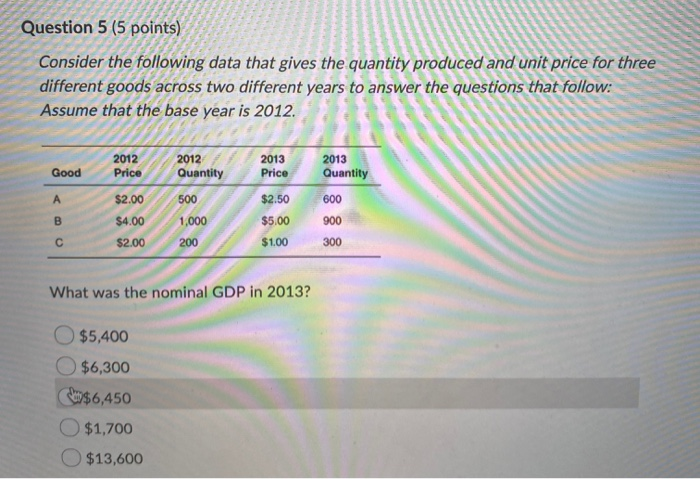
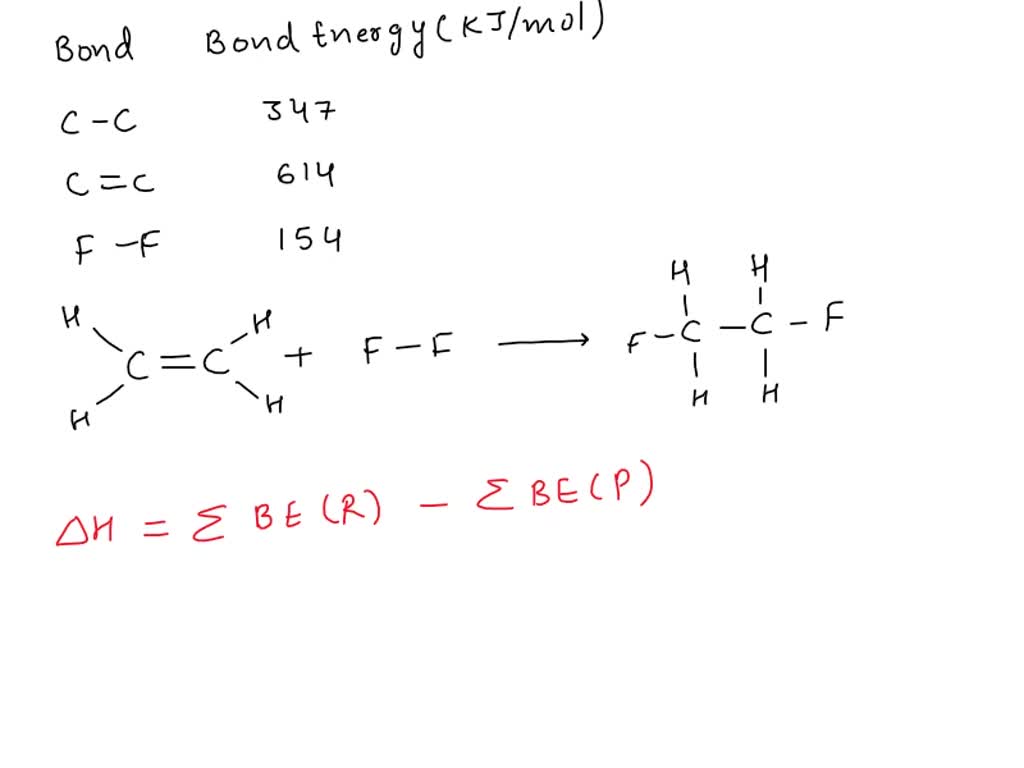Consider The Following Data For Fluorine:
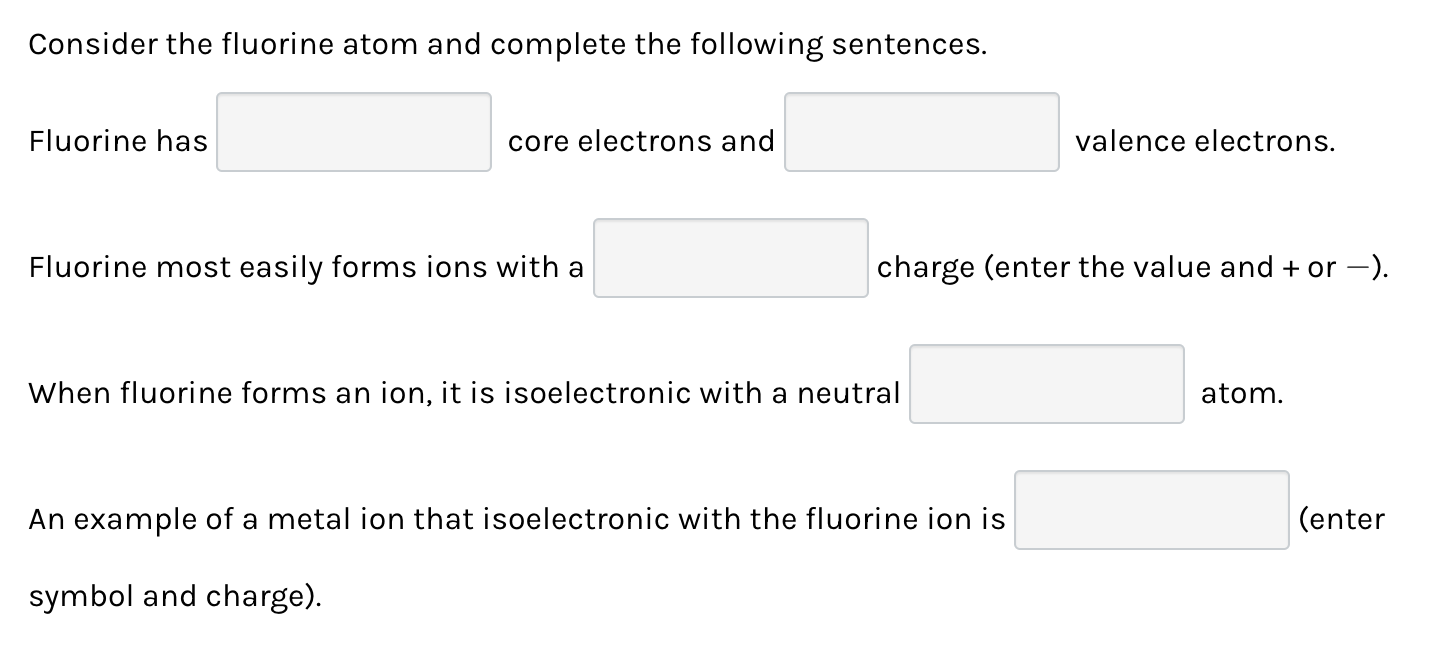
Alright, gather 'round, folks, and let's have a little chat about something that sounds way more boring than it actually is. We're talking about fluorine. Yeah, I know, I know, "fluorine? Is this going to involve a pop quiz on the periodic table?" Relax, my friends, because this is less textbook and more like eavesdropping on a particularly wild chemical gossip session. Think of me as your friendly neighborhood element whisperer, spilling the tea on this feisty little character.
So, what's the big deal with fluorine? Well, imagine the periodic table as a giant family reunion. You've got your chill, easy-going elements, your a bit-too-loud uncles, and then you've got fluorine. Fluorine is that relative who walks into the room, grabs everyone's attention, and probably causes a minor incident. This guy is the most electronegative element out there. What does that even mean, you ask? It means fluorine is a total drama queen when it comes to electrons. It wants them. It really wants them. It's like the ultimate electron hoarder, snatching them up from anything it can get its tiny, invisible hands on.
