How Many Water Molecules Can Urea Bond With
Ever wondered about the tiny, invisible world that makes up everything around us? It's a fascinating place, and sometimes, even the simplest questions can lead to surprisingly interesting discoveries. Today, we're going to dip our toes into the world of chemistry with a question that might sound a little out there: How many water molecules can urea bond with? It might not be the first thing you think about during your morning coffee, but understanding these interactions is actually quite useful and can be a really fun way to explore the science behind everyday things.
For beginners, this is a fantastic way to start thinking about how different substances behave when they meet. It's like learning how your favorite LEGO bricks connect! For families, it can spark curiosity in kids, turning a simple glass of water with a pinch of something into a science experiment. Imagine explaining that even though urea looks like a simple white powder, it's got its own little handshake with water. Hobbyists, whether they're into gardening (urea is a fertilizer!) or even brewing, might find this knowledge surprisingly applicable to optimizing their processes.
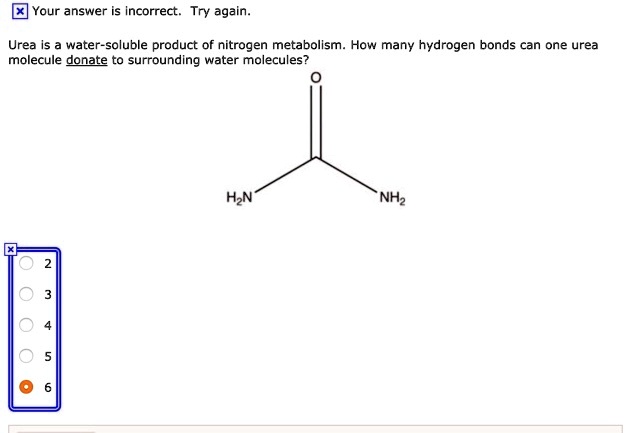
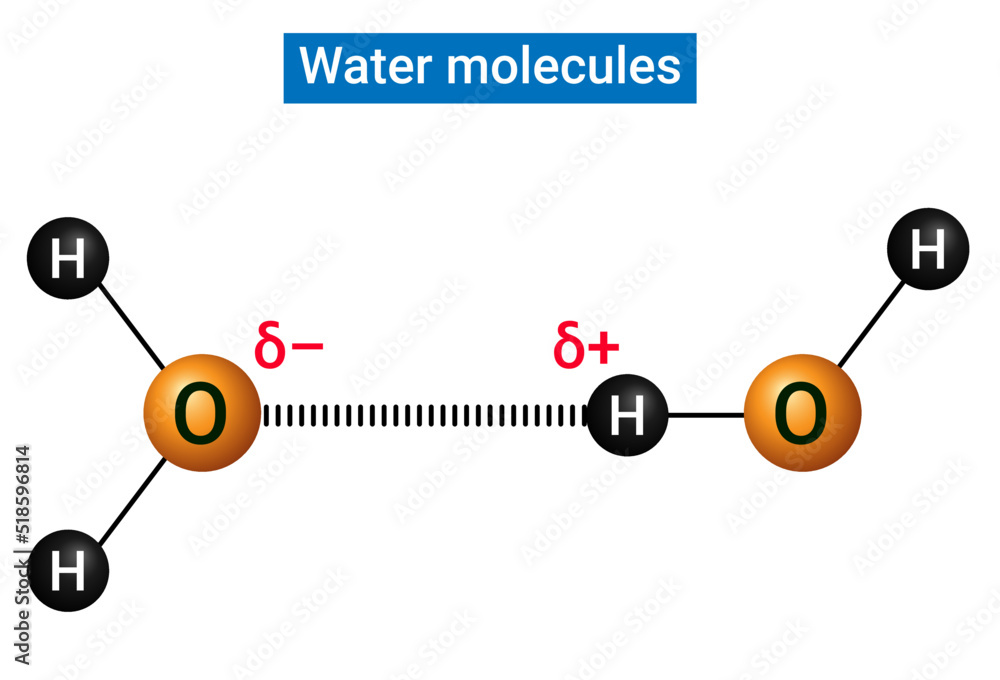
So, let's break it down. Urea is a molecule that's quite good at dissolving in water. Think of it like a tiny magnet that can attract water molecules. But here's the exciting part: it's not just one water molecule that gets cozy with urea. Urea actually has a couple of spots where it can form these bonds, specifically hydrogen bonds. These are like little sticky fingers that water molecules use to hold onto urea. In a nutshell, a single urea molecule can typically form bonds with four water molecules. It's like urea is having a little party and inviting four water molecules to join in!
Must Read
Now, this number can be a bit flexible depending on the conditions, but four is a great general rule of thumb. You might see variations if you're dealing with extremely concentrated solutions or at very low temperatures, but for most everyday scenarios, that's the magic number. It's this ability of urea to interact with water that makes it so soluble and useful in many applications.
Getting started with this idea is super simple. You don't need a fancy lab! Next time you're adding something to water, just think about the molecules. For example, salt (sodium chloride) dissolves in water too, but it interacts differently than urea. Salt breaks down into charged particles (ions) that are then surrounded by water molecules. It's a different kind of handshake! Another variation to consider is sugar. Sugar also dissolves well, and like urea, it forms hydrogen bonds with water, but it can often bond with even more water molecules than urea can, depending on the type of sugar.
![[GET ANSWER] 8. Urea is a water-soluble product of nitrogen metabolism](https://cdn.numerade.com/ask_images/6da2255547ef4048a23b3f1dd9991c8f.jpg)
If you want to explore this further, a great practical tip is to simply observe. Mix a little bit of urea (you can find it in some garden fertilizers, but be careful and always supervise children!) into water. Notice how easily it dissolves. You can then compare this to something that doesn't dissolve as well, like sand. This simple observation can lead to bigger questions about why things dissolve. It's a gateway to understanding the fascinating, invisible world of molecular interactions that shape our reality.
So, the next time you encounter urea, whether it's in a garden or a science experiment, remember its little molecular party with water. It's a small piece of knowledge, but it opens up a whole new way of looking at the world, making everyday science a little more enjoyable and a lot more valuable.