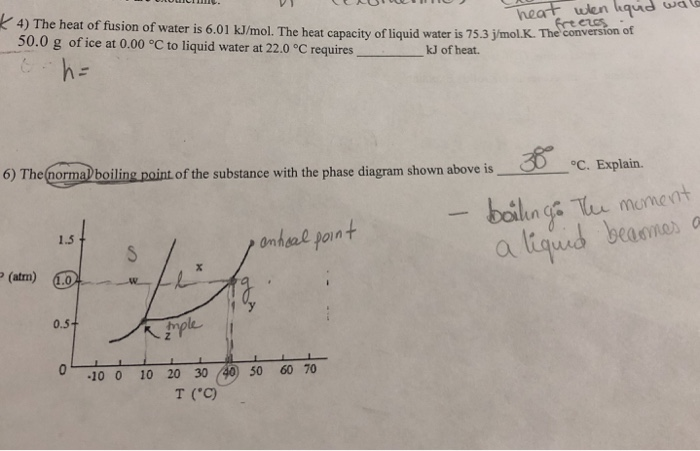
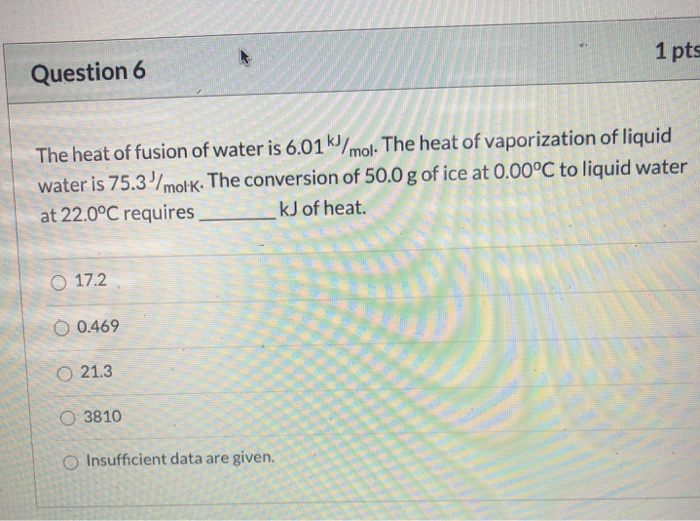
The Heat Of Fusion Of Water Is 6.01 Kj Mol

Ever wondered why ice cubes make your drink so refreshingly cold, or why steam can be so powerful? It all comes down to a fascinating property of water: its heat of fusion. This isn't some scary, complicated science term meant only for chemists in lab coats. In fact, understanding a little about the heat of fusion of water, which is approximately 6.01 kilojoules per mole, can be surprisingly fun and even useful in our everyday lives!
So, what exactly does "heat of fusion" mean in plain English? Simply put, it's the amount of energy needed to change water from a solid (ice) to a liquid at its melting point, without changing its temperature. Think of it as the energy it takes to break those tiny ice bonds and let the water molecules start flowing. And that 6.01 kJ/mol figure? It's a measure of that energy. It might sound small, but when you consider how much water is around us, it adds up!
Why should you care? For beginners exploring the world of science, it's a fantastic gateway into understanding phase changes – how things go from solid to liquid, liquid to gas, and back again. For families, it can spark curiosity in kids about why ice melts so slowly on a hot day or why a steaming cup of tea can keep your hands warm for ages. Hobbyists, especially those interested in things like making ice cream from scratch or even understanding how certain crafts work with temperature, will find this knowledge incredibly practical.
Must Read
Let's look at some everyday examples. When you add ice to your drink, it doesn't just get colder immediately. The ice absorbs a lot of heat from the drink to melt, which is why your beverage stays chilled for so long. That's the heat of fusion at work! Think about how much energy it takes to melt a big block of ice compared to just warming up a cup of cold water. Or consider steam burns – they are often more severe than hot water burns because steam also releases its latent heat of vaporization (a related concept!) when it condenses on your skin, in addition to its initial heat.
Want to get a feel for this? Try a simple experiment at home. Put a thermometer in a glass of ice water. You'll notice the temperature stays right around 0°C (32°F) until all the ice has melted. That plateau is the heat of fusion doing its thing! Another fun variation is to see how long it takes for different amounts of ice to melt in the same amount of warm water. You'll see that more ice means more heat is absorbed, and the water stays colder for longer.
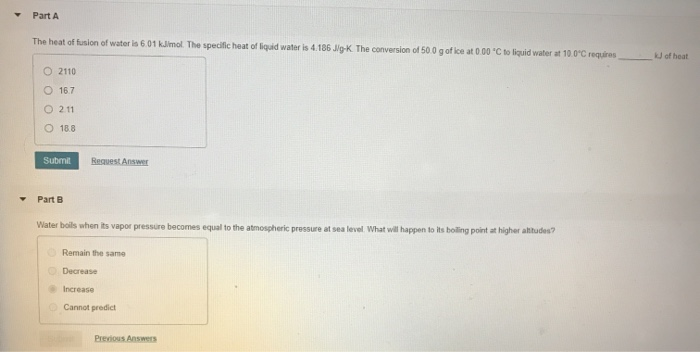
Getting started is easy. You don't need fancy equipment. A glass, some ice cubes, a thermometer, and a little bit of curiosity are all you need. Observe how things change, ask "why," and perhaps do a little online searching about melting points and phase changes. It’s a wonderful way to connect with the physical world around us.
So, the next time you enjoy a cold drink or watch snow melt, remember the heat of fusion of water. It's a simple scientific principle that makes our world cooler, warmer, and infinitely more interesting. It’s a small piece of knowledge with a big impact, and exploring it is a genuinely enjoyable experience!