Seriously, this electron-grabbing isn't just a hobby; it's practically fluorine's entire personality. It’s so eager to grab electrons that it’s practically the most reactive element on the block. Think of it this way: most elements are happy to share their electrons or take one or two. Fluorine is like, "Hand 'em over! All of them! And while you're at it, can I have your car keys and that artisanal cheese you brought?" It’s a bit much, to be honest.
Must Read
Because of this intense need for electrons, fluorine doesn’t really hang out by itself. You won't find a lonely fluorine atom chilling in a gas cloud. Nope. It’s always partnered up, usually with another fluorine atom to form F₂, or, more commonly, it's busy bonding with everything else. It’s the ultimate social butterfly, but in a slightly terrifying, "I will consume you" kind of way. It’s like that friend who’s super enthusiastic about joining your friend group, but then ends up accidentally stealing everyone’s snacks and wearing all their favorite sweaters.
Now, here's where things get interesting, and maybe a little bit mind-bending. This super-reactive, electron-snatching nature actually makes fluorine incredibly useful. You'd think something that wants to react with everything would be a recipe for disaster, right? And sometimes, it is! But controlled fluorine is like a highly trained, slightly unhinged superhero. It can do amazing things when pointed in the right direction.

For starters, think about your teeth. Yes, your pearly whites! You probably owe them a silent thank-you to our friend fluorine. When we add small amounts of fluoride (which is basically fluorine in a more polite, less aggressive form) to our toothpaste and drinking water, it does something pretty neat. It interacts with the enamel on your teeth, making it stronger and more resistant to cavities. It’s like giving your teeth a tiny superhero cape!
How does it work? Well, the fluoride ions sneak into the crystal structure of your tooth enamel. They don't actually steal electrons from your enamel, that would be too much. Instead, they integrate themselves in a way that makes the enamel less susceptible to the acids produced by the bacteria in your mouth that cause decay. So, while fluorine itself is a bit of a chemical bully, fluoride is a surprisingly good bodyguard for your molars.
But wait, there's more! Fluorine isn't just about dental hygiene. It's also a key ingredient in some seriously cool materials. Remember that non-stick pan you love that makes flipping pancakes a breeze? That slick, slippery surface is often thanks to Teflon, which is made using compounds containing fluorine. It’s like fluorine decided to get into the kitchenware business, and frankly, we’re all better off for it. Imagine trying to cook scrambled eggs on a regular pan – the horror!
