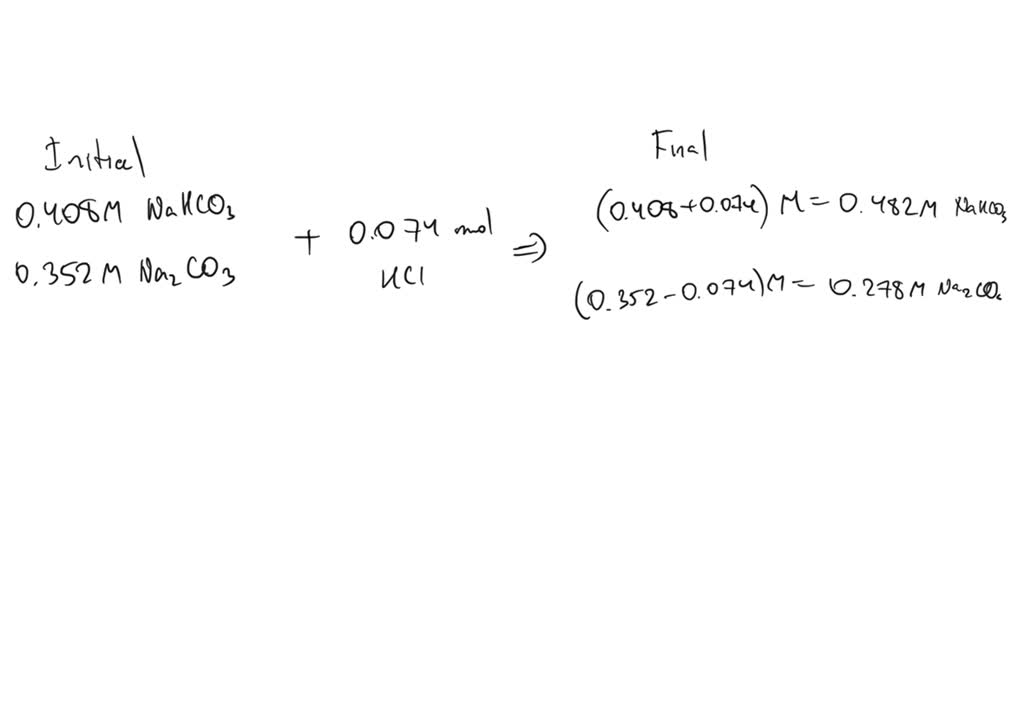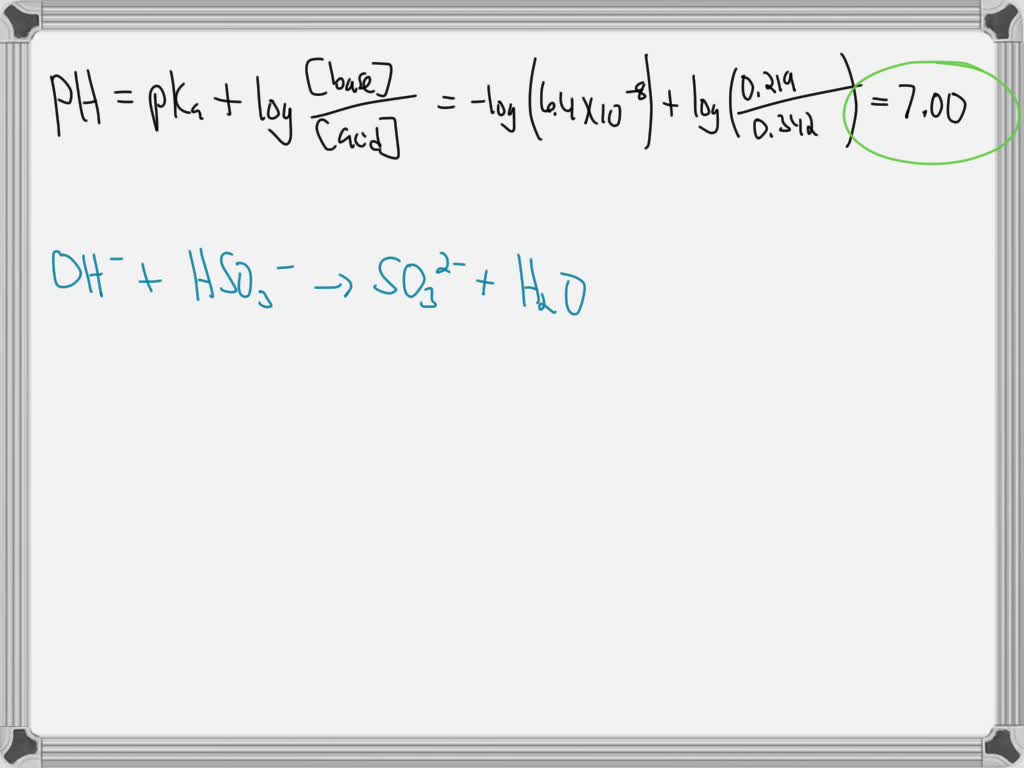Buffer Solution Containing Na2co3 And Nahco3

Hey there, science adventurers! Ever feel like your body is a bustling city, with all sorts of chemical reactions going on, some that want to make things a little too acidic and others that are a bit too basic? Well, guess what? Your body, and a lot of other cool stuff in the world, have tiny, unsung heroes working overtime to keep things perfectly balanced. Today, we're going to talk about one of these super-duper, pH-preserving pals: the Sodium Carbonate and Sodium Bicarbonate power couple!
