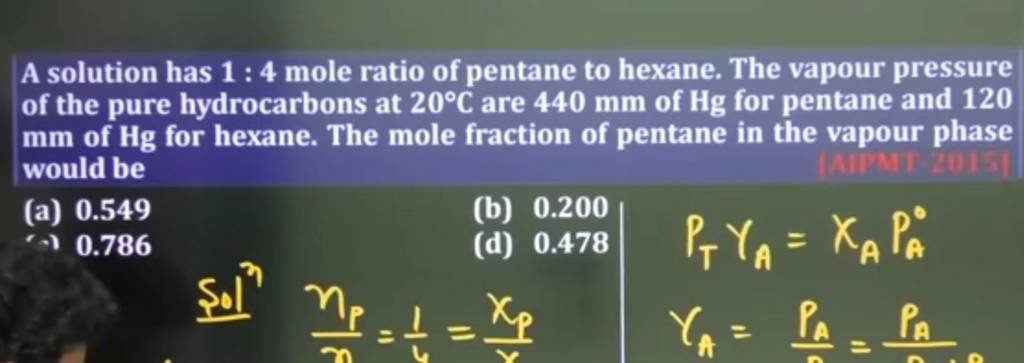
What Is The Mole Ratio Of Oxygen To Pentane

Hey there, science explorers and curious minds! Ever find yourself staring at a gas burner, or maybe just a really fancy candle, and think, "You know, I wonder about the tiny dance happening right here?" Well, buckle up, buttercups, because today we're diving into a question that might sound a little… niche. We’re talking about the mole ratio of oxygen to pentane. I know, I know, it sounds like something straight out of a chemistry textbook, but trust me, there's a surprising amount of fun and inspiration hidden in these numbers!
Think about it. We’re all breathing, right? And most of us have probably enjoyed a picnic with some grilled goodness, or maybe even just lit a birthday cake. All of these involve a chemical reaction, a super-cool chemical reaction called combustion. And pentane? Well, pentane is a pretty common ingredient in things like gasoline, and it’s also found in some camping stoves. So, this isn't just abstract science; it's the stuff of everyday life!
So, what exactly is this "mole ratio" we’re babbling about? Don't let the fancy words scare you. In the world of chemistry, a "mole" is just a way of counting things. It's a huge number, like a baker's dozen, but for atoms and molecules. Imagine trying to count every single grain of sand on a beach – a mole is like a super-powered counter that makes that job ridiculously easy for chemists.
Must Read
And the "ratio"? That’s just a comparison. Like comparing how many apples you have to how many oranges. Simple, right? So, the mole ratio of oxygen to pentane is simply telling us how many moles of oxygen molecules are needed to completely react with a certain number of moles of pentane molecules. It's like the perfect recipe for a fire!
Now, why should you care about this? Because understanding these ratios unlocks a whole new level of appreciation for the world around you. It’s like learning the secret language of the universe! When you see a flame, you can mentally picture the molecules doing their thing, a synchronized ballet of atoms.
Let’s get down to the nitty-gritty. Pentane has the chemical formula C5H12. This means for every molecule of pentane, there are 5 carbon atoms and 12 hydrogen atoms. Oxygen, our ever-present friend, is O2. That little '2' is important; it means oxygen molecules come in pairs. Now, when pentane burns, it reacts with oxygen to produce carbon dioxide (CO2) and water (H2O). It’s a chemical transformation, a bit like a magic trick, but it follows precise rules.

To figure out the mole ratio, we need to balance the chemical equation. This is where the "recipe" comes in. We want to make sure we have the right amount of each ingredient so that everything is used up perfectly. Think of it like baking a cake – if you have too much flour and not enough eggs, it’s not going to turn out right, is it?
The balanced chemical equation for the combustion of pentane looks like this:
C5H12 + 8O2 → 5CO2 + 6H2O

Take a moment to marvel at that! It's a thing of beauty, isn't it? On the left side, we have our reactants – pentane and oxygen. On the right side, we have our products – carbon dioxide and water. See those big numbers in front of the molecules? Those are the coefficients, and they tell us the mole ratios!
The Grand Reveal!
Look at the equation again. What’s in front of the pentane (C5H12)? It’s a little ‘1’ that we usually don’t write. And what’s in front of the oxygen (O2)? It's an ‘8’. So, what does this tell us about our mole ratio of oxygen to pentane?
For every 1 mole of pentane that burns, you need exactly 8 moles of oxygen.
![[ANSWERED] 30 A solution has a 1 4 mole ratio of pentane to hexane - Kunduz](https://media.kunduz.com/media/sug-question-candidate/20210513032935244443-2921068.jpg?h=512)
Isn’t that neat? It’s like a universal rule for how these molecules party together. This 1:8 ratio is the secret handshake between pentane and oxygen.
Why is this so cool? Well, imagine you're designing a fuel-efficient engine. Understanding these ratios helps engineers figure out exactly how much oxygen is needed for optimal combustion, leading to less wasted fuel and cleaner emissions. It’s science making our world a little bit better, one balanced equation at a time!
Or think about it on a smaller scale. Maybe you’re into camping and you’re using a portable stove that runs on pentane. Knowing this ratio can give you a deeper understanding of how your stove works. It’s not just a black box; it’s a system governed by these elegant chemical principles. You’re not just a user; you're an informed participant in a chemical process!
![[ANSWERED] A solution has a 1 4 mole ratio of pentane to hexane The](https://media.kunduz.com/media/sug-question-candidate/20210607090612264131-3317355.jpg?h=512)
This isn't about memorizing facts for a test. This is about seeing the hidden order and intricate beauty in the world. It's about appreciating the invisible forces that shape our reality. Every time you see a flame, you can remember that dance of molecules, the precise ratio that makes it all possible.
And the best part? This is just the tip of the iceberg! The world of chemistry is filled with these fascinating relationships, these ratios that govern everything from the air we breathe to the stars in the sky. Each new concept you explore opens up a new window of understanding, a new perspective on the amazing universe we inhabit.
So, the next time you encounter a chemical formula or a reaction, don’t shy away. Lean in! Ask questions. Because within those seemingly complex symbols lies a world of wonder, a story waiting to be told, and a whole lot of inspiration. You might just find that understanding the mole ratio of oxygen to pentane is the spark that ignites a lifelong love affair with science. Keep exploring, keep questioning, and never stop being amazed by the magic of the natural world!