The Criss Cross Method Of Balancing Charge
Okay, so let's talk about something that might sound a little bit… well, let’s just say it’s not exactly the star of the chemistry show. We’re talking about balancing charges. You know, those little pluses and minuses that go whizzing around in the world of atoms and molecules. It’s like a tiny, invisible dance party.
Most folks learn about this stuff in school. It’s presented as this super important, totally serious business. And sure, it is important. But sometimes, I think we get a little too bogged down in the jargon. We forget the simple, almost silly, beauty of it all.
My personal, slightly controversial, take on this is that there’s a super easy way to get a handle on it. It’s so simple, it feels like cheating. But it’s not cheating! It’s just… smart. It's like finding a secret shortcut on a video game.
Must Read
I call it the Criss Cross Method. Why? Because it just sounds cool, doesn't it? And honestly, that’s the most scientific explanation I can give you. It involves a little bit of crossing and a little bit of switching. Like trading places at a party.
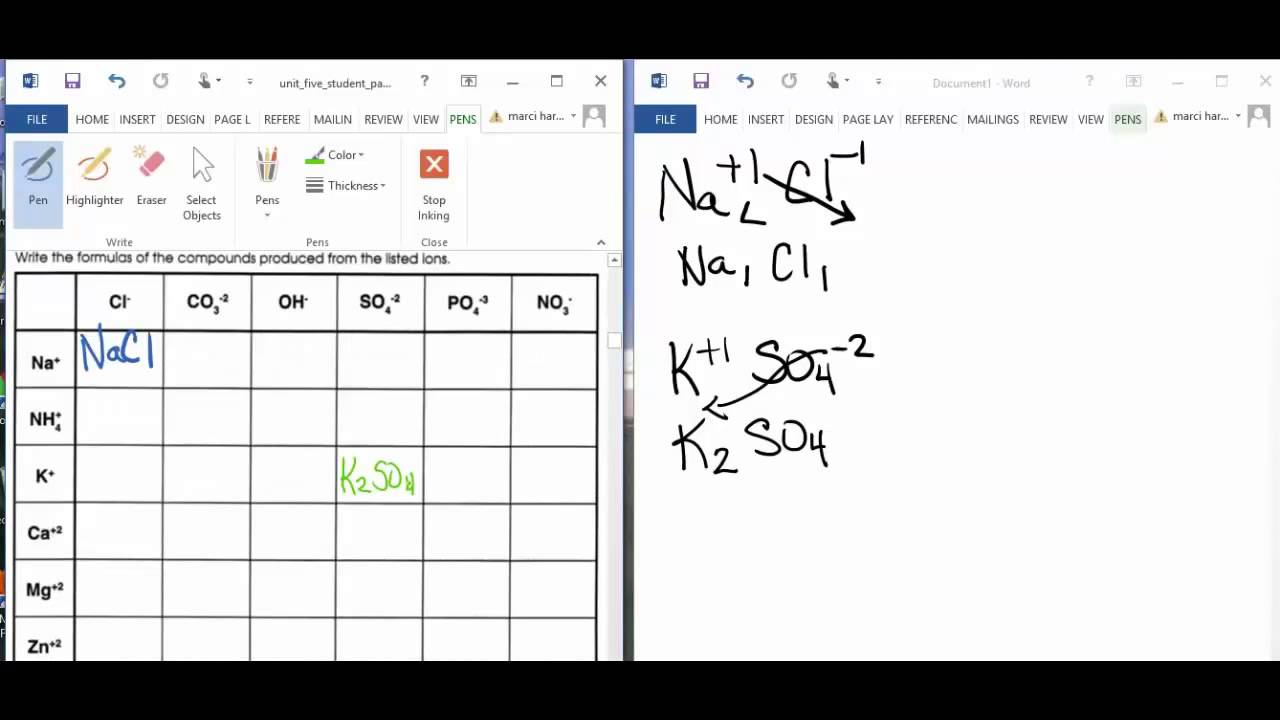
Think of it like this: you have two partners in this charge-balancing tango. Let's call them Partner A and Partner B. Partner A has a certain number of charges. Partner B has a different number. They want to team up to create something stable, something neutral.
Now, instead of getting all mathematical and angsty, we just do this. We take the number of charges from Partner A and we give it to Partner B. And we take the number of charges from Partner B and we give it to Partner A. It’s a swap! A friendly exchange.
So, if Partner A has, let's say, a charge of 2+ (that’s two positive feelings, apparently), and Partner B has a charge of 1- (one negative feeling), we don't get all stressed. We just… switch.
The 2+ from Partner A becomes the number of Partner B we need. And the 1- from Partner B becomes the number of Partner A we need. See? Simple as pie. Or maybe simple as a really well-organized cookie jar.

