Electrophilic Addition Of Water To An Alkene

Hey there, awesome science explorer! So, let's dive into something that might sound a bit intimidating at first: the electrophilic addition of water to an alkene. But don't you worry, we're going to break it down, make it super easy, and maybe even have a little chuckle along the way. Think of me as your friendly neighborhood chemistry guide, armed with a metaphorical beaker and a whole lot of enthusiasm!
First off, what's an alkene? Imagine a hydrocarbon chain, like a little string of carbon atoms. Now, if there's a double bond between two of those carbon atoms, you've got yourself an alkene! It's like the carbon atoms are holding hands, but not just with one hand – with two! This double bond is the star of our show today, because it's super reactive and ready for some action. It’s like a party waiting to happen, and water is invited!
Now, what about "electrophilic addition"? Sounds fancy, right? Let's break that down too. An electrophile is basically something that loves electrons. Think of it as an electron-deficient species, always on the lookout for a nice, electron-rich place to hang out. And guess what? That double bond in our alkene? It's absolutely bursting with electrons. It's like a giant buffet for our hungry electrophile!
Must Read
The "addition" part is pretty straightforward. It means that something is going to be added across that double bond. The double bond is going to break, and the atoms from our incoming molecule will attach themselves to the carbon atoms that were involved in the double bond. It's like the double bond is saying, "Okay, you two, come on in! Let's make room for you!"
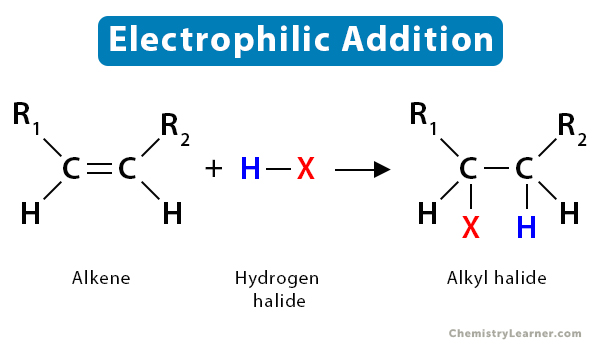
So, putting it all together, "electrophilic addition of water to an alkene" means that an electrophile, which will be derived from water, is going to attack the electron-rich double bond, causing the double bond to break and the components of water to get added to the alkene molecule.
The Star Player: The Double Bond
Let's talk more about this double bond. It's made up of two parts: a sigma bond and a pi bond. The sigma bond is like the strong, stable handshake. The pi bond, however, is weaker and sticks out above and below the sigma bond. This pi bond is where all the electron action is! It's like the loose change in your pocket – easy to grab!
Because the pi electrons are more exposed and less tightly held, they are the first to get attracted to anything that's a bit electron-hungry – our electrophile. It's the most accessible part of the double bond, the easiest target. So, when our electrophile shows up, it's going to go straight for that pi cloud. Think of it as a magnet, but instead of metal, it's attracting electrons.
Enter the Acid Catalyst: The Wingman!
Now, plain old water by itself isn't usually the best electrophile. It's a bit too polite, you know? So, to get this party started, we usually need a little help from an acid catalyst. Common catalysts are strong acids like sulfuric acid (H₂SO₄) or phosphoric acid (H₃PO₄). These guys are really good at giving up a proton (H⁺). And guess what? A proton is the ultimate electron-seeker!
The acid catalyst basically plays the role of the matchmaker. It helps to create a more powerful electrophile that can then attack the alkene. It's like the catalyst is saying, "Psst, water, buddy, you need to be a little more assertive! Here, take this proton and go get 'em!"
Step 1: The Electrophile Makes Its Move!
