Compare The Stability Of A Lithium Atom With

Okay, so imagine you've got your trusty old bicycle. It's been with you through thick and thin, seen you through countless trips to the grocery store, and probably endured a few questionable parking jobs. This bike, in its own way, is pretty darn stable. It's got its wheels firmly on the ground, its handlebars are where they should be, and it generally does what you expect it to do. Now, let's talk about atoms, specifically a lithium atom, and see how it stacks up against our bike buddy.
We're not talking about rocket science here, folks. We're just gonna peek under the hood of what makes things… well, things. And when it comes to stability, it’s like deciding if you’re going to binge-watch that new show for the fifth time or finally tackle that pile of laundry. Some things just want to stay put, while others are constantly looking for a bit of excitement.
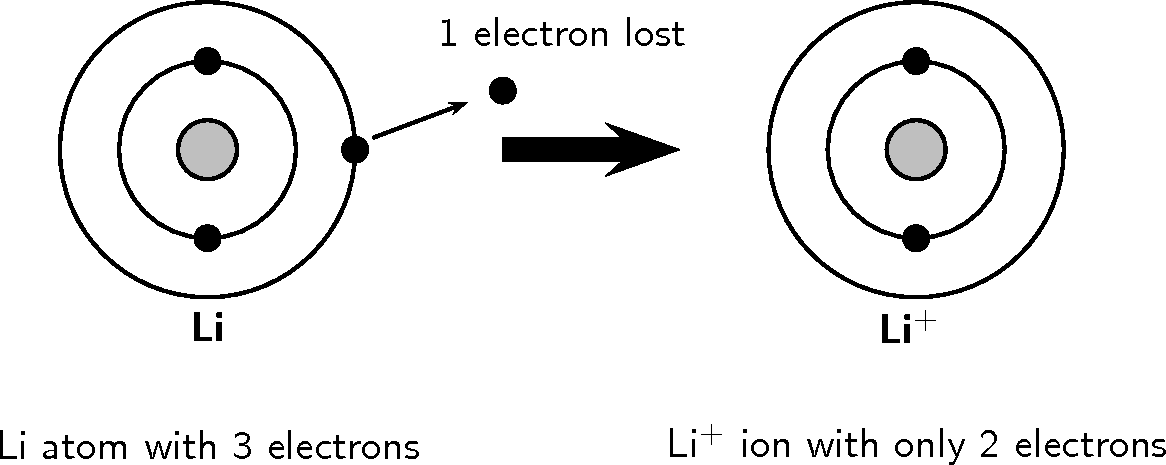
So, what's the deal with lithium? Think of a lithium atom as a tiny, energetic toddler. Seriously. It's got this one extra little electron zipping around its nucleus, like a toy it’s not quite sure what to do with. It’s like having that one rogue sock that always manages to escape the laundry basket. It’s just… there, and it's kind of disrupting the peace.
Must Read
The nucleus of the lithium atom is like the toddler’s very patient (or perhaps not-so-patient) parent. It’s got protons and neutrons chilling together, a bit like a comfy sofa. But then there’s this electron, this little rebel, doing its own thing. And this electron, this outermost electron, is what we call a valence electron. It’s the one that gets to play in the playground of the outside world, while the inner electrons are more like the kids who are happy to stay in the cozy living room.
Now, stability in the atomic world is all about having a happy, content electron configuration. Think of it like having a perfectly balanced set of shelves. Everything is in its place, and nothing is wobbling precariously. Atoms love that feeling of completeness, like when you finally find that missing piece to your jigsaw puzzle and everything just clicks into place. They want to have a full outer shell of electrons. It's their ultimate goal, their atomic nirvana.
Lithium, bless its little heart, is not quite there. It’s got its inner shell all snug and happy, like a perfectly arranged display of antique teacups. But that one valence electron? It's like a lone Lego brick that doesn't quite fit anywhere in the main castle. It's a bit of an outlier, a bit of an odd one out.
Because it has this extra electron that's not really doing much for its overall stability, lithium is like that friend who’s always eager to share their snacks. They’ve got something extra, and they’re just looking for someone to take it. This makes lithium very reactive. It’s not content to just sit there. It wants to do something with that electron. It wants to get rid of it to achieve that perfect, stable, full-shell configuration.

Compare this to, say, a noble gas, like Neon. Neon is the ultimate chill dude of the atomic world. Its outer electron shell is already full. It's like that friend who always brings their own snacks to the party and is perfectly happy just observing. Neon doesn't need to give anything away, and it doesn't need to take anything. It's just… there. Perfectly content. Completely stable. It's the atomic equivalent of a cat napping in a sunbeam, utterly unbothered by the world.
Lithium, on the other hand, is more like a squirrel who’s found a whole bag of nuts. It’s excited, it’s a little frantic, and it’s desperately trying to bury them all before anyone else can get them. It’s so eager to shed that one electron that it will readily give it up to any atom that’s willing to take it. This eagerness is why lithium is used in batteries – it's practically begging to give away its electron to make something happen.
