Is The Autoionization Of Water Endothermic

Okay, let's talk about something that sounds super fancy but is actually a bit of a homebody. We're diving into the tiny world of water. You know, the stuff you drink, shower in, and use to make your morning coffee. It seems so simple, right?
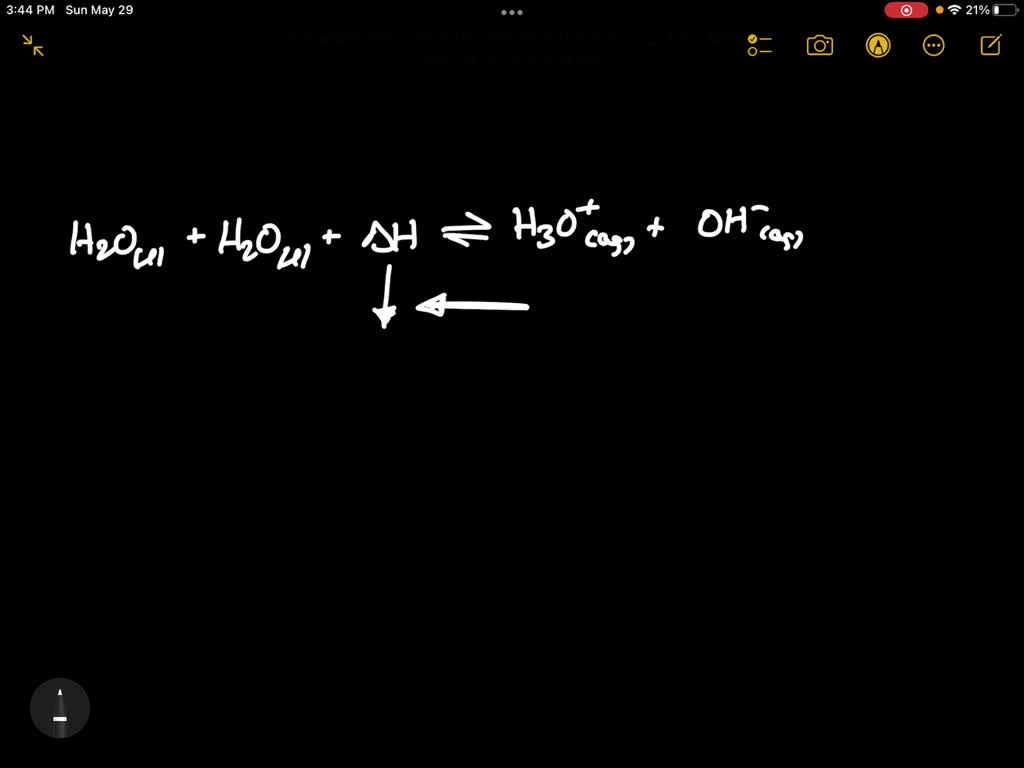
But even water has its little secrets. It’s not just sitting there being wet. It’s actually a little bit of a drama queen, performing a tiny dance all by itself. This little dance is called autoionization. Sounds dramatic, I know. Like a tiny water rave.
So, what's this autoionization thing all about? Basically, a few water molecules, feeling a bit restless, decide to break up. They split into two pieces. It’s like a tiny divorce, but way less paperwork.
Must Read
These broken pieces are called ions. One is a grumpy little proton, which we chemists often write as H+. The other is a rather bewildered hydroxide, looking lost without its other half, which is OH-. They’re like the loneliest singles at a molecular party.
Now, the big question that’s been whispered in hushed tones (okay, maybe just in my head) is about how much energy this whole breakup requires. Is it a warm hug or a chilly shove?
