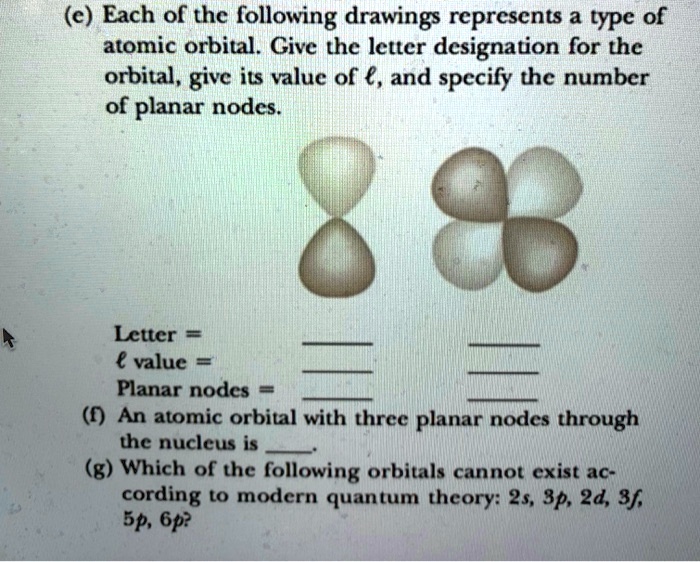
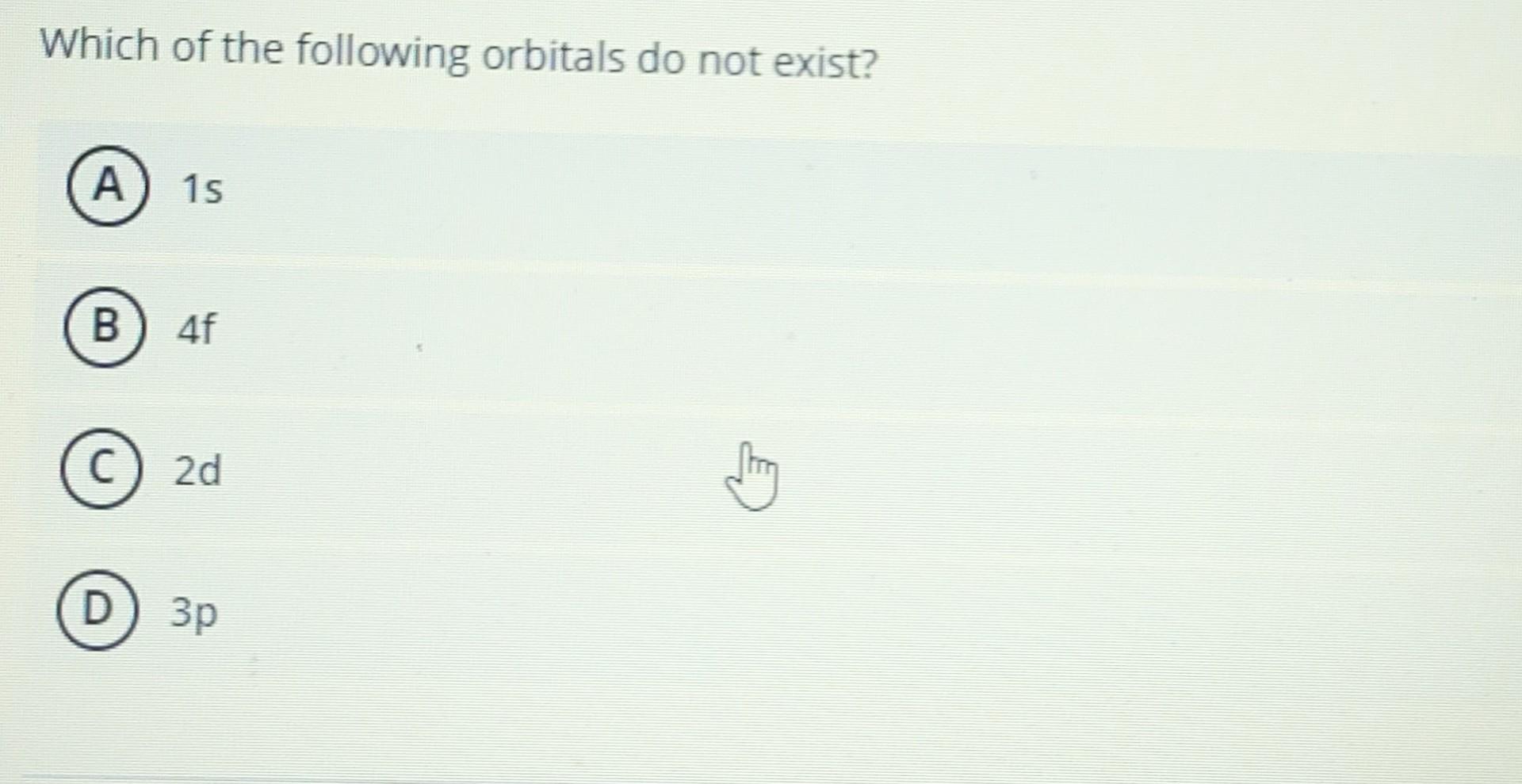
Which Of The Following Orbitals Cannot Exist
Hey there, curious minds! Ever feel like the universe is playing a little game of hide-and-seek with its secrets? Well, sometimes, it’s not about finding something hidden, but about realizing something can’t be there in the first place. Today, we’re diving into a fun little puzzle from the world of atoms that’s surprisingly… well, electrifying!