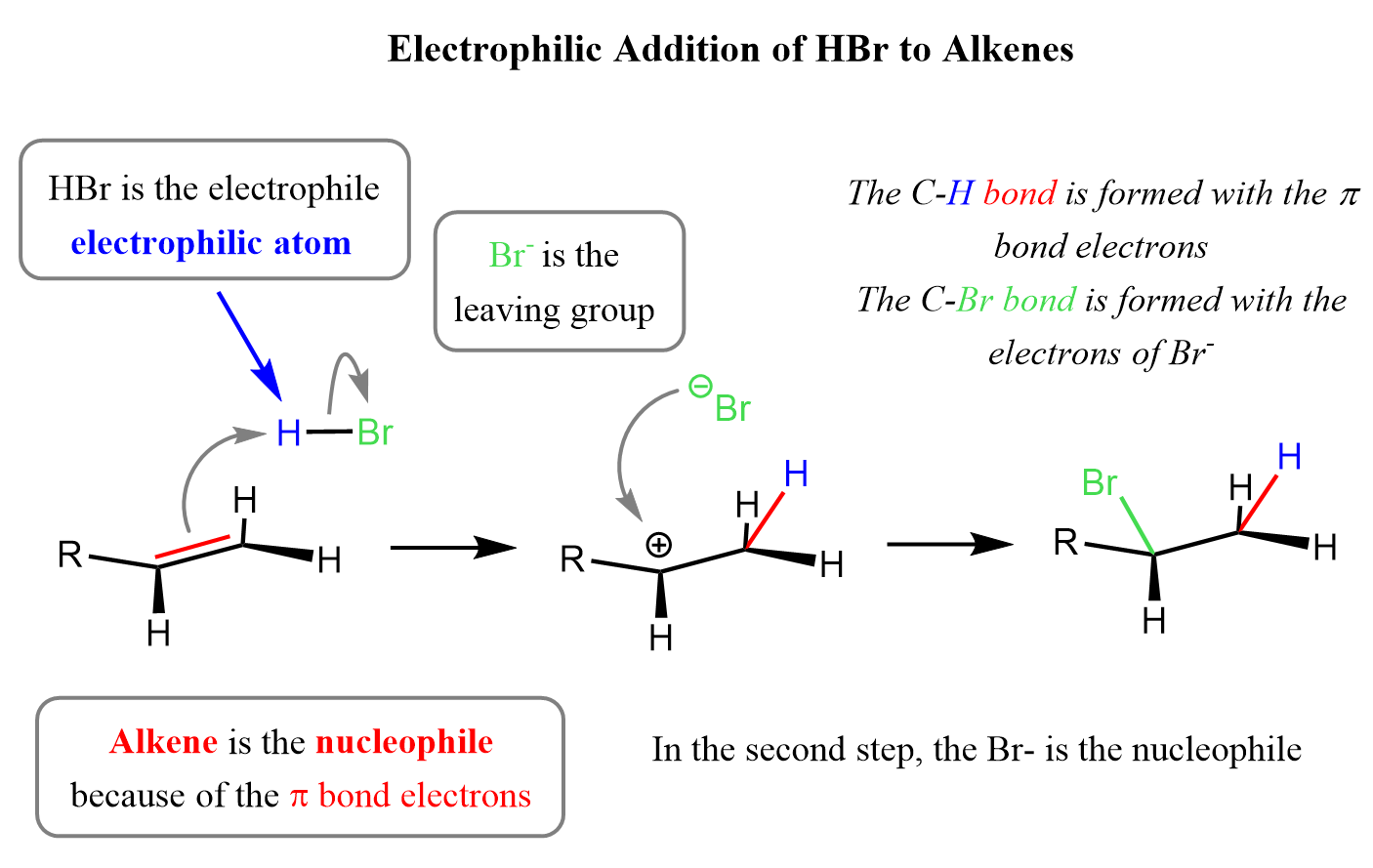
Alright, here's where the magic begins! The first step is the proton from the acid catalyst (our electrophile) getting attracted to the electron-rich pi bond of the alkene. The proton bumps into the double bond, and – poof! – the pi bond breaks. One of the carbon atoms in the former double bond grabs that proton, becoming more stable.
But here's the catch, and it's a crucial one. When the pi bond breaks, it leaves behind a positively charged carbon atom on the other carbon that was part of the double bond. This positively charged carbon is called a carbocation. Think of it as a carbon atom that's just lost its best friend and is now feeling a bit lonely and desperate for some company (or electrons!).