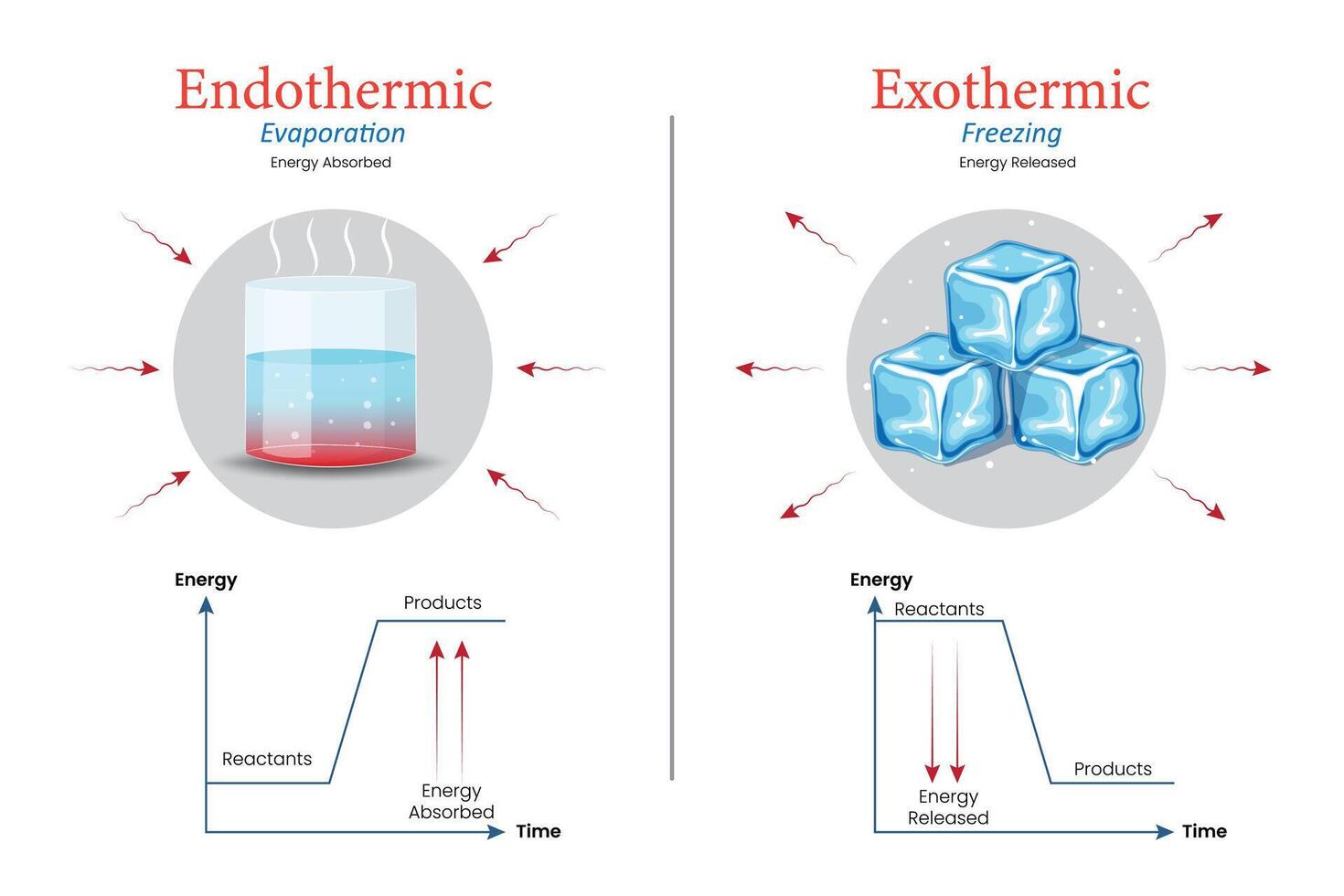
We're talking about whether this whole water-splitting business is endothermic. For those who slept through chemistry class (no judgment, I sometimes dozed off too), endothermic means it needs energy to happen. Like needing a warm blanket on a cold day.
Think about it. Water molecules are pretty happy, cozy, and well-behaved in their little clusters. They’re all holding hands, enjoying their liquid life. Why would they just decide to split up for no reason?
It feels like they’d need a little nudge, right? A bit of a pep talk, or maybe some caffeine, to get them going. This suggests they’re asking for energy. They aren't just spontaneously deciding to go their separate ways.
Imagine a group of friends having a really good time. They’re chatting, laughing, maybe sharing some snacks. Would they suddenly decide to have a fight and storm off in different directions without any provocation?

