Eight Components Present In Nucleic Acids

Alright, pull up a chair, grab your latte, and let's dish about something that sounds way more intimidating than it is: the eight magical components that make up nucleic acids. Yeah, yeah, I know, "nucleic acids" sounds like something you'd only encounter in a Mad Scientist's basement, probably bubbling ominously. But honestly, it's just DNA and RNA, the original superstar molecules of life. Think of them as the universe's tiny, incredibly important recipe books.
Now, these recipe books aren't just a jumble of random letters. They’re meticulously constructed, like the world’s most complex IKEA furniture, but with way fewer tiny screws and a lot more life-altering consequences if you mess up. And to build this molecular marvel, you need some key ingredients. We're talking about eight essential building blocks that are cooler than a penguin in a tuxedo. Let’s break ‘em down, shall we?
The Sugar Rush: Sweet, Sweet Pentoses!
First up, we’ve got the sugars. But not just any sugars, oh no. We’re talking about pentose sugars. Pent-a-what-now? It means they have five carbon atoms. Think of them as the sweet little scaffolding that holds everything together. Without these guys, your DNA and RNA would be flatter than a pancake left out in the rain.
Must Read
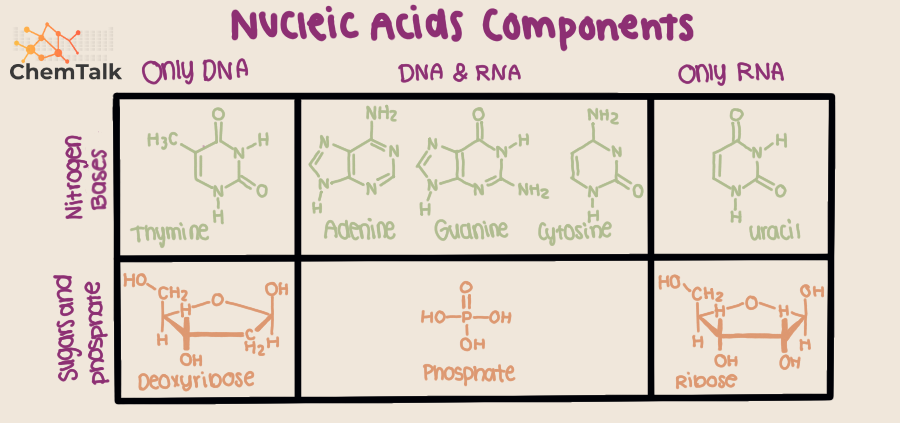
Now, here's where things get interesting. There are actually two main types of pentose sugars involved, depending on whether you’re dealing with DNA or RNA. For DNA, the star player is deoxyribose. The "deoxy" part? It means it’s missing a little something-something: an oxygen atom on a specific carbon. This little omission is actually a huge deal, making DNA more stable, which is pretty crucial for a molecule that’s supposed to keep your genetic blueprint safe for, you know, generations.
And then for RNA, we have ribose. Ribose is basically deoxyribose’s slightly more oxygenated sibling. It’s got that extra oxygen. This makes RNA a bit more reactive, which is perfect because RNA is more of the “working-class hero” molecule, hopping around doing various jobs, not necessarily needing the long-term stability of DNA. So, think of deoxyribose as the sturdy, reliable dad and ribose as the energetic, busy mom. Both essential!

