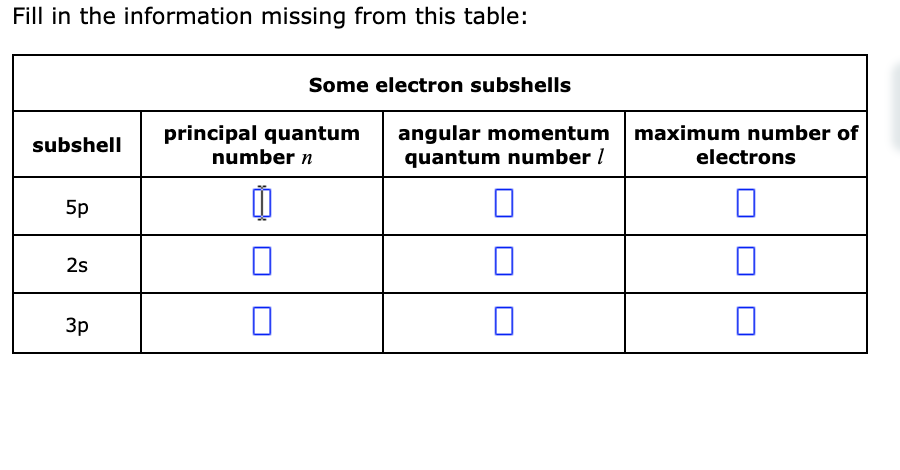
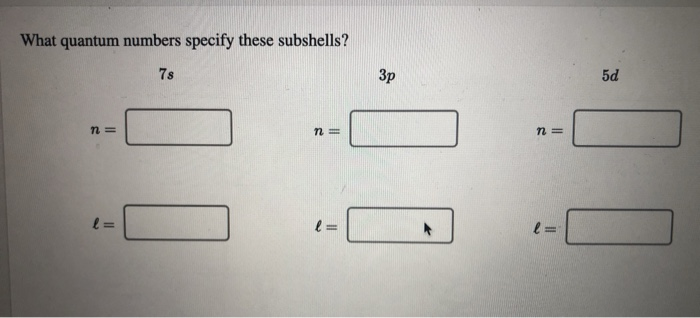
What Quantum Numbers Specify These Subshells

Hey there, ever feel like you're just… a bit fuzzy around the edges? Like you know you exist, but pinning down exactly where or how can be a whole thing? Well, welcome to the wild and wonderful world of quantum numbers, where even the tiniest particles get their own unique descriptions!
Think of it like this: if you were trying to describe your best friend to someone who'd never met them, you wouldn't just say "they're a person." That's way too vague! You'd say, "Oh, they're tall, have curly brown hair, usually wear bright colors, and are super into [their favorite hobby]." You're giving them a set of characteristics that make them them.
Atoms, those tiny building blocks of everything, are kind of like that. But instead of hair color and hobbies, they have a set of "quantum numbers" that tell us all about their electrons – those zippy little things that buzz around the nucleus like hyperactive toddlers. These quantum numbers are like an electron's personal ID card, giving us a surprisingly detailed picture of its life within the atom.
Must Read
The Principal Quantum Number: How Far Out Are We Talking?
Let's start with the big picture. The first quantum number, called the principal quantum number (symbolized by a little 'n'), is basically telling us how much energy an electron has and, consequently, how far away from the nucleus it's likely to be. Imagine a cozy apartment building. 'n' is like the floor number.
So, 'n=1' is the ground floor, closest to the bustling city center (the nucleus). Electrons here are pretty low-key, not a whole lot of energy. As 'n' gets bigger – 'n=2', 'n=3', and so on – we're moving up to higher floors. These electrons have more energy and are generally found further from the nucleus. It's like moving from a studio apartment to a spacious penthouse suite – more room, more energy!
Why should you care? Well, these energy levels are super important for how atoms interact. Think about fireworks! The brilliant colors you see are because electrons are jumping between these energy levels, absorbing and releasing light. The 'n' number dictates how high they can jump and how much energy they can let go of, which in turn determines the color of the show.