This carbocation is a very important intermediate. It's like a temporary holding cell before the next step. And its stability is a big deal. We'll get to that in a bit, but for now, just remember: proton lands on one carbon, double bond breaks, and a carbocation forms on the other carbon.
Markovnikov's Rule: The Predictable Party Guest
Now, before we move on, we have to talk about a really important rule that governs where that proton actually goes. It's called Markovnikov's Rule. This isn't some grumpy old chemist telling you what you can and can't do; it's more like a helpful tip for predicting the outcome of this reaction. Basically, Markovnikov's Rule states that when a protic acid (like H-X, where X is a halogen, or in our case, we're thinking about the proton from water) adds to an alkene, the hydrogen atom (the "hydro" part) tends to attach itself to the carbon atom of the double bond that already has the most hydrogen atoms attached to it.
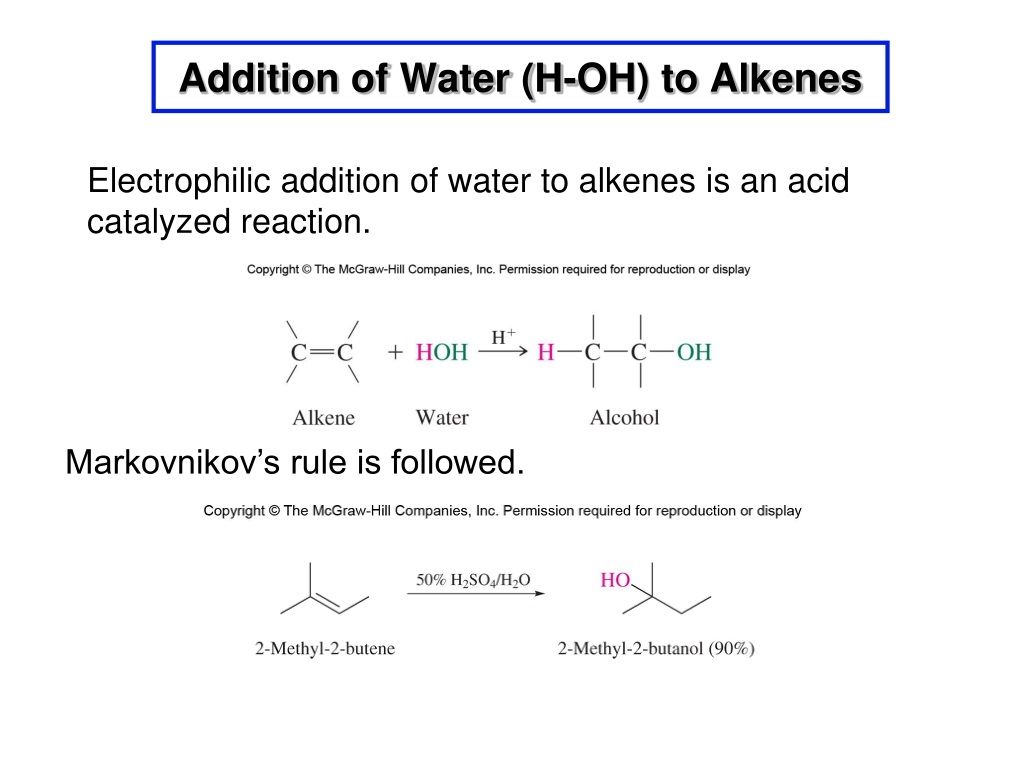
Why? Because it leads to the formation of the most stable carbocation intermediate. Remember that lonely, positively charged carbon? Well, some carbocations are more stable than others. Tertiary carbocations (where the positive carbon is attached to three other carbons) are more stable than secondary carbocations (attached to two other carbons), which are more stable than primary carbocations (attached to one other carbon). It’s like having more friends to lean on when you’re feeling down!
So, the proton will preferentially add to the carbon with more hydrogens, leaving the positive charge to form on the carbon that can better stabilize it, usually the one with more alkyl groups attached. This is a super handy rule for predicting which product you'll get!
Step 2: Water to the Rescue!
Okay, so we've got our carbocation – that lonely, electron-hungry carbon. And what do we have plenty of in our reaction mixture? Water! Remember, water (H₂O) has an oxygen atom with two lone pairs of electrons. These lone pairs are just begging to find a positive charge. It's like a thirsty person finding an oasis!