Think of your blood like a fancy restaurant kitchen. You've got chefs (enzymes) making all sorts of amazing dishes (chemical reactions), but if the temperature or the acidity goes wonky, the whole meal can be ruined! We're talking a soggy souffle or a burnt roast. Not good!
That's where our dynamic duo comes in. We've got sodium carbonate (Na₂CO₃) and its best buddy, sodium bicarbonate (NaHCO₃). Together, they form a superhero team, ready to tackle any pH shenanigans that try to crash the party.
Must Read
Imagine you're making a really delicate cake. If you add too much baking soda (which is basically sodium bicarbonate!), your cake might rise a little too enthusiastically and end up tasting a bit… soapy. Ew! On the other hand, if you don't have enough of the right ingredients, your cake might be flatter than a deflated balloon.
Well, our sodium carbonate and sodium bicarbonate solution is like the ultimate cake-baking supervisor. It's got the built-in ability to say, "Whoa there, party animals! Let's chill!" whether things are getting too acidic or too basic. It's the ultimate peacekeeper for your tiny chemical kingdom.
Let's dive a little deeper into how these two amazing compounds work their magic. Think of sodium bicarbonate as the friendly, easy-going one. It loves to hang out in water and doesn't mind being a little bit basic. It's like the helpful neighbor who always offers you a cup of sugar.
Then there's sodium carbonate. This one is a bit more of a powerhouse. It's a stronger base, meaning it's really good at soaking up those pesky acid molecules that try to sneak in and mess things up. It's like the bouncer at a party, making sure only the right kind of guests are allowed in.

But here's the truly mind-blowing part, the secret sauce that makes this combination so incredible. When you have both sodium carbonate and sodium bicarbonate hanging out together in the same solution, they create something called a buffer solution. It's like a chemical shield, a force field of stability!
What does a buffer solution do? It resists changes in pH. So, if some acidic stuff tries to barge in, the buffer solution is there to neutralize it. And if some basic stuff tries to crash the party, the buffer solution is also ready to calm things down. It’s the ultimate chameleon, adapting to whatever situation it faces.
Think about it like this: you’re at a playground, and a couple of kids are arguing. One is saying, "It's too hot!" (acidic) and the other is shouting, "It's too cold!" (basic). A good mediator (our buffer solution!) would come in and say, "Okay, let's find a middle ground. Maybe we can open a window or turn on a fan."
Our sodium carbonate and sodium bicarbonate system does exactly that for chemical reactions. If the environment starts to become too acidic, the bicarbonate ions (from sodium bicarbonate) are like little sponges, ready to grab onto those excess acid molecules. They transform into carbonic acid, which is much weaker and doesn't throw the whole system off balance.

And if, by some crazy chance, the environment starts getting too basic, the carbonate ions (from sodium carbonate) are the ones who step up. They’re like the grumpy but ultimately helpful old uncle who tells everyone to settle down. They can react with water to produce bicarbonate, helping to bring the pH back to normal.
It’s a beautifully choreographed dance of chemical cooperation. Sodium carbonate and sodium bicarbonate are constantly working together, each playing their role to maintain that perfect pH sweet spot. It's like having a synchronized swimming team for your chemical processes – graceful, efficient, and always in perfect harmony.
This is why these guys are so darn important. Your body uses a similar system in your blood to keep it at a very specific pH, around 7.4. If your blood pH swings even a little bit too far in either direction, it can be a very, very big problem. Imagine your blood becoming as sour as a lemon or as bitter as a strong cup of coffee! Not exactly ideal for all those important cells doing their jobs.
But thanks to these amazing buffer systems, your blood stays just right. It’s like having tiny, invisible guardians patrolling your bloodstream, ensuring everything runs smoothly. They are the unsung heroes of your internal well-being, working silently and effectively.