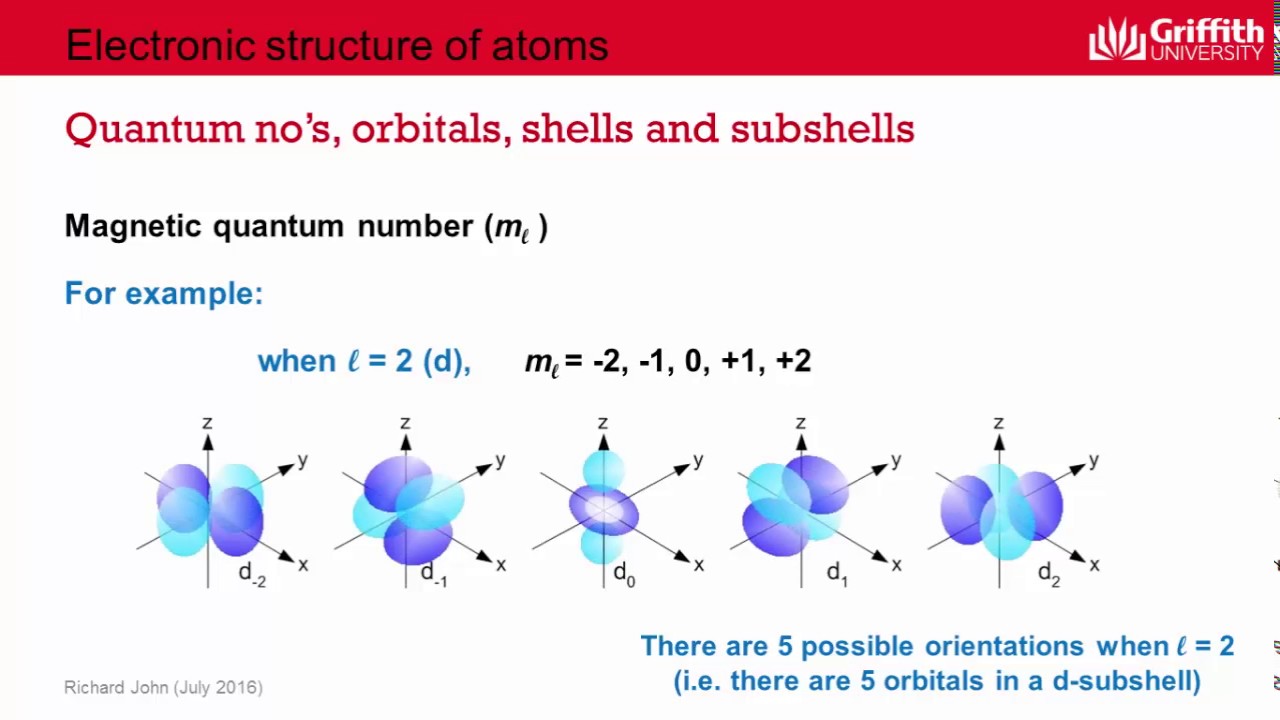
The Angular Momentum Quantum Number: The Shape of Things
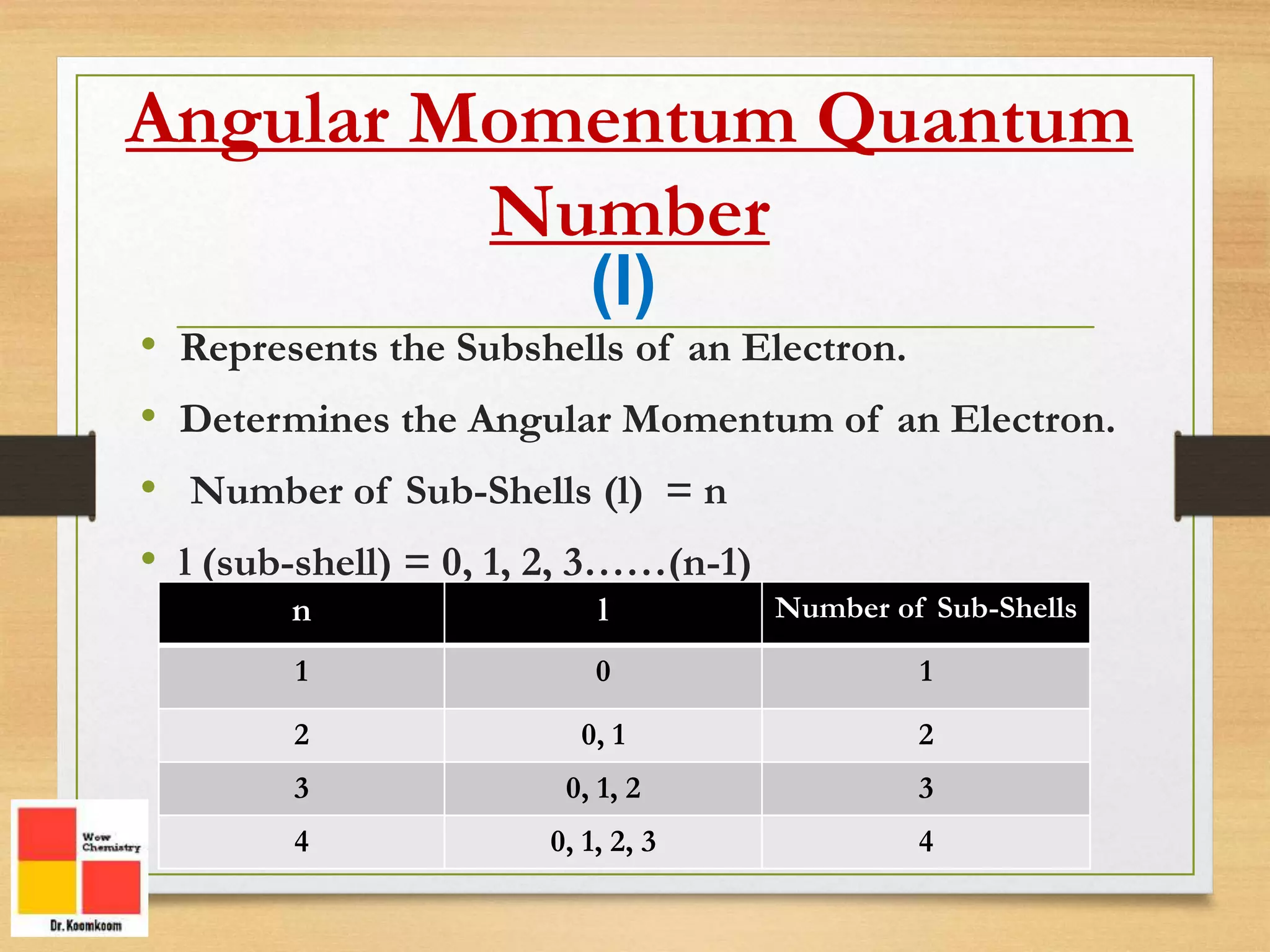
Now, if 'n' is the floor, the next number, the angular momentum quantum number (symbolized by 'l'), is like the shape of the apartment. Electrons don't just orbit the nucleus in perfect circles; they exist in different shapes of regions called orbitals.
