Is Cough Syrup Heterogeneous Or Homogeneous

Ah, cough syrup! That ubiquitous, syrupy elixir that’s been a staple in medicine cabinets for generations. We've all reached for it during those unwelcome bouts of sniffles and tickles. But have you ever paused, a spoonful poised, and wondered about the science behind its comforting consistency? Today, we’re diving into a surprisingly delightful question: Is cough syrup heterogeneous or homogeneous? And more importantly, what can its very nature inspire in us?
For the creative soul, the hobbyist, or even the curious casual learner, understanding the fundamental properties of everyday items can unlock a world of inspiration. Think of it this way: just as a painter mixes pigments to achieve the perfect hue, scientists mix ingredients to create solutions with specific properties. Cough syrup, in its quest to soothe, offers a fantastic real-world example of these principles. It’s a playground for understanding mixtures and how they behave, a concept that can fuel artistic endeavors, from conceptualizing color palettes to designing intricate patterns. For hobbyists exploring DIY projects or even just appreciating the world around them, it's a gentle nudge to look closer and ask "why?".
So, back to our question. Cough syrup, for the most part, is a homogeneous mixture. This means its ingredients are evenly distributed throughout, creating a uniform appearance and composition. When you look at a bottle of clear cough syrup, it appears as one single substance. There aren't visible clumps or separate layers. This uniformity is crucial for its medicinal function, ensuring each dose delivers the same concentration of active ingredients. Think of it like a perfectly blended smoothie – you can’t pick out individual berries or chunks of banana; it’s all one smooth, delicious entity. This characteristic is what makes it predictable and effective.
Must Read
However, some cough syrups, particularly those with suspensions like guaifenesin or decongestants, might present as heterogeneous. You might notice a slight cloudiness or a need to shake well before use. This indicates that the solid particles aren't fully dissolved but are dispersed throughout the liquid. While this might seem less "perfectly mixed," it serves a vital purpose for certain medications. It’s akin to a beautifully swirling nebula in space – distinct components contributing to a larger, dynamic whole. The key takeaway is that both homogeneous and heterogeneous properties in common items demonstrate clever design and scientific understanding.
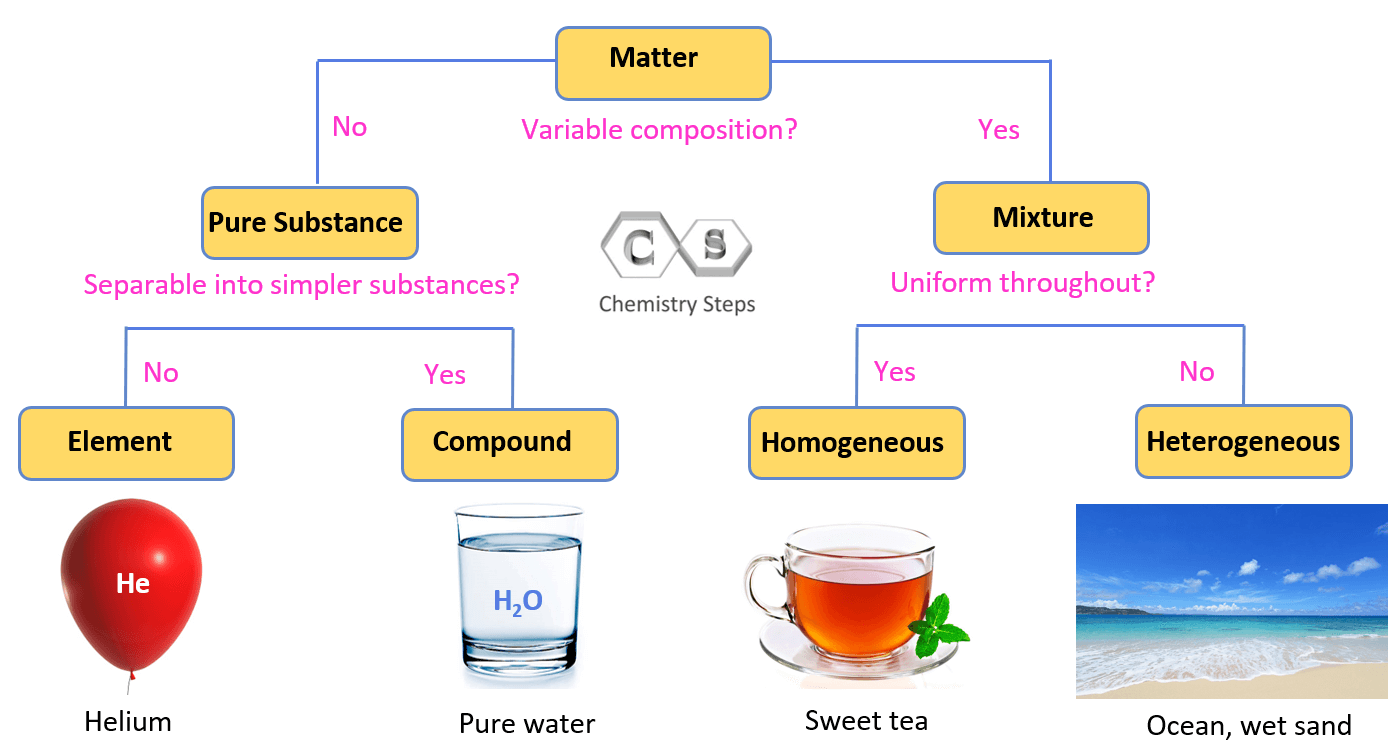
Curious to explore these concepts further? You don't need a lab coat! Try some simple at-home experiments. Take a glass of water and stir in a spoonful of sugar – observe how it dissolves completely (homogeneous). Then, try stirring in some sand – you'll see it settle (heterogeneous). You can even mimic the "shake well" aspect with a simple mixture of oil and water in a sealed jar. You’ll see them separate, requiring a vigorous shake to momentarily blend.
Ultimately, whether you’re contemplating the uniform beauty of a homogeneous solution or the dynamic interplay of a heterogeneous one, there's a quiet joy in understanding the world around us. It’s a reminder that even the most mundane objects hold fascinating secrets, waiting to be discovered and, perhaps, to spark your next great idea. So, the next time you reach for that familiar bottle, remember the science, embrace the curiosity, and enjoy the simple, inspiring power of a well-mixed mixture!