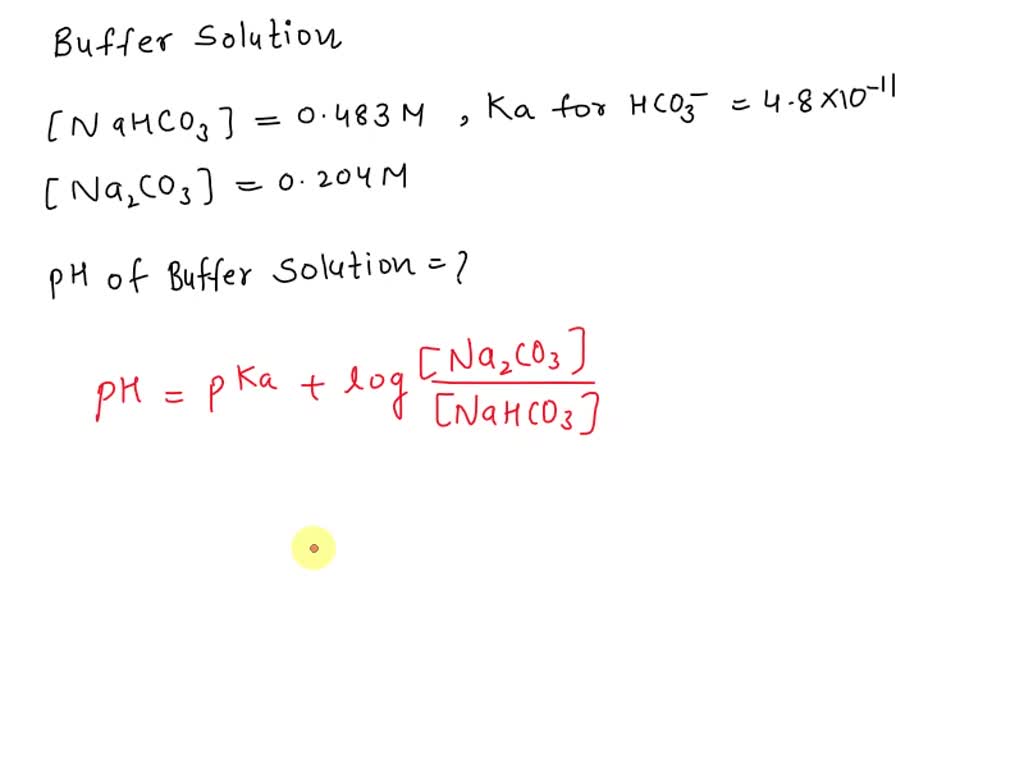
And it’s not just in your body! This amazing buffering power of sodium carbonate and sodium bicarbonate is used in all sorts of places. Think about your favorite shampoo. You want it to be gentle on your hair, right? Well, a buffer solution helps make sure it’s not too acidic or too basic, so your hair doesn't end up feeling like straw.
Or what about those fizzy antacids you might take when you have a tummy ache? Yep, you guessed it! While they might contain other ingredients, the principle of neutralizing excess stomach acid often involves some form of buffering, and the chemical cousins of our dynamic duo are often involved. They’re like the superhero cleanup crew for your digestive system.
Even in baking, besides the baking soda we mentioned, you might find these compounds (or related ones) helping to control the acidity of ingredients like buttermilk or yogurt. This ensures your cakes rise beautifully and have the perfect texture. It’s the secret behind many perfectly baked goods!
So, next time you feel perfectly fine, or enjoy a delicious, well-baked treat, take a moment to appreciate the incredible work of chemical substances like sodium carbonate and sodium bicarbonate. They are the silent guardians of balance, the unsung heroes of pH stability, and the reason why so many things in our world, from our bodies to our baked goods, work just the way they should.

They are truly a marvel of nature and chemistry, working together in perfect harmony. It’s a reminder that even the smallest components can have the biggest impact. They are the ultimate team players, always ensuring that the conditions are just right for everything else to thrive. How cool is that?!
The real magic happens when sodium carbonate and sodium bicarbonate join forces. They create a buffer solution, a chemical superhero that keeps pH from going wild!
So, there you have it! A little peek into the wonderful world of sodium carbonate and sodium bicarbonate and their incredible ability to maintain a stable pH. They are the quiet backbone of many essential processes, and it’s pretty amazing to think about the science happening behind the scenes to keep everything in check.
It’s like having a tiny, invisible team of engineers constantly monitoring and adjusting the environment. They are the guardians of equilibrium, ensuring that the delicate balance required for life and function is always maintained. It’s a testament to the power of simple compounds working together in a sophisticated way.
So, let’s give a big round of applause for these chemical marvels! They might not get the spotlight, but they are undeniably essential. They are the quiet champions of consistency, ensuring that the world around us, and within us, operates with remarkable precision.