The Phosphate Party: The Backbone Boogie!
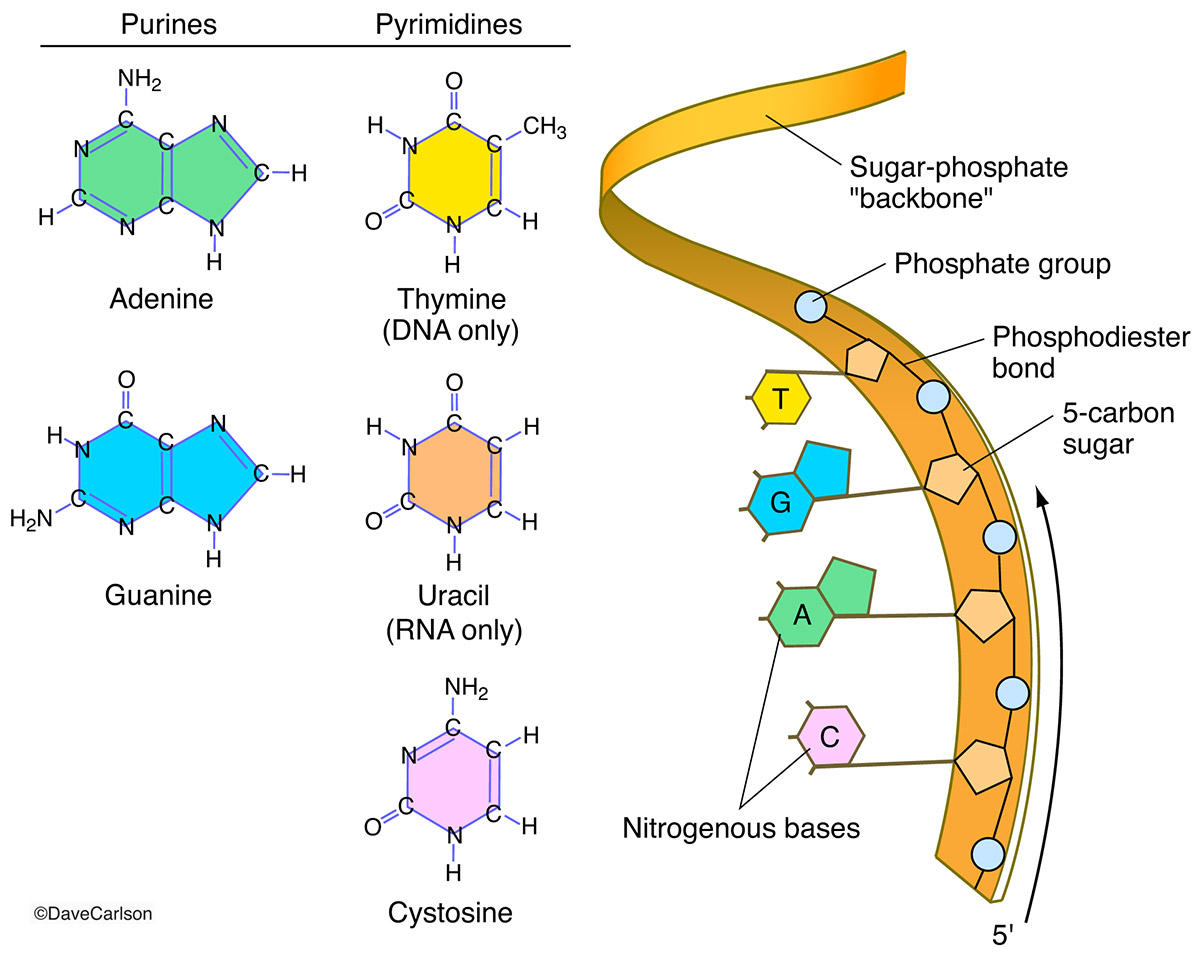
Next on our ingredient list are phosphate groups. These little guys are like the glue that holds the sugar scaffolding together. They form the phosphate backbone of nucleic acids, which sounds super rigid, right? But it's more like a flexible, powerful spine. Imagine tiny molecular springs connecting all the sugar units.
A phosphate group, in case you were wondering (and you totally were, admit it), is basically a phosphorus atom bonded to four oxygen atoms. Sounds simple, but these guys are negatively charged. This gives the entire nucleic acid molecule a negative charge. So, if you ever spilled your DNA on the floor, it would probably cling to the positively charged dust bunnies. Science!
These phosphate linkages are called phosphodiester bonds. Catchy, I know. They’re incredibly strong, which is good because you don’t want your genetic code falling apart when you’re doing your happy dance. These bonds are the unsung heroes, the silent workhorses ensuring the integrity of your DNA and RNA.
The Nitrogenous Bases: The Alphabet of Life!
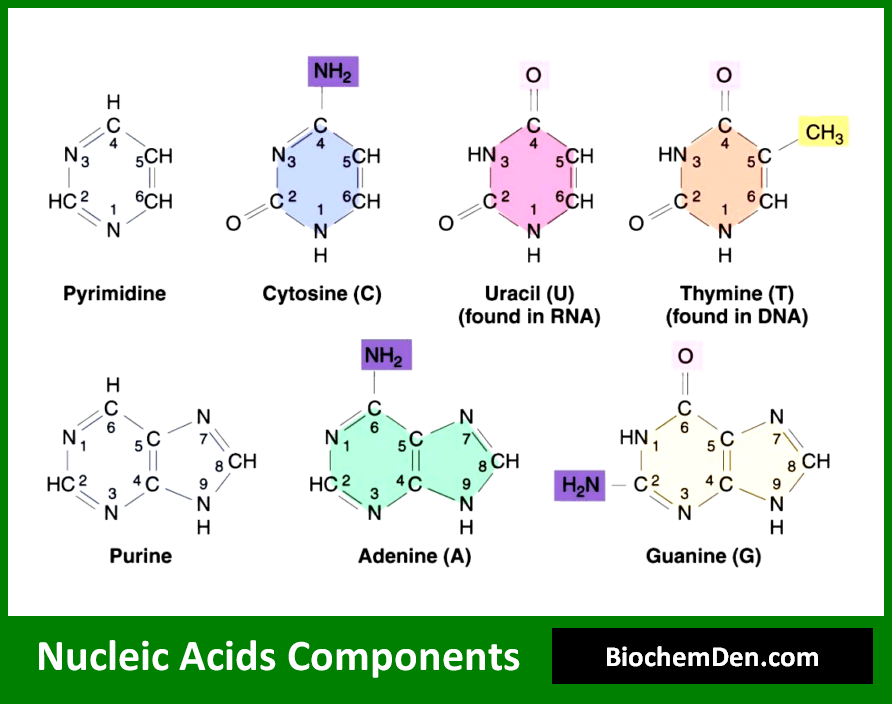
Okay, now for the really fun stuff: the nitrogenous bases. These are the actual letters of your genetic alphabet. They’re the part that carries the information, the codons, the secrets of who you are and why you have that weird obsession with collecting rubber ducks. There are five different types of these bases, and they come in two stylish flavors: purines and pyrimidines.
First, the purines! These are the double-ringed big shots. They’re like the rock stars of the base world. We have adenine (A) and guanine (G). They’re a bit larger and more complex. Think of them as the foundational pillars of your genetic code. They're present in both DNA and RNA.

