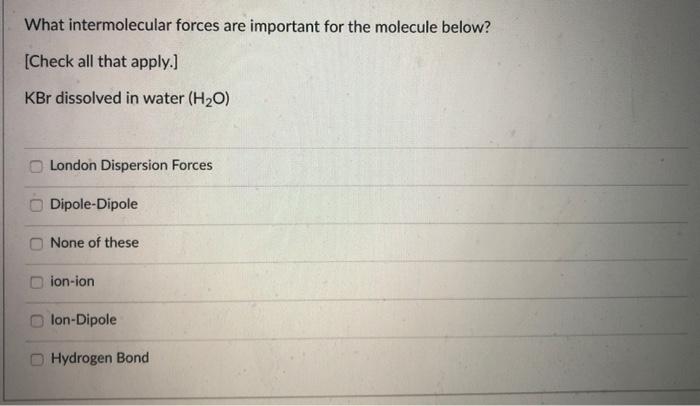
Kbr Dissolved In Water Intermolecular Forces
Hey there! So, you wanna chat about KBr dissolving in water? Awesome! It’s one of those everyday science things that’s actually pretty neat, you know? Like, you’ve probably tossed some salt – or, you know, potassium bromide, which is kinda like fancy salt – into your water and watched it just disappear. Poof! Gone. But why does it do that? It’s not like it sprouts little legs and runs away. Nope. It’s all about the microscopic dance party happening between the KBr and the H2O. And guess what? It’s all thanks to these things called intermolecular forces. Sounds super sci-fi, right? But it's really just how molecules like each other (or don't!).
So, let's break it down. We've got our trusty KBr. What is that, anyway? It's an ionic compound. Think of it like a really stable couple. Potassium (K) and Bromine (Br) got together, and one of them totally gave an electron to the other. This makes Potassium a positively charged ion, or a cation (yay for science terms!). And Bromine? It’s now a negatively charged ion, an anion (double yay!). They’re stuck together like magnets, but like, super strong magnets, thanks to this ionic bond. They form this neat, orderly crystal lattice. It's like a perfectly stacked pile of LEGOs, all holding hands. Very organized, very committed.
Now, let's bring in water. Ah, water. The universal solvent. The stuff of life. The reason we can make tea and do laundry. But water is also a bit of a drama queen. It's a polar molecule. What does that mean? It means it's got a little bit of a split personality. Oxygen is a bit greedy for electrons, so it pulls them closer to itself. This gives the oxygen end of the water molecule a slight negative charge. And the hydrogen ends? They’re left feeling a bit electron-deprived, so they get a slight positive charge. It's like one side of the molecule is always a little grumpy and the other is always a little cheerful. This uneven charge distribution is what makes water so special, and so good at dissolving things!
Must Read
So, picture this: you dump your KBr crystal into your nice, muggy water. The water molecules, with their grumpy negative oxygen ends and cheerful positive hydrogen ends, start to surround the KBr crystal. They're like little curious onlookers, all buzzing around. They notice those strong ionic bonds holding K+ and Br- together. And they think, "Hmm, I bet I can break that up!"
The positive hydrogen ends of the water molecules are naturally drawn to the negative bromide ions (Br-). Remember, opposites attract, right? So, these positive hydrogens start to snuggle up close to the bromide ions. They’re not forming a new bond, mind you, just getting really, really close. It’s like a group hug, but with a specific target!

At the same time, the negative oxygen ends of the water molecules are doing their own thing. They’re attracted to the positive potassium ions (K+). Again, opposites attract! So, these negative oxygens start to surround the potassium ions, getting all cozy. It’s like another group hug, but for the potassium ions this time. You can almost hear the water molecules whispering, "Come on over, we've got room for you!"
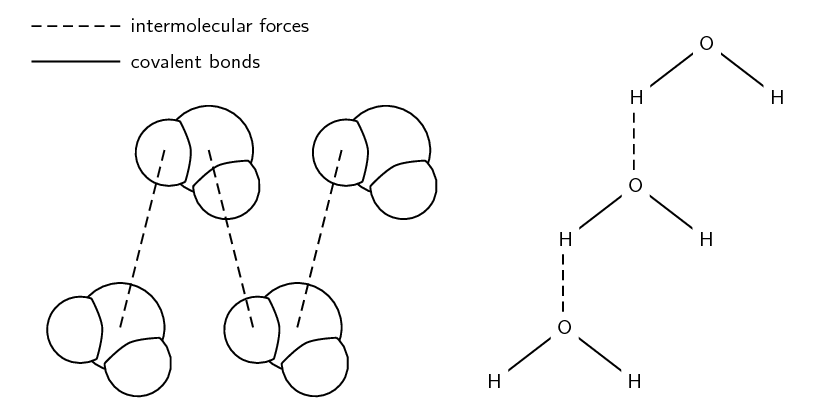
This process, where water molecules surround and pull apart ions from an ionic compound, is called hydration. It's like the water is giving each ion its own personal entourage. Each K+ ion gets its own little crew of water molecules, and each Br- ion gets its own little posse. These water molecules are essentially solvating the ions, meaning they’re forming a protective layer around them. This layer prevents the ions from clumping back together.

