Which Event Is An Example Of Vaporization

I remember this one scorching summer day when I was a kid. We were at the local park, and my mom had packed a picnic. She’d made this absolutely delicious watermelon, chilled to perfection. We’d sat it out on the checkered blanket, and as the sun beat down, I noticed something… weird. Little beads of water were forming on the outside of the plastic cooler. And then, as if by magic, those beads started to… disappear. Not drip, not run off, but just… vanish. I was convinced the sun was eating our picnic supplies! My mom, bless her patient soul, just chuckled and explained it was something called condensation. But what about the opposite of that? What about when things, well, become air?
That’s where we get to the really fun stuff, the topic of today’s little chat: vaporization. It sounds super science-y, I know, but stick with me because it’s actually happening all around you, all the time. And once you start noticing it, you’ll be pointing it out like I used to point out the disappearing water beads.
So, What Exactly Is Vaporization?
Alright, let’s break it down. In its simplest form, vaporization is the process where a substance in a liquid or solid state changes into a gas or vapor. Think about it: you’ve got stuff, usually in a nice, organized, liquidy or solidy form, and then POOF! It decides to break free and become all floaty and diffuse, like a ghost at a rave.
Must Read
There are actually two main ways this can happen: evaporation and boiling. And while they both lead to the same end result – becoming a gas – they’re a little different in how they get there. It’s like taking the stairs versus taking the elevator. Both get you to the top floor, but the experience is… well, you get it.
Evaporation: The Chill Way to Become Air
Let’s start with evaporation. This is the more subtle, everyday kind of vaporization. It happens when a liquid turns into a gas without reaching its boiling point. Yep, you heard that right. It’s like that shy friend who gradually drifts away from the party instead of making a grand exit.
Think about a puddle after it rains. The sun comes out, it’s not exactly a raging inferno, but slowly, steadily, that puddle shrinks and eventually disappears. Where did the water go? It evaporated! The water molecules at the surface gained enough energy from the surroundings (even a little bit of sun or just the ambient air temperature) to break free from the liquid and float off as water vapor. It’s a gentle, persistent process.
This is also what happens when you hang your laundry outside. Those wet clothes gradually dry. The water molecules escape into the air. It’s a beautiful, low-energy way for things to become… gaseous. And isn’t it kind of neat that we rely on this natural process for clean socks?
Another classic example? That sweat on your skin when you’re working out or on a hot day. Your body is releasing liquid to cool you down, and as that sweat evaporates, it takes heat energy with it. That’s why feeling that cool breeze after sweating feels so good. It's your body's built-in air conditioning, powered by evaporation!

Boiling: The Dramatic Entrance
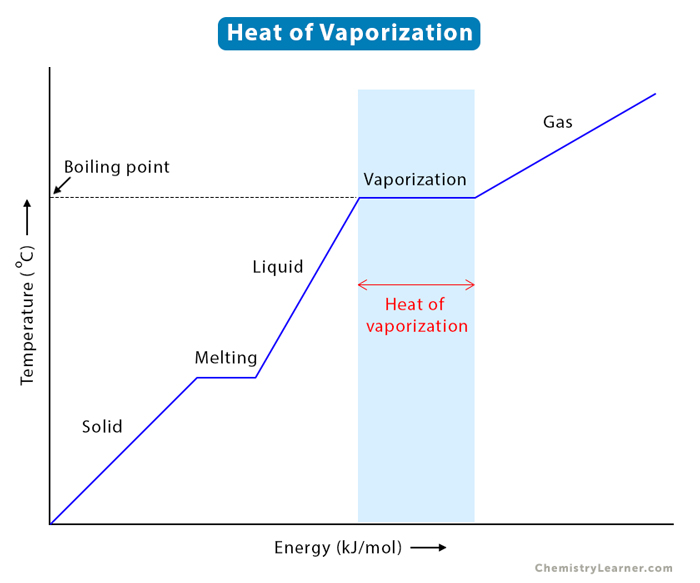
Now, boiling is a whole different kettle of fish. This is vaporization that happens when a liquid reaches its specific boiling point. This is the point where the liquid has enough energy to turn into a gas throughout the entire bulk of the liquid, not just at the surface.
You know this one instinctively. You put a pot of water on the stove, turn up the heat, and eventually, it starts to bubble and steam. Those bubbles you see forming are the water turning into steam (water vapor) from within the liquid, not just at the top. It’s a much more energetic, dramatic event. The liquid is practically throwing a party and inviting all its molecules to become gas.
The boiling point is specific for each liquid at a given pressure. For water, at standard atmospheric pressure, it’s 100 degrees Celsius (or 212 degrees Fahrenheit). If you’re at a higher altitude, the pressure is lower, and water actually boils at a lower temperature. Pretty wild, right? It’s like the water’s saying, “Oh, this is too easy, I can become gas even at a lower temp!” Conversely, if you increase the pressure, you’d need to heat water to a higher temperature to make it boil.
So, while evaporation is happening constantly, boiling is a more specific event tied to a particular temperature and energy input. You can have evaporation at room temperature, but you definitely can’t boil water at room temperature (unless you have some really fancy equipment that manipulates pressure, but let’s not get ahead of ourselves).
Which Event Is An Example Of Vaporization?
Okay, so now that we’ve got the definitions sorted, let’s get to the fun part: spotting vaporization in the wild! The question is, which event is an example of vaporization? The answer is… pretty much anything where a liquid or solid turns into a gas!

