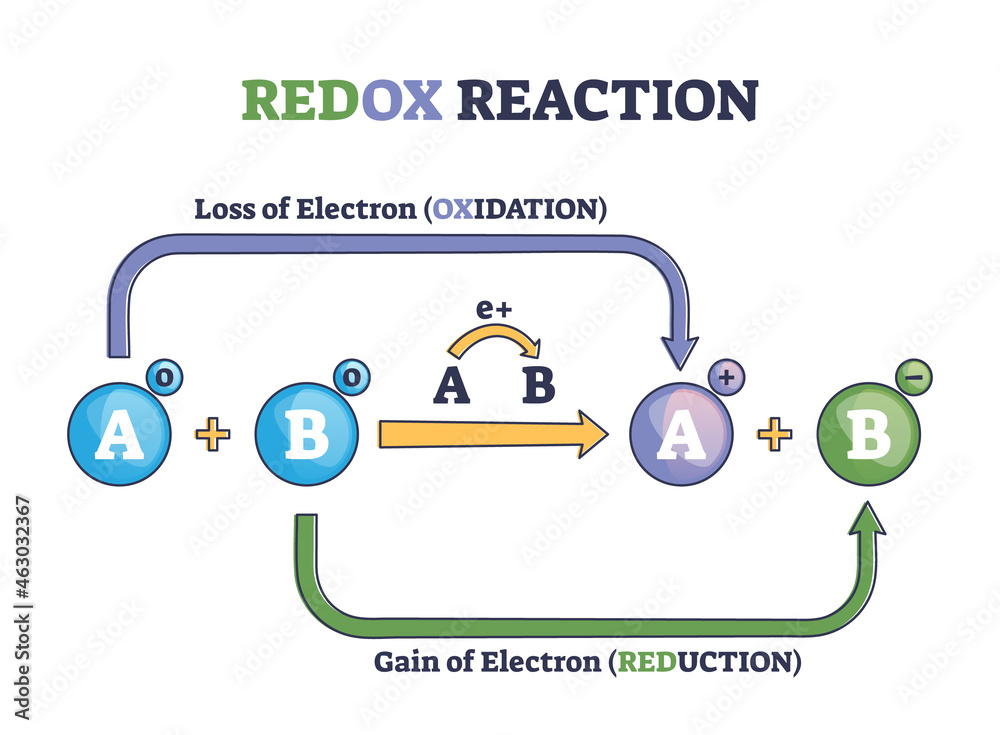
Identify The Oxidation State Of Cl In Clf+4
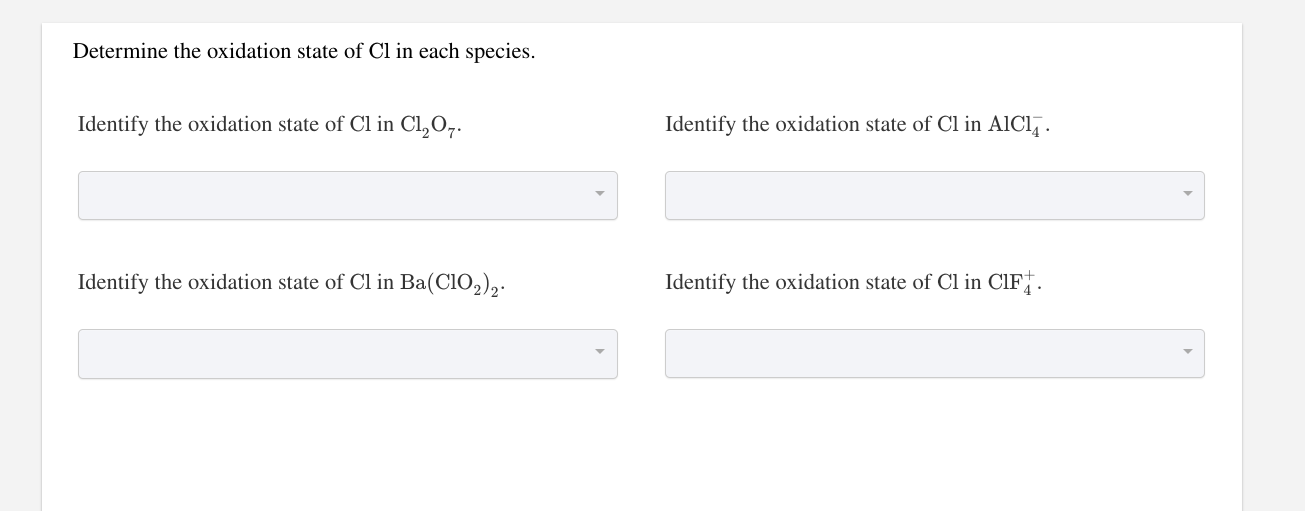
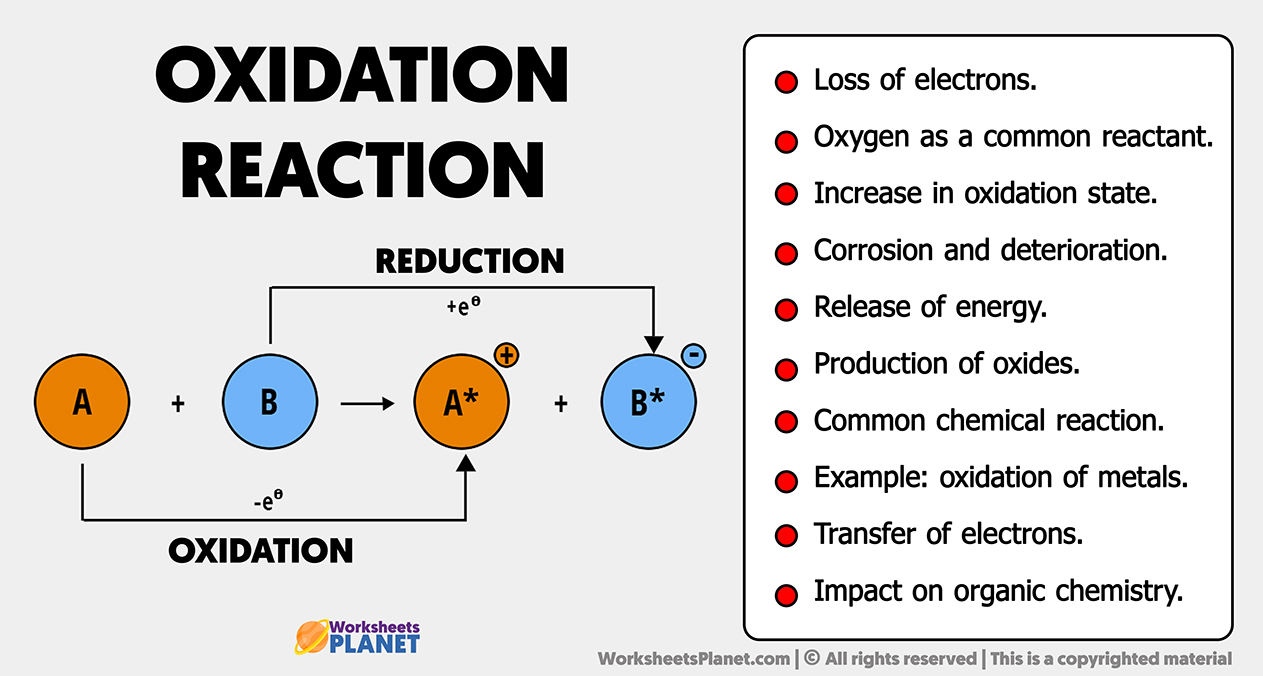
Have you ever felt a spark of curiosity, a desire to unravel a tiny mystery that dances just beyond your immediate grasp? Well, prepare to be delighted! Today, we're diving into a world that, while sounding quite scientific, offers a surprising amount of creative potential and a wonderful sense of accomplishment, even for the most casual learner. We're talking about the fascinating realm of identifying oxidation states, and we'll be focusing on a particularly intriguing example: Cl in ClF4+.
Now, you might be thinking, "Oxidation states? That sounds complicated!" But here's the exciting part: understanding these fundamental chemical concepts can be incredibly rewarding. For artists, it can unlock new ways to think about color mixing and material interactions. For hobbyists, it's a gateway to understanding more complex chemical processes, from baking to gardening. And for casual learners, it’s a chance to experience the thrill of solving a small puzzle, building confidence and a sense of intellectual engagement.
Think of it like this: just as an artist chooses specific pigments to create a desired effect, chemists determine oxidation states to understand how elements will behave. In the case of ClF4+, we're looking at a positively charged molecule where chlorine (Cl) is bonded to four fluorine (F) atoms. The beauty of this exercise is that it encourages us to think logically and systematically. You'll learn to apply simple rules, much like following a recipe or a pattern in a craft project. It’s a process of deduction that’s both engaging and deeply satisfying.
Must Read
Imagine the satisfaction of looking at this formula and, through a few straightforward steps, arriving at the precise charge of the chlorine atom. It’s a moment of clarity, a small victory in understanding the language of molecules. This isn't about memorizing complex equations; it's about grasping a core principle that underlies so much of the chemical world around us.

Ready to give it a try? For ClF4+, the key is to remember that fluorine is almost always assigned an oxidation state of -1 when it's in a compound with another element (except for other halogens in certain circumstances). Since the overall charge of the ion is +1, and we have four fluorine atoms each contributing -1, we can use this to find the oxidation state of chlorine. You'll be surprised at how quickly you can solve it!
The real joy in identifying oxidation states, especially with fun examples like ClF4+, lies in its accessible complexity. It’s like unlocking a hidden door to understanding. You don't need a full laboratory; just a curious mind and a willingness to explore. So, the next time you encounter a chemical formula, don't shy away – embrace the puzzle! You might just find yourself enjoying the process of discovery more than you ever imagined.