It's like when you're playing a game with friends. You need a certain number of blue tokens and a certain number of red tokens to win. If you have too many blue and your friend has too many red, you just swap some. Boom! Everyone's happy.
This Criss Cross Method bypasses all the confusing intermediate steps. It gets you straight to the answer. It's the VIP lane for charge balancing. No waiting in the general admission line.
Let's try another example. Imagine we have Ion X with a charge of 3+ and Ion Y with a charge of 2-. We want them to join forces.
Instead of staring at the numbers and wondering about common denominators or something equally frightening, we just… criss cross.
The 3+ from Ion X becomes the number of Ion Y we need. So we need three Ion Ys.
And the 2- from Ion Y becomes the number of Ion X we need. So we need two Ion Xs.
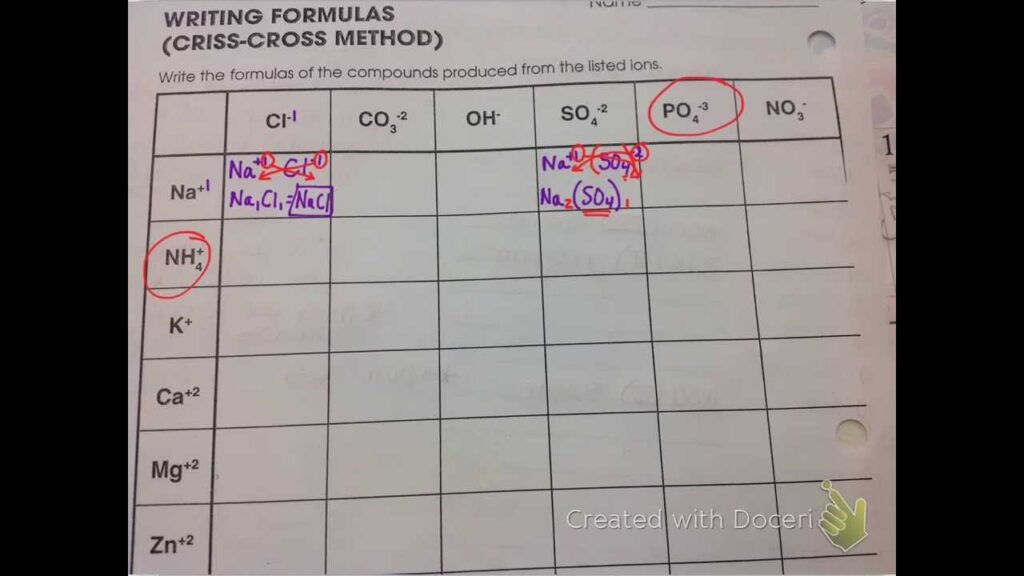
Voila! We have two Ion Xs and three Ion Ys. The charges totally cancel each other out. It’s like a chemical high-five. A successful union.
It works because we're aiming for neutrality. A charge of zero. And this method just makes that happen efficiently. It's a little bit of magic, powered by simple numbers.
Now, I know what some of you might be thinking. "But what about the electron configuration? What about electronegativity?" And yes, those are important. For, you know, actual chemists.
But for the rest of us? For those of us who just want to get through the homework without pulling our hair out? The Criss Cross Method is a lifesaver. It’s the comfy sweater of chemical bonding.
It’s an intuitive approach. It feels right. It’s not some abstract rule you have to memorize. It’s a visual trick that sticks. Like remembering the alphabet by singing it.

I’ve seen students struggle with these concepts for ages. They get lost in the equations. They get tangled up in the subscripts. It’s a mess.
Then, I show them the Criss Cross Method. And their eyes light up. It's like a tiny spark of understanding ignites. A little ‘aha!’ moment.
And it’s not just for simple ions. You can use it for compounds too. It’s a versatile little tool. A Swiss Army knife of charge balancing.
Think about forming ionic compounds. That’s when positive and negative ions get together. They form a solid structure. A crystal. Like a tiny, organized city.
The Criss Cross Method helps you figure out how many of each ion you need to build that city. To make sure it's sturdy and balanced.
So, the next time you see those pesky charges and feel a pang of dread, remember my little secret. Remember the Criss Cross Method.

Don't overthink it. Just cross. Just switch. Let the numbers do their dance.
It’s the easy way. The fun way. The way that doesn’t require a PhD in theoretical chemistry.
And if anyone tells you it's not the "proper" way, just smile and tell them you’re optimizing for efficiency. You’re practicing advanced chemical ergonomics.
Because sometimes, the most brilliant solutions are the ones that are also the simplest. And the most entertaining.
So go forth and criss cross! Balance those charges with confidence and a smile. You’ve got this.
It’s a beautiful, simple dance. And you’re now invited to lead.