But since we’re talking about specific examples, let’s pick a few that really highlight the concept. And trust me, some of these might surprise you!
The Steaming Pot of Soup
This is a textbook, no-brainer example of boiling, which is a type of vaporization. You’re heating up your delicious chicken noodle soup (or whatever your comfort food of choice is), and you see that visible steam rising. That steam is water vapor escaping from the soup. The liquid soup molecules are gaining enough energy from the heat to break free and become a gas. It’s pure, unadulterated boiling happening right there. Just try not to burn your tongue on it!
Dry Ice Sublimating
Now, this one is a little cooler (literally!). You’ve probably seen dry ice in action, right? It’s that mysterious, smoky stuff that makes spooky Halloween decorations or keeps things super cold. Dry ice is solid carbon dioxide (CO2). And when it’s at typical atmospheric pressure, it doesn’t melt into a liquid. Nope, it goes straight from a solid to a gas. This process is called sublimation, and it’s a special kind of vaporization where a solid skips the liquid phase entirely and turns directly into a gas. So, when you see that fog rolling off dry ice, you're witnessing vaporization in its most direct, solids-to-gas form. Pretty neat, huh?
It’s important to note that sublimation is technically a form of vaporization, as it involves a phase change into a gaseous state. It's just a shortcut!
A Wet Towel Drying in the Sun
Remember our laundry example? This is another perfect illustration of evaporation. You’ve just finished your workout, you’re all sweaty, and you grab a towel. It’s damp, maybe even a little drippy. You hang it up, and later, it’s dry. The water molecules that were clinging to the towel’s fibers have absorbed enough energy from the surrounding air and sunlight to break free and become water vapor. This is evaporation at its finest, a gentle, continuous conversion of liquid to gas. No boiling involved, just good old-fashioned solar-powered drying.

The Disappearing Puddle
This brings us back to my childhood memory. A puddle after a rain shower is a prime example of evaporation. The water molecules on the surface of the puddle are constantly gaining and losing energy. When they gain enough energy from the sun or even just the movement of air, they can overcome the attractive forces holding them together as a liquid and escape into the atmosphere as water vapor. It’s a slow but sure process, and it’s happening all over the world, all the time, keeping our planet’s water cycle in motion.
A Perfume Bottle Left Open
Ever forgotten to put the cap back on your favorite perfume? Yeah, I’ve done that. And what happens? The scent starts to fade, and eventually, the liquid is gone. The alcohol and fragrance oils in the perfume are volatile, meaning they evaporate easily. At room temperature, these liquids gain enough energy to vaporize and diffuse into the air, carrying that lovely scent with them. It’s a more rapid evaporation than water, due to the nature of the liquids involved, but it’s still evaporation at play. It’s like the perfume is saying, "I’m too good to be contained!"
The Kettle Whistling
This is a classic! When you’re boiling water in a whistling kettle, the steam that builds up inside is under pressure. As this steam is forced out through the narrow opening of the whistle, it creates the distinctive sound. This is a direct result of boiling, and therefore, vaporization. The water has reached its boiling point, turned into steam, and is now escaping the confines of the kettle.
It’s not just the sound, though. The steam itself is a visible sign of water turning into a gas. It’s a constant process as long as the water is heated sufficiently.
Ice Cubes in a Freezer (over a long time)
This one is a bit more of a slow burn, but it’s still vaporization! If you leave ice cubes in your freezer for a really, really long time, you might notice they get a little… smaller. Or maybe they develop little frosty edges that seem to recede. This is because even at temperatures below freezing, some water molecules at the surface of the ice have enough energy to directly turn into water vapor. This process is called sublimation, just like with dry ice, but it happens much, much slower at these cold temperatures. It’s ice slowly, gradually, turning into air. It’s the ultimate slow-motion vaporization!

Why Does It Matter?
So, we’ve seen that vaporization is everywhere. From the dramatic boil of a kettle to the subtle disappearance of a puddle, it’s a fundamental process. But why should we care about how water or dry ice or perfume turns into a gas?
Well, for starters, it’s crucial for the water cycle. Evaporation from oceans, lakes, and rivers puts water vapor into the atmosphere, which eventually condenses to form clouds and then falls back to Earth as precipitation. Without vaporization, we wouldn’t have rain, snow, or even the drinking water we rely on!
It’s also responsible for cooling. As we mentioned with sweat, evaporation takes heat away from surfaces. This is why we use fans and air conditioning, and why plants transpire (which is essentially plant sweat!). It’s a natural cooling mechanism that’s vital for life on Earth.
And then there are the industrial and scientific applications. Distillation, for example, uses vaporization to separate liquids based on their boiling points. It’s essential in everything from producing distilled water to separating components in crude oil. Even the way our bodies function involves vaporization – think about the lungs releasing water vapor when we exhale!
So, the next time you see steam rising from a mug of tea, a puddle drying up, or even just notice your skin feeling cooler after a shower, remember: you’re witnessing vaporization in action. It’s a fundamental force of nature, quietly working its magic all around us, turning liquids and solids into the invisible gas that fills our world.
It’s a reminder that even the most common occurrences have fascinating scientific explanations. Keep your eyes open, and you’ll start seeing examples of vaporization everywhere you look. Happy vaporizing!