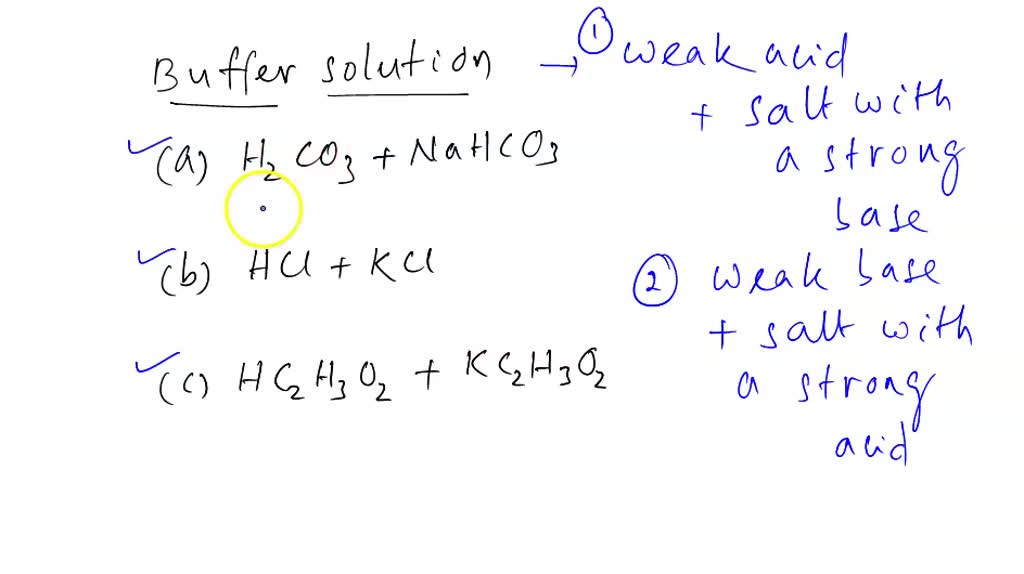
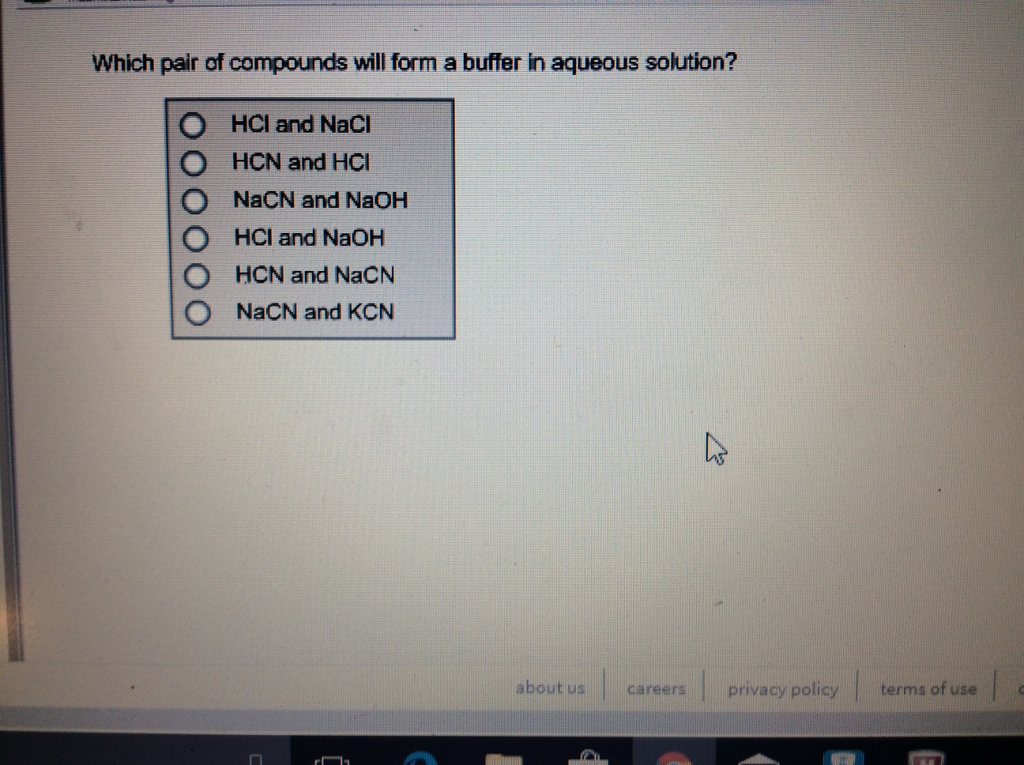
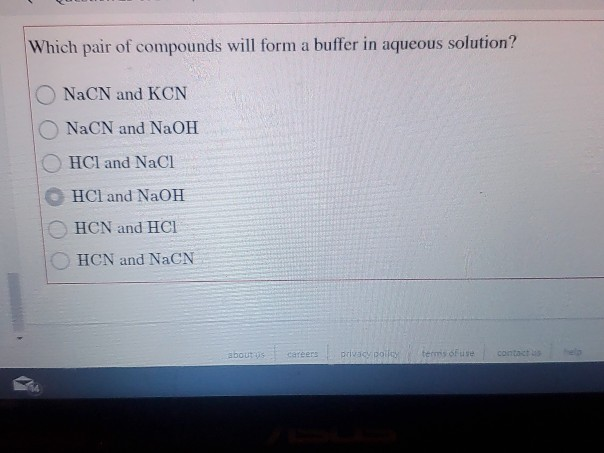
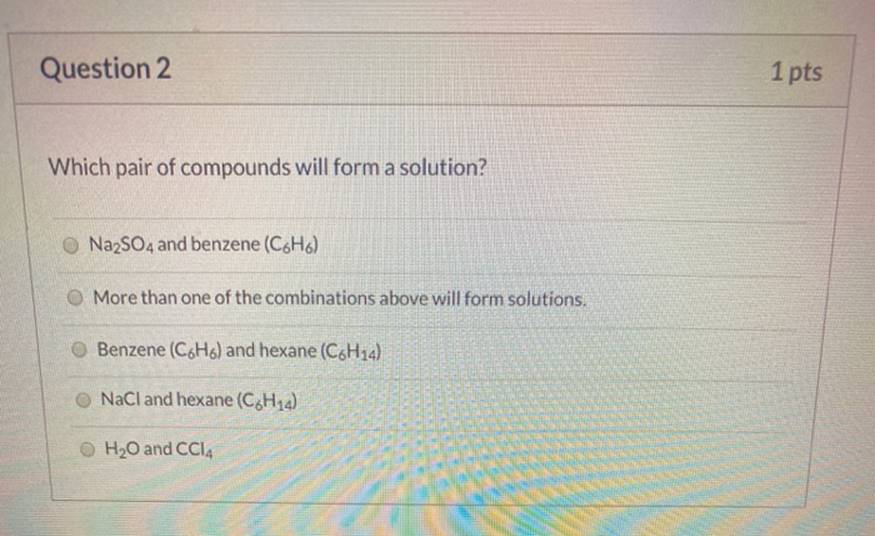
Which Pair Of Compounds Will Form A Solution
Have you ever wondered why oil and water just don't mix, but salt disappears into water like magic? It's all about finding the right partners when it comes to making solutions! This little bit of science isn't just for chemists in lab coats; it's a fun and useful peek into how the world around us works. Understanding which compounds will happily join forces to form a solution can be surprisingly handy, whether you're whipping up a homemade cleaner, brewing a perfect cup of tea, or just satisfying your curiosity about everyday phenomena.
For beginners, this topic is a fantastic entry point into the world of chemistry. It demystifies common observations and replaces confusion with understanding. Families can turn this into a playful learning experience for kids, conducting simple experiments at home that are both educational and entertaining. Imagine the delight on a child's face as sugar "vanishes" into water! Hobbyists, from bakers to gardeners, can leverage this knowledge to improve their crafts. Knowing how to dissolve fertilizers for plants or create even dough consistency in baking all comes down to understanding how different ingredients interact.
The core principle boils down to a simple phrase: "like dissolves like." This means that polar substances tend to dissolve in other polar substances, and nonpolar substances dissolve in other nonpolar substances. Water, for instance, is a polar molecule. Think of it like having a slightly positive end and a slightly negative end. Salt (sodium