So, when we talk about the stability of a lithium atom, we’re essentially talking about its internal desire for perfection. It’s not perfectly stable because it has this one electron that’s making it feel a bit… incomplete. It’s like having one squeaky hinge on an otherwise magnificent door. It works, sure, but it’s not perfect. And lithium is constantly striving for that perfection.
Think about it in human terms. You know those people who are just always looking for something more? They’ve got a good job, a nice house, but they’re still restless, always chasing the next big thing. That’s a bit like lithium. It’s not unhappy, it’s just not… fully satisfied with its current electron arrangement.

Now, imagine a truly stable atom. It’s like that perfectly brewed cup of coffee in the morning. It’s exactly what you need, when you need it. No fuss, no drama, just pure, unadulterated satisfaction. Noble gases are the royalty of this satisfaction. They’ve got their electron shells packed tighter than a sardine can, and they’re not about to let any of those precious electrons go, nor do they need any more. They’re the ultimate introverts of the chemical world, perfectly happy in their own little electron bubbles.
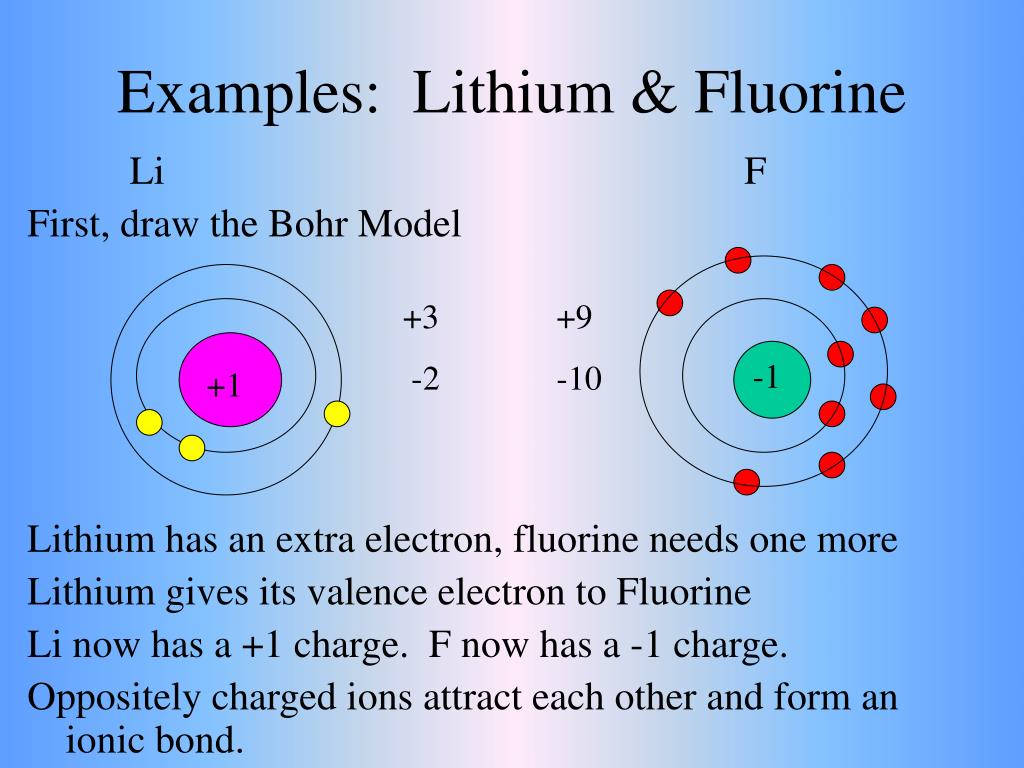
Lithium, by contrast, is the life of the party. It's the one who's always initiating conversations, always looking for interaction. That single valence electron is its social butterfly wing, ready to flit off and join another atom's dance. This makes lithium a highly reactive metal. It readily forms chemical bonds because it's so keen to achieve that electron configuration of its nearest noble gas neighbor (which, in lithium’s case, is Helium – a very stable character indeed!).
So, the stability of a lithium atom isn't about it being unstable in the sense of falling apart. It’s about its internal potential for change. It’s like a coiled spring, ready to unleash its energy. It's not going to spontaneously combust, but it's certainly not going to sit around doing nothing if there's an opportunity for a chemical reaction. It’s like a toddler who’s just discovered the joy of kicking a ball – they’re going to keep kicking it!
The key difference lies in that single, lonely valence electron. It’s the element that prevents lithium from being as supremely stable as, say, a neon atom or an argon atom. These noble gases are the ultimate in atomic contentment. They’ve got their outer electron shells completely filled, so they have no incentive to gain, lose, or share electrons. They are the hermits of the periodic table, perfectly happy in their solitude.
Lithium, however, is the friendly neighbor who’s always willing to lend a hand (or an electron, in this case). It has just one electron in its outermost shell, making it eager to shed it to achieve a more stable, filled shell configuration. This is why lithium is so reactive and forms compounds so easily. It's like a person who’s just finished a huge meal and is absolutely stuffed, looking around for someone to share their leftovers with.
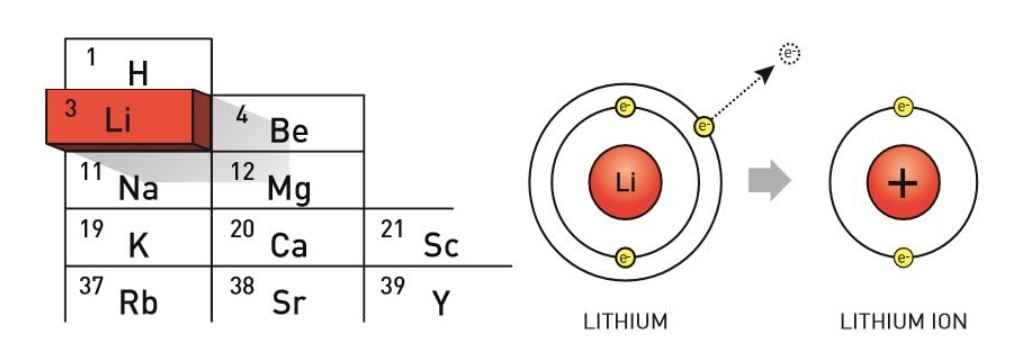
Think of it like this: Imagine you have a collection of beautifully arranged figurines on a shelf. That's a stable arrangement. Now, imagine you have one extra figurine that doesn't quite fit anywhere. It’s just sitting there, looking a bit out of place. You might be tempted to give it to a friend who collects that particular type of figurine. That's your lithium atom, wanting to offload its extra electron.
The atomic world, much like our own, has its characters. Some are content to be left alone, perfectly balanced and unbothered. Others are a bit more… dynamic. They have a surplus of energy, a desire for interaction, and a tendency to make things happen. Lithium falls firmly into the latter category.
So, when we’re comparing the stability of a lithium atom to, say, the stability of a brick wall, it’s important to understand what we mean by ‘stability’. A brick wall is stable because it’s heavy, dense, and not going anywhere unless you actively try to move it. It’s not going to spontaneously decide to rearrange its bricks. Lithium, on the other hand, is stable in the sense that it exists as a distinct element, but it’s dynamically stable. It’s not static; it’s always on the verge of interaction.
It’s like comparing a rock to a puppy. The rock is incredibly stable. It's been there for ages and will likely be there for ages more, doing nothing in particular. The puppy? It’s stable in that it’s a puppy, but it’s got boundless energy, it’s always wanting to play, and it’s prone to doing all sorts of unexpected things. That’s your lithium atom. It’s got that single, easily lost electron, making it a prime candidate for chemical reactions. It’s like having a single loose button on your favorite jacket – it’s not going to cause the whole jacket to unravel, but you’re constantly aware of it and might eventually decide to sew it back on or, in lithium’s case, give it away.

The desire for a full outer electron shell is the atomic equivalent of wanting to complete a set. Think of collecting trading cards. You have a few cards, and you’re happy. But when you get that last card to complete the whole set, oh boy, that’s a feeling of pure satisfaction! Atoms are no different. Lithium has that one electron that makes its outer shell almost complete, but not quite. It’s like having 99 cards out of 100. You’re so close, but you’re still looking for that final one.
This inherent drive to achieve a stable electron configuration makes lithium highly electropositive. It's practically yelling, "Take my electron! I don't need it!" This is in stark contrast to nonmetals like fluorine, which are highly electronegative and are desperate to gain an electron to achieve their own stable configuration. It’s like a really generous person handing out gifts versus a person who’s desperately trying to receive one.
So, the stability of lithium is a funny thing. It's stable enough to exist, to be a lithium atom. But it's not inertly stable like a noble gas. It's more like a well-trained athlete who's ready for competition. They’re physically sound and capable, but they’re looking for the opportunity to perform. Lithium is looking for an atom to react with. It’s not going to cause trouble on its own, but put it next to something that needs an electron, and watch the sparks fly (metaphorically, of course!).
Ultimately, the 'stability' of an atom is a spectrum. Lithium sits on the more reactive end of that spectrum. It's not a wilting flower, but it's certainly not a stoic rock either. It's got that one little electron doing its best to make things happen, and that’s what makes it so fascinating and so useful in our everyday lives, from the batteries in our phones to the alloys in our cookware. It’s all thanks to that one energetic, slightly restless, lithium atom, just looking for a good chemical home.
So next time you’re using a device powered by lithium, you can imagine that tiny atom, not quite perfectly content, but perfectly ready to do its job. It’s the atomic equivalent of that friend who’s always up for an adventure, a little unpredictable, but ultimately, incredibly valuable.