The oxygen atom in the water molecule, with its electron-rich lone pairs, is attracted to the positively charged carbocation. It swoops in and forms a new bond with the carbon. So, now the carbon is happy again, but the oxygen from the water molecule is now bonded to the carbon and is carrying a positive charge itself. It's a bit like someone generously offering their coat to a shivering person, but then feeling a bit chilly themselves!
This positively charged oxygen species is called an oxonium ion. It's still not quite our final product, but we're getting closer! It's like the first draft of a masterpiece – good, but needs a bit more refinement.
Step 3: Deprotonation – The Final Touch!
We've got our oxonium ion, which is a bit unstable with that positive charge on the oxygen. To become a nice, neutral alcohol, it needs to get rid of that extra proton. So, another water molecule (or even the conjugate base of our acid catalyst, if present) comes along and acts as a base. It plucks that extra proton off the positively charged oxygen.
And voilà! The oxygen is now neutral, and we have formed our final product: an alcohol! The double bond is gone, replaced by a single bond, and a hydroxyl group (-OH) has been added to one of the carbons that was originally part of the double bond. The proton that initiated the whole cascade has also ended up on the other carbon of the original double bond. It's a beautiful transformation!
Regioselectivity: It Matters Where Things Land
This whole process is often described as regioselective. That's just a fancy way of saying that the reaction has a preferred direction. Thanks to Markovnikov's Rule, we can predict with pretty good accuracy which carbon will get the hydroxyl group and which will get the hydrogen. It's not a random jumble; there's a predictable pattern to where everything lands.
Think of it like a carefully choreographed dance. The reactants move in a specific order, forming specific intermediates, and leading to a specific, preferred product. It's not just chaotic splashing; it's a structured event, all thanks to the chemistry involved!
The Big Picture: From Alkene to Alcohol
So, to recap, the electrophilic addition of water to an alkene is a three-step process:
- Protonation: An acid catalyst provides a proton (H⁺), which attacks the alkene's pi bond, forming a carbocation. Markovnikov's Rule dictates where the proton adds to form the most stable carbocation.
- Nucleophilic Attack: Water, acting as a nucleophile (because of its electron-rich oxygen), attacks the positively charged carbocation, forming an oxonium ion.
- Deprotonation: A base (often another water molecule) removes a proton from the oxonium ion, yielding the final alcohol product and regenerating the acid catalyst.

This reaction is incredibly important in organic chemistry because it's a fundamental way to create alcohols from readily available alkenes. Alcohols are super versatile and are used in everything from solvents and fuels to pharmaceuticals and flavors. So, next time you see a nice cold drink or a potent cleaning solution, you can think, "Hey, maybe some of that alcohol got here thanks to an electrophilic addition of water!"
Why is this so cool?
Beyond the obvious utility, this reaction showcases some really neat chemical principles. It demonstrates the power of electron-rich pi bonds, the role of electrophiles and nucleophiles, the formation of reactive intermediates like carbocations, and the elegance of regioselectivity guided by principles like Markovnikov's Rule. It’s a beautiful illustration of how atoms and molecules interact, driven by the fundamental forces of attraction and repulsion.
And guess what? The acid catalyst gets regenerated at the end of the reaction. That means it can go off and start another round of the reaction with more alkene and water. It's like a tireless party starter, making sure the fun never stops! This catalytic nature makes the process very efficient.
So, there you have it! Electrophilic addition of water to an alkene. It might sound like a mouthful, but it's really just a series of well-orchestrated steps driven by the fundamental attraction of electrons. From a reactive double bond to a useful alcohol, it’s a testament to the cleverness of chemical reactions.
Isn't chemistry amazing? It's like a giant puzzle, and with each reaction we understand, we unlock another piece of the universe's incredible design. So, keep exploring, keep asking questions, and never be afraid to dive into the wonderfully weird and fascinating world of molecules. You've got this, and who knows what incredible discoveries you'll make next! Keep that scientific curiosity shining bright – it’s your superpower!