chloride) is also polar, so its charged particles are easily attracted to the charged ends of water molecules, causing it to dissolve. Oil, on the other hand, is nonpolar. Its molecules are balanced and don't have distinct positive or negative ends. Because oil and water have different "personalities" (polar vs. nonpolar), they don't attract each other and stay separate.
Must Read
Let's look at a few more examples. Ethanol (the alcohol in drinks) is polar, which is why it mixes so well with water – think of alcoholic beverages. However, trying to dissolve oil in water is like trying to mix sand and marbles; they just don't want to get together. Another variation is when you have something that's a bit of both, like soap. Soap molecules have a polar "head" that likes water and a nonpolar "tail" that likes oil, making them excellent at bridging the gap and helping to clean grease off dishes!

Getting started with this is super easy. You don't need fancy equipment. Grab some water (your universal solvent for many common substances!), a bottle of rubbing alcohol, some cooking oil, and a few common household items like salt, sugar, and even baking soda. In clear containers, try mixing small amounts of these substances. Observe what happens. Does it disappear? Does it form layers? Simple observation is your best tool here. You can even try mixing the alcohol and oil – you'll find they do mix!
Exploring the world of solutions is a fantastic way to engage with science in a tangible and enjoyable way. It's about understanding the invisible connections that make everyday things happen. So next time you're making lemonade or cleaning up a spill, take a moment to appreciate the delightful chemistry at play!