Probably not. They’d likely need something to disrupt their good vibes. Some external force or a collective decision to seek new adventures. So, it makes sense that water molecules would need a little energy boost to break their bonds.
This is where my (slightly unconventional) theory comes in. I have this feeling, this gut instinct if you will, that the autoionization of water is indeed endothermic.
It's like a small, energetic demand. The water molecules are saying, "Hey, we're kind of comfy here. If you want us to break up and become these ions, you gotta give us some juice!" It’s not a spontaneous, energetic explosion of ions.
I picture it like this: you’re all snuggled up on the couch, watching a good movie. You’re warm, you’re content. Suddenly, someone says, "Okay, time for a spontaneous dance-off!" You'd probably think, "Uh, can I at least grab a snack first? I need energy for this!"
The breaking of bonds between water molecules seems like it would require that kind of effort. They’re not just falling apart like a stale biscuit. They have to actively split.
And where does this energy usually come from? Well, in the grand scheme of things, it’s usually the surrounding environment. If the water is warm, it has more of this 'oomph' to go around.
So, when water gets warmer, it’s more likely to have these little molecular breakups. It's like the warmer it gets, the more energetic and willing the water molecules are to try this ion thing.

This is why the autoionization constant of water, a number that tells us how much autoionization is happening, increases with temperature. More heat, more breakups. It's a simple cause and effect, and it fits my endothermic hunch perfectly.
Think of it as a tiny, molecular investment. The water molecules are investing a bit of energy to get a return of ions. These ions then go on to do all sorts of things, like making things acidic or basic.
It’s not like freezing water, which releases energy (that’s exothermic, the opposite). Freezing is like the molecules deciding to huddle together for warmth, saying, "Phew, glad we’re not split up anymore!"
But autoionization? It feels like a little step away from that perfect, unified state. A step that requires a little push, a little spark, a little energy input.
Now, I know what some of you might be thinking. "But what about what the textbooks say? What about all those fancy scientific papers?" And you’re right, there are established facts. But sometimes, it’s fun to just ponder the feeling of it.
My feeling, my unwavering, slightly stubborn belief, is that this little molecular divorce isn't a freebie. It’s a transaction. Water gives up some of its own internal calm, its cozy togetherness, and asks for energy in return.
It's an unpopular opinion, maybe. The "official" answer might be more complex, or perhaps even slightly different in the eyes of rigorous science. But the intuition is strong.
The very idea of breaking something apart, of disrupting a stable, bonded structure, screams "energy needed!" It’s like unzipping a jacket. You have to pull the zipper up. It doesn't just zip itself.
And for water, those little zipper pulls are the energy that drives the autoionization. The proton and hydroxide ions are the result of this energetic effort.
So, next time you’re sipping on a glass of water, or even just looking at a puddle, remember this tiny drama. The ceaseless, quiet autoionization. And maybe, just maybe, smile and agree with me that it’s all a little bit endothermic. A little bit of a water-powered energy quest.
It’s the little things, isn’t it? The subtle energy exchanges happening all around us, even in something as ordinary as water.
It’s like a tiny, ongoing scientific soap opera. Who’s breaking up? Who’s joining forces? And how much energy does it all take?
For me, the answer is pretty clear. It takes energy. It’s endothermic. And that, my friends, is a beautiful thing. A very, very watery thing.

Think of it as water's little energetic investment in becoming more interesting. It’s not just plain old H2O; it’s a dynamic, ion-producing entity. And that takes effort!
So, while the chemists might have their precise equations and rigorous proofs, let's not forget the simple, elegant logic of everyday observation. Breaking things usually requires a bit of a push. And water, in its own quiet way, is pushing itself apart.
This is why I firmly stand by my gentle, slightly whimsical belief: water's autoionization is endothermic. It's a small, energetic transaction that makes our world a little more chemically interesting.
It’s just a hunch, a feeling. But it’s a feeling that makes perfect sense to me. And hopefully, it brings a little smile to your face too.
The universe is made of stories, not of atoms.
— Muriel Rukeyser
And the story of water autoionization, in my humble opinion, is an endothermic one.