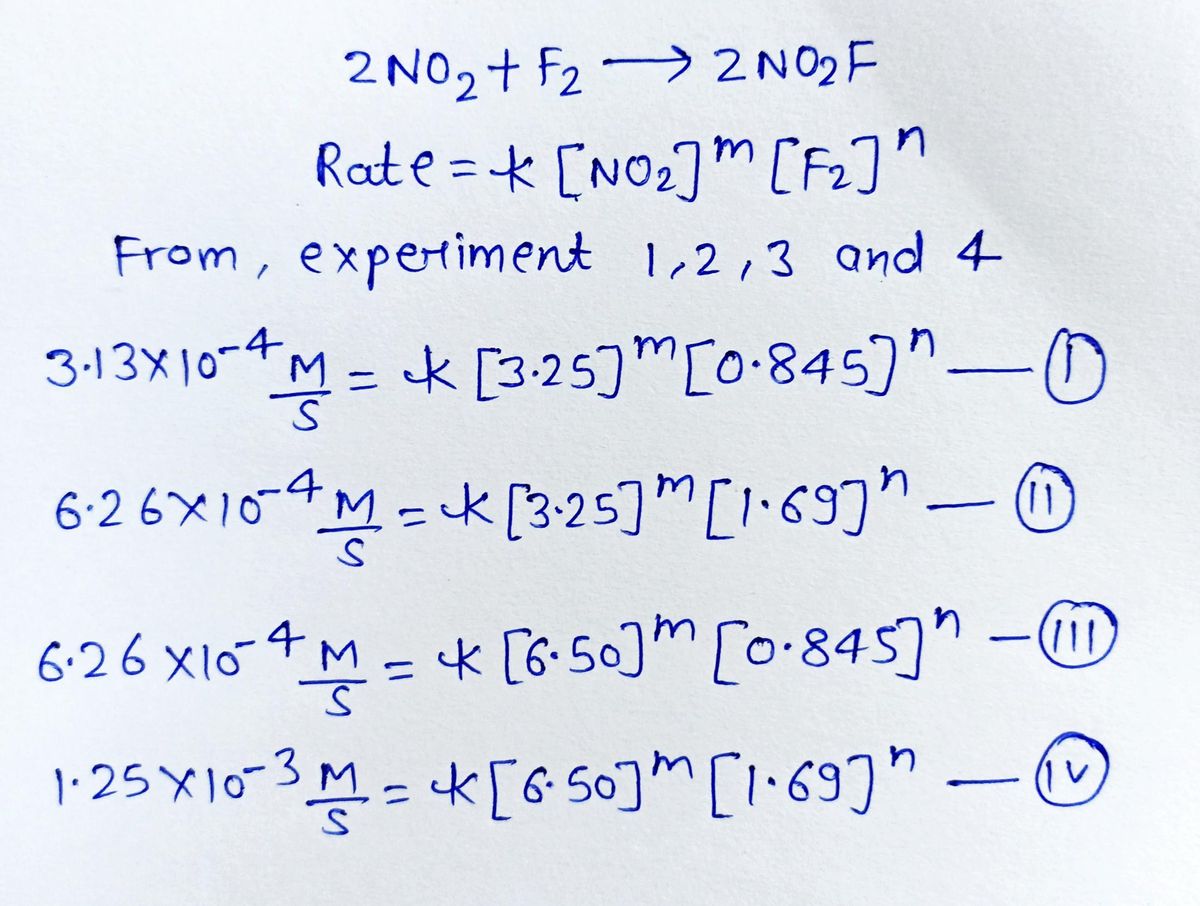
And what about those fancy medical procedures? Fluorine plays a role there too. Some specialized plastics used in medical devices, like catheters and IV tubing, incorporate fluorine. This makes them more durable and resistant to wear and tear, which is pretty important when you’re dealing with life-or-death situations. So, in a weird, roundabout way, fluorine might be helping save lives. Who knew that the most reactive element could be so… helpful?
Let's talk about some of the more extreme examples of fluorine's personality. Because it's so reactive, pure, elemental fluorine (F₂) is incredibly dangerous. It's a pale yellow gas that's highly toxic and can cause severe burns. It's like meeting that guy who's a bit too intense at a party – you want to keep a safe distance. Seriously, imagine trying to bottle that stuff. It would be like trying to hug a greased lightning bolt. Scientists who work with it have to wear some seriously protective gear, probably looking like they're preparing for a zombie apocalypse.

It’s also incredibly powerful. It can react with things you wouldn't expect, like glass! You can't store pure fluorine in a glass container because it will just eat through it. That’s right, it’s so aggressive it can dissolve glass. So, if you ever see a jar of what looks like glowing yellow liquid that’s melting the shelf it’s on, you’re probably looking at some serious fluorine action. Don't poke it. Just… don't.
Another wild fact: fluorine is essential for the production of certain refrigerants, which are used in air conditioning and refrigeration systems. So, that chilly blast of air on a hot summer day? You might have fluorine to thank for that. It’s like, "I'll make your teeth strong, make your pans non-stick, and then I'll even help keep you cool." This element is really pulling its weight in the world, even if it is a bit of a handful.
So, the next time you're brushing your teeth, or enjoying a perfectly cooked pancake, or even just feeling the cool relief of air conditioning, take a moment to appreciate the incredible, and sometimes terrifying, power of fluorine. It’s a reminder that even the most seemingly obscure parts of science can have a huge impact on our everyday lives. And who knew that the scariest element on the periodic table could be so… useful? It’s a chemical paradox, and honestly, I'm here for it.


![[ANSWERED] 3 Regression Consider following data X 3 6 20 14 Y 55 55 10](https://media.kunduz.com/media/sug-question-candidate/20220619195016175602-3516426.jpg?h=512)