We’re talking about atomic orbitals, those quirky zones where electrons love to hang out. Think of them like the rooms in a super-tiny, invisible house. Each room has a specific shape and energy level. We’ve got the simple, spherical 's' rooms, the more complex 'p' rooms that look like dumbbells, and the super-fancy 'd' and 'f' rooms that are like modern art sculptures. Pretty neat, right?
But here’s where the fun begins. Imagine you’re trying to build a house, and you’re given a blueprint. The blueprint tells you what kind of rooms are possible. Now, what if someone handed you a sketch of a room that just… doesn't make sense according to the rules of architecture? That’s essentially what we’re going to explore: which of these atomic "rooms" are the universe's architectural impossibilities? It’s like a cosmic scavenger hunt where the prize is understanding a fundamental law of nature!
Must Read
The Case of the Impossible Orbitals
So, the big question is: Which of the following orbitals cannot exist? We’re going to throw some possibilities at you, and you, my clever reader, get to be the detective! These aren't just random letters; they represent specific shapes and energy levels. We've got our familiar friends: 1s, 2p, 3d. Then there are some that might make you scratch your head. What about a 1p orbital? Or a 2s? Or perhaps a 3f?
Let's break down why some of these are perfectly fine and others are, well, a cosmic no-go. It all boils down to a few simple rules that govern how electrons can arrange themselves. Think of these rules as the strict zoning laws of the atom!
The Quantum Numbers: The Atom's Address System
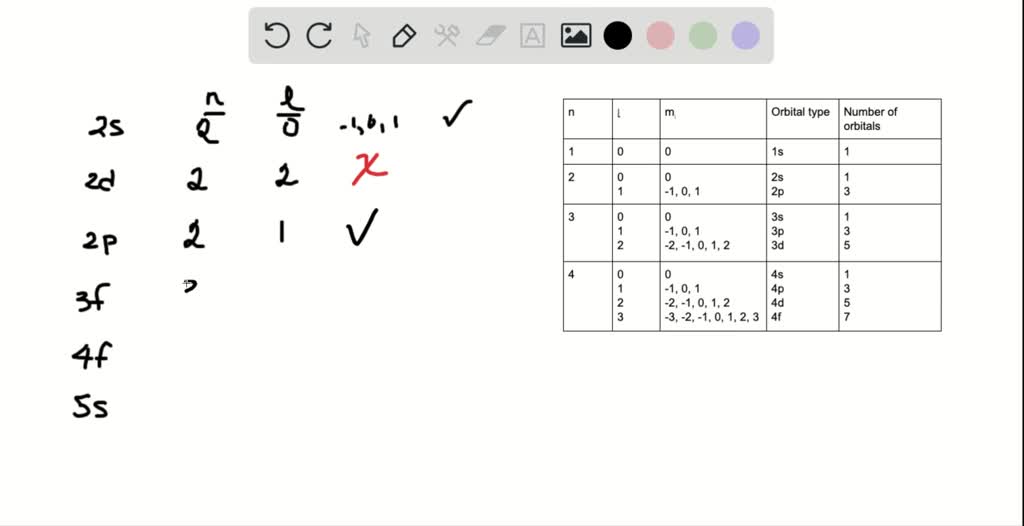
To understand which orbitals are real and which are imaginary, we need to talk about quantum numbers. Don't let the fancy name scare you! They’re basically like an electron’s full address. There are four of them, and each one gives us a piece of information about where an electron is and how it behaves.

The first one, the principal quantum number (n), tells us the main energy level, or shell, the electron is in. Think of it as the floor number in our atomic apartment building. Higher numbers mean higher energy and generally further from the nucleus. So, n can be 1, 2, 3, and so on, going up infinitely (in theory!). This one is pretty straightforward, right?
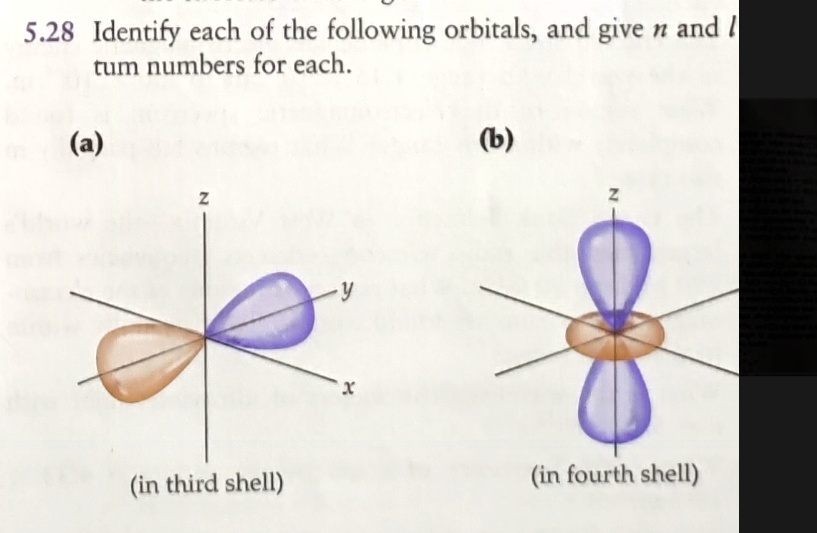
Next up is the angular momentum quantum number (l). This is the one that dictates the shape of the orbital, our room's design. This is where things get really interesting! For a given principal quantum number (n), the possible values of l range from 0 up to (n-1). So, if n is 1, l can only be 0. If n is 2, l can be 0 or 1. If n is 3, l can be 0, 1, or 2. See the pattern?
