What Is The Oxidation State Of Cl In Hclo3