Then, we have the pyrimidines. These are the single-ringed, more streamlined players. They're a bit simpler in structure. The main pyrimidines are cytosine (C), thymine (T), and uracil (U). Cytosine is a team player, showing up in both DNA and RNA. Thymine is DNA’s exclusive VIP guest – it's only found in DNA.
And here’s the plot twist! Uracil is RNA’s version of a party crasher, or maybe a substitute player. It’s found in RNA but not in DNA. So, when DNA says "T," RNA sometimes says "U." It’s like they have their own secret language, but we’ve figured it out. It’s all about keeping things organized and ensuring the right messages get sent. Clever, right?

The Grand Total: Eight Amazing Components!
So, let’s count our blessings, or rather, our components. We’ve got:
- Two types of pentose sugars: deoxyribose (for DNA) and ribose (for RNA).
- Phosphate groups, which form the backbone.
- Four nitrogenous bases that are commonly found: adenine (A), guanine (G), cytosine (C), and thymine (T) in DNA, and adenine (A), guanine (G), cytosine (C), and uracil (U) in RNA.
Wait, that’s only seven if you list them out individually like that! Ah, but remember the two types of sugars are distinct entities. So, deoxyribose is one component, ribose is another. Then we have the phosphate group. And then the four bases (A, G, C, and T/U depending on the nucleic acid). Thus, we have our magical eight components. It’s like a molecular Avengers assembly, each piece playing a vital role in the grand opera of life. Pretty neat for something so tiny you need a microscope to even think about seeing it!
So, next time you hear about DNA or RNA, just picture these eight essential components – the sugars, the phosphates, and the bases – all dancing together to create the symphony of you. And if that doesn't sound like a good story for your next café chat, I don't know what does!

:max_bytes(150000):strip_icc()/nucleotide_base-5b6335bdc9e77c002570743e.jpg)