What do these 'l' values mean in terms of shapes? Well, l=0 corresponds to the spherical 's' orbitals. l=1 gives us the dumbbell-shaped 'p' orbitals. l=2 gives us the more intricate 'd' orbitals, and l=3 gives us the even more complex 'f' orbitals. So, the value of 'l' directly tells us what kind of orbital we're dealing with.
The third quantum number is the magnetic quantum number (m_l). This one describes the orientation of the orbital in space. Think of it as which way the room is facing – is the dumbbell pointing along the x, y, or z axis? For a given 'l', m_l can range from -l to +l, including 0. So, if you have a 'p' orbital (l=1), you can have m_l values of -1, 0, and +1, which correspond to the three different p orbitals (p_x, p_y, p_z).

And finally, the spin quantum number (m_s). This one is pretty simple: it just tells us the intrinsic angular momentum of an electron, often thought of as its "spin" – either spin up or spin down. Electrons are a bit like tiny magnets, and they can spin in one of two directions. So, m_s can only be +1/2 or -1/2.
Putting the Rules to the Test!
Now, let’s play the guessing game with our hypothetical orbitals. Remember the rule: for a given 'n', 'l' can only go up to 'n-1'. This is our golden rule, the cosmic handshake that makes an orbital valid.
Let's consider a 1p orbital. Here, n=1. According to our rule, the maximum value for 'l' should be n-1, which is 1-1 = 0. But here we have l=1. Oops! That doesn't fit the rules of the universe's architectural firm. There’s no room for a 'p' orbital on the first floor (n=1) of the atomic building. It's like asking for a two-story house on a single-plot lot – it just doesn't compute!
So, the 1p orbital? It cannot exist. It's a phantom, a theoretical blip that violates the fundamental laws of quantum mechanics. Pretty cool, right? We just figured out an impossibility!
What about the others? Let's check them out to make sure you're a seasoned orbital detective.
A 2s orbital. Here, n=2. The maximum 'l' allowed is n-1 = 2-1 = 1. We have l=0 for an 's' orbital. Is 0 less than or equal to 1? Yes! So, a 2s orbital is perfectly valid. It's a spherical room on the second floor. Totally normal!
A 3f orbital. Here, n=3. The maximum 'l' allowed is n-1 = 3-1 = 2. We have l=3 for an 'f' orbital. Is 3 less than or equal to 2? Nope! Uh oh. This is another one that, according to the rules, cannot exist. We've reached the f-orbitals at n=4. Think of it like needing a higher floor number to accommodate those super-complex f-shapes. They just won't fit on the lower levels!
Now, let's clarify something. You might see some higher energy levels where f-orbitals do exist, like in the 4f orbitals. But the key is that the 'l' value must be less than or equal to 'n-1'. For a 3f orbital, n=3 and l=3. Since 3 is not less than or equal to 3-1 (which is 2), it's a no-go. It’s all about that relationship!
The beauty of understanding these "impossible" orbitals isn't about dwelling on what's not there. It's about appreciating the elegant, underlying order of the universe. It’s like a perfectly designed game with incredibly consistent rules. And who doesn't love a game with fair rules, even if they lead to some curious impossibilities?
Making Science Fun and Electrifying!
This stuff might sound a bit abstract, but it's the bedrock of everything around us! The way atoms bond, the colors we see, the very functioning of our bodies – it all hinges on these quantum rules and the orbitals electrons occupy. When you grasp these concepts, you're not just memorizing facts; you're unlocking a deeper understanding of the world's intricate dance.
It’s a fantastic reminder that even in the vastness of the cosmos and the tininess of atoms, there’s a profound logic at play. And the more we learn about that logic, the more we can appreciate the marvel of existence. So, next time you hear about 's', 'p', 'd', or 'f' orbitals, remember the cosmic building codes and the orbitals that are simply not in the architectural plans!
Learning about these seemingly small details can open up a universe of understanding. It’s a journey of discovery that’s not just about textbooks, but about seeing the world with fresh, inspired eyes. Keep asking questions, keep exploring, and who knows what other amazing cosmic impossibilities (or possibilities!) you'll uncover. The universe is full of wonders, and the more you learn, the more you realize how much more there is to be amazed by!



:+d+orbitals+can+exist+on:.jpg)