And here’s the really cool part: the intermolecular forces between the water molecules and the ions are actually stronger than the ionic bonds holding the KBr crystal together, and even stronger than the forces holding the water molecules to each other. It’s like the KBr crystal tried to have a family reunion, but the water molecules threw such an awesome party with all the ions that they decided to stay and hang out in the water instead. The energy released when these new ion-dipole attractions form is enough to overcome the energy required to break the ionic lattice and the hydrogen bonds in water.
Think about it this way: you have a super-tight hug with your best friend (that's the ionic bond in KBr). Then, a bunch of your other friends (water molecules) come along, and they're really good at giving hugs. They start hugging both you and your best friend individually, and their hugs are so good, and there are so many of them, that you and your best friend eventually let go of each other to get these new, awesome hugs from the water friends. And then you're all just floating around, having a grand old time.
So, that KBr crystal? It’s not actually gone. It’s just broken down into individual potassium ions (K+) and bromide ions (Br-), each happily surrounded by a swarm of water molecules. They’re dispersed throughout the water, so you can’t see them anymore. They’re dissolved! It’s like a microscopic rave where the ions are the VIPs and the water molecules are the enthusiastic dancers.

This whole thing is a prime example of the "like dissolves like" rule in chemistry. Polar solvents, like water, tend to dissolve polar solutes and ionic compounds. Nonpolar solvents, like oil, tend to dissolve nonpolar solutes. KBr is ionic, which is pretty darn polar in its own way with those charges, and water is super polar. So, they're a match made in heaven. Or, you know, in a beaker.
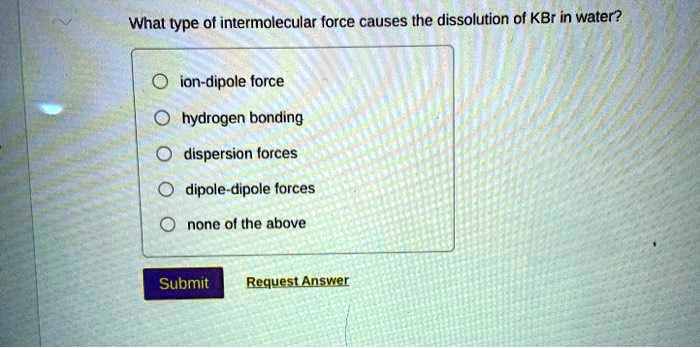
The forces involved are mostly ion-dipole forces. That's the attraction between an ion (like K+ or Br-) and a dipole (like a water molecule, with its positive and negative ends). These forces are strong enough to pull the ions away from the crystal lattice. Then, within the water itself, you have hydrogen bonding. Water molecules form these weak, fleeting bonds with each other. When the ions get hydrated, these hydrogen bonds in the water get temporarily disrupted to make room for the ion-dipole interactions. It's a constant ebb and flow of attractions!
It's pretty amazing when you think about it. You take something solid, something that looks pretty stable, and you introduce it to something else, and poof, it vanishes. It’s not magic; it’s just the universe at its molecular best. The intricate dance of attractions and repulsions, the way molecules interact based on their charges and structures. It’s all happening right there in your glass of water!
And it’s not just KBr, of course. This is how most salts dissolve in water. Sodium chloride (table salt, NaCl)? Same deal. Magnesium sulfate (Epsom salt)? Yep, that too. They all have those ionic bonds that water, with its polar personality and its ability to form strong intermolecular forces, can break apart. It's like water is the ultimate mediator, always ready to break up a solid union and get everyone mingling.
So, next time you're dissolving something in water, take a moment to appreciate the invisible ballet of molecules. The attraction between the positive and negative poles, the way water molecules flock to ions like paparazzi to a celebrity. It's a beautiful, chaotic, and utterly essential part of how our world works. Who knew that dissolving a little salt could be such a grand spectacle of intermolecular forces, right? Makes you wonder what other hidden dramas are playing out in everyday substances. Probably a lot! Now, who wants more coffee?