This number 'l' tells us the shape of these orbitals. It can take on values from 0 up to 'n-1'. So, on the first floor ('n=1'), 'l' can only be 0. This corresponds to a spherical shape, like a perfectly round balloon. We call this an 's' orbital.
On the second floor ('n=2'), 'l' can be 0 (still a sphere, the '2s' orbital) or 1. When 'l' is 1, we get a different shape. These are called 'p' orbitals, and they look more like dumbbells or little infinity symbols, oriented in different directions in space. Think of them as more directional, like a spotlight.
As we go to higher floors ('n=3', 'n=4'), we get even more complex shapes – 'd' orbitals (which can be like clover leaves) and 'f' orbitals (even weirder shapes!).

Why care? These shapes are crucial for how atoms bond together to form molecules. It's like how different shaped puzzle pieces fit together. The 's', 'p', 'd', and 'f' orbitals have specific ways they can overlap and interact with other atoms, dictating whether you'll get water (H₂O), salt (NaCl), or something much more complicated. They’re the fundamental building blocks of chemistry’s architecture!
The Magnetic Quantum Number: Which Way Is It Pointing?
So we've got the energy level (the floor) and the shape of the orbital (the apartment's layout). What's next? The magnetic quantum number (symbolized by 'ml') tells us the orientation of these orbitals in space. Think of it as the direction the dumbbell or clover leaf is pointing.
For a spherical 's' orbital ('l'=0), there's only one way to orient it – it's round, it doesn't matter which way you turn it. So, ml can only be 0. Easy peasy.

But for those dumbbell-shaped 'p' orbitals ('l'=1), there are three possible orientations. We call them px, py, and pz, pointing along the x, y, and z axes respectively. So, ml can be -1, 0, or +1. It’s like having three identical dumbbells but placing them in different directions in your room.
For 'd' orbitals ('l'=2), there are five possible orientations, and for 'f' orbitals ('l'=3), there are seven. Each orbital shape can exist in multiple spatial arrangements.
Why is this important? Imagine a crowded concert hall. The seats are like orbitals. Even if two seats are the same shape, their location and orientation matter for where you can sit. Similarly, the spatial orientation of orbitals affects how electrons can interact between different atoms. It plays a role in things like magnetism, where the alignment of electrons in their orbitals can create a magnetic field.
The Spin Quantum Number: Spinning Like a Top
Finally, we have the spin quantum number (symbolized by 'ms'). This one is a bit like saying whether a dancer is spinning clockwise or counter-clockwise. Electrons, even though they're tiny, have an intrinsic property called "spin." It’s not literal spinning like a top, but it behaves similarly and creates a tiny magnetic field.
This spin can have one of two values: +1/2 or -1/2. It’s like the electron is a tiny little magnet that can point "up" or "down."
The crucial rule here, called the Pauli Exclusion Principle, states that no two electrons in an atom can have the same set of all four quantum numbers. This means that if two electrons are in the same orbital (same 'n', 'l', and 'ml'), they must have opposite spins. It's like having two people in the same tiny room; they have to agree on something to share the space peacefully!
Why does this matter? This simple rule is the reason why elements have the properties they do. It dictates how electrons fill up those orbitals, step by step, creating the unique electronic configuration for each element. This configuration is the key to understanding why iron rusts, why sodium reacts so vigorously with water, and why the colors of the rainbow are produced by different elements when heated. It’s the bedrock of all chemical behavior!
So, next time you hear about atoms and electrons, remember their quantum numbers. They’re not just abstract scientific terms; they’re the incredibly precise descriptions that govern the behavior of the universe at its most fundamental level. They’re the secret sauce that makes everything, from the air you breathe to the stars you see, do what it does. Pretty neat, right?