Are Anions Smaller Than Their Parent Atoms

Have you ever wondered what happens when an atom gets a little something extra? You know, like when it decides to pick up an extra electron? It's like a tiny atom party, and things can get surprisingly interesting! Today, we're diving into the super cool world of anions and asking a question that might sound a little tricky: are these electron-gaining atoms smaller than their parent atoms? Get ready for a fun adventure into the miniature universe of chemistry!
Imagine an atom as a tiny solar system. You have the nucleus in the center, like the sun, packed with protons and neutrons. Then, zipping around the nucleus, you have the electrons, like planets orbiting their star. These electrons are crucial. They're constantly moving, occupying different energy levels, or "shells," around the nucleus. Think of them like little workers doing their jobs in designated areas.
Now, an atom is usually pretty happy and balanced when it has a specific number of electrons. It's like having just the right amount of workers for the factory. But sometimes, for all sorts of chemical reasons, an atom might decide to grab an extra electron. It's like a factory suddenly getting a new hire, someone who might be a bit eager and enthusiastic!
Must Read
When an atom gains an electron, it transforms into something called an anion. So, instead of just being a regular atom, it's now an ion with a negative charge. That little extra electron is a big deal! It joins the existing crowd of electrons buzzing around the nucleus. This is where things get really intriguing.
Think about those electron shells. They're like designated lanes on a highway. When a new electron arrives, it needs a place to go. It's not just going to stand around outside! It'll find a spot, often in the outermost shell. But here's the twist: this new electron doesn't just squeeze into an already crowded space without affecting anything. Oh no, it's a bit more dynamic than that!

When that extra electron arrives, it brings its negative charge with it. Now, all the electrons in the atom are negatively charged. And what do like charges do? They repel each other! It's like having two magnets with their negative ends facing each other – they push apart. So, this new electron starts to push away the other electrons. It’s a bit like a polite but firm nudge to give everyone a little more personal space.
This "personal space" requirement has a fascinating effect. The electrons, which were already occupying certain energy levels, are now being pushed slightly further out from the nucleus. They can't get too far, because the positive charge of the protons in the nucleus is still holding onto them. But that repulsive force from the new electron makes them spread out a bit more. It's like a crowd at a concert, and when a few more people join, everyone naturally shifts outwards to make a bit more room. They don't abandon their seats, but they might lean back a little further.
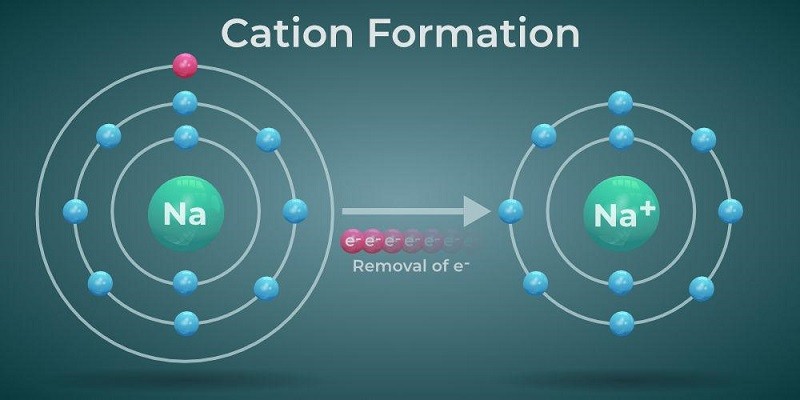
So, what does this spreading out of electrons mean for the overall size of the atom? Well, the "size" of an atom is generally measured by how far its outermost electrons extend. If those outermost electrons are pushed a little further out, the entire electron cloud gets a bit bigger. It's like inflating a balloon slightly more – the outer edge expands.
Therefore, in most cases, when an atom gains an electron and becomes an anion, it actually becomes larger than its original, neutral parent atom. Isn't that neat? It's the opposite of what you might intuitively expect! You might think adding something would make it denser or more compact, but in the world of atoms and electrons, adding an electron often leads to expansion.

This phenomenon is one of the many subtle yet profound reasons why chemistry is so endlessly fascinating. It’s these tiny, almost imperceptible changes at the atomic level that lead to all the different properties of elements and how they interact. The behavior of a tiny electron can influence the size of an entire atom, and that, in turn, affects how that atom behaves in reactions.
Think about it: a single, extra electron. It's so small, almost insignificant. Yet, its presence can subtly rearrange the entire electron system of an atom, causing it to swell just a little. It's like a tiny pebble dropped in a pond, creating ripples that spread outwards. The analogy is quite fitting!

This is why studying chemistry is so rewarding. It's about understanding these fundamental interactions. It’s about realizing that even the smallest things can have significant impacts. So, the next time you encounter the term anion, remember this little secret: it's often a slightly bigger, more spread-out version of its parent atom, all thanks to the power of that extra electron and the wonderful push-and-pull of electrical charges.
It's a captivating dance of forces, a miniature ballet of charged particles. The nucleus provides the steady gravitational pull, while the electrons, with their repulsions and attractions, create a dynamic, ever-shifting cloud. And when an anion forms, it's a testament to how adding a single element can alter the entire shape and size of this delicate atomic structure. It’s a beautiful reminder that sometimes, adding more can lead to a bit of expansion, and that’s perfectly natural in the tiny, exciting universe of atoms!