Why Is Formic Acid Stronger Than Acetic Acid

Hey there, science curious pals! Ever feel like you're just muddling through, not quite reaching your full potential? Well, guess what? Even a humble little acid can have a similar existential crisis! Today, we're diving into the surprisingly fun world of acids, specifically why formic acid, the zippy little guy, packs a bigger punch than its more laid-back cousin, acetic acid (that's the stuff in vinegar, by the way!). No complicated jargon, just pure, unadulterated chemical charm!
So, picture this: you've got two molecules, right? They're like tiny building blocks that make up everything. Formic acid and acetic acid are both part of a big, happy family called carboxylic acids. Think of them as siblings. They share a lot of the same awesome features, but they also have their own unique quirks. And it's these quirks that make all the difference!
Now, when we talk about acids, we're not talking about something that’s going to dissolve your kitchen counter (unless, you know, you’re dealing with some really serious stuff, which we’re not today!). We’re talking about their ability to be a bit… giving. Acids like to share a hydrogen ion. It’s like they’re saying, "Here, take this little bit of me! It'll make you feel more stable!"
Must Read
The strength of an acid is all about how readily it’s willing to share that hydrogen ion. The easier it is for them to let go, the stronger the acid. Makes sense, right? It’s like being a generous friend – the more eager you are to share your last cookie, the more generous you’re considered.
So, why is formic acid the overachiever in this little scenario? It all comes down to the microscopic architecture, the tiny little details that make each molecule special. Imagine our two acid siblings are trying to hold onto their hydrogen ion. Formic acid has a really straightforward structure. It’s like a minimalist apartment – clean, simple, and nothing extra to get in the way.
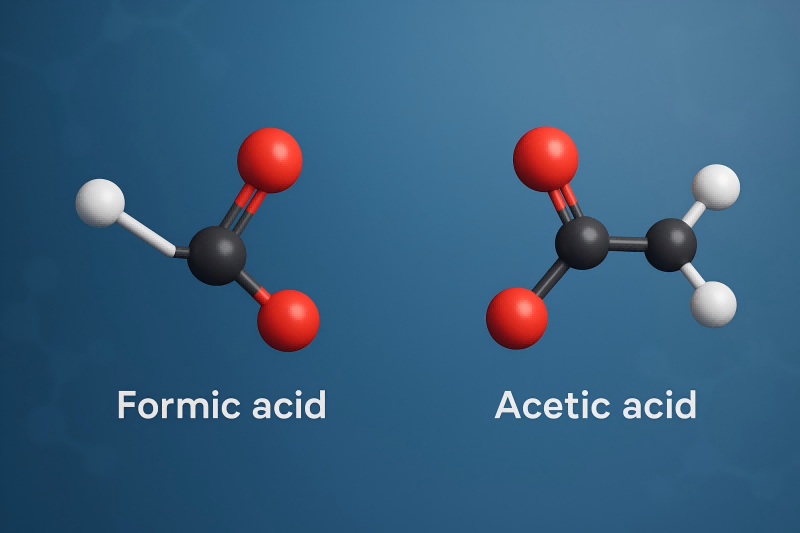
Acetic acid, on the other hand, has a little extra something attached. It's got a whole methyl group hanging around. Think of it like a comfy armchair in that apartment. This armchair, while cozy and perhaps even contributing to the overall stability of the molecule, also makes it a tiny bit harder for the hydrogen ion to escape. It’s like the hydrogen ion is a guest who’s gotten a bit too comfortable in the armchair and doesn’t want to leave!
This seemingly small difference in structure has a big impact. The methyl group in acetic acid has a slight tendency to push electrons towards the rest of the molecule. This “electron-donating” effect makes the bond between the hydrogen and the oxygen a little bit stronger. So, the hydrogen ion is like, "Hmm, this is a pretty secure relationship. Maybe I’ll stick around for a bit."

Formic acid, lacking this extra electron-donating group, has a less stable connection with its hydrogen ion. It’s like that hydrogen ion is standing at the door, keys in hand, ready for an adventure. This makes formic acid more likely to release that hydrogen ion when it’s in a suitable environment, like water. And poof! It becomes a stronger acid.
Think about it like this: you’re trying to get your friend to come out and play. If they’re already sitting on the couch with a blanket and a good book (acetic acid with its comfy methyl group), it might take a bit more convincing. But if they’re just standing by the door, ready to go (formic acid), a simple invitation is all it takes!

This difference isn't just some abstract chemical concept; it has real-world implications! Formic acid, being stronger, is actually used in some really interesting ways. It's used in the tanning of leather, which is pretty neat when you think about it. It’s also found naturally in the sting of ants – hence the name "formic," from the Latin word for ant, "formica." So, the next time an ant gives you a little nip, you can impress yourself by knowing it’s a tiny dose of strong acid!
Acetic acid, being milder, is great for everyday things. It helps preserve food (hello, pickles!), and it's the backbone of salad dressings. It’s the reliable friend who’s always there, doing its job without any fuss.

It’s fascinating, isn't it? How a few extra atoms, a slight rearrangement of things at the molecular level, can create such a difference in behavior. It's a gentle reminder that even the smallest details matter. And that sometimes, a little bit of extra "push" or a simpler, unadorned structure can lead to greater things.
This whole formic versus acetic acid showdown is a fantastic example of how understanding the "why" behind things can be incredibly satisfying. It’s not about memorizing facts; it’s about seeing the elegant logic of the universe at play, even in the smallest of molecules. It sparks a curiosity, doesn't it? You start looking at the world around you and wondering, "What makes this thing tick? What are its little secrets?"
So, don't be afraid to dig a little deeper, ask those "why" questions, and explore the fascinating world of chemistry, or any subject that piques your interest! You never know what amazing insights and fun facts you might uncover. The universe is full of these little marvels, waiting for you to discover them. So go forth, be curious, and let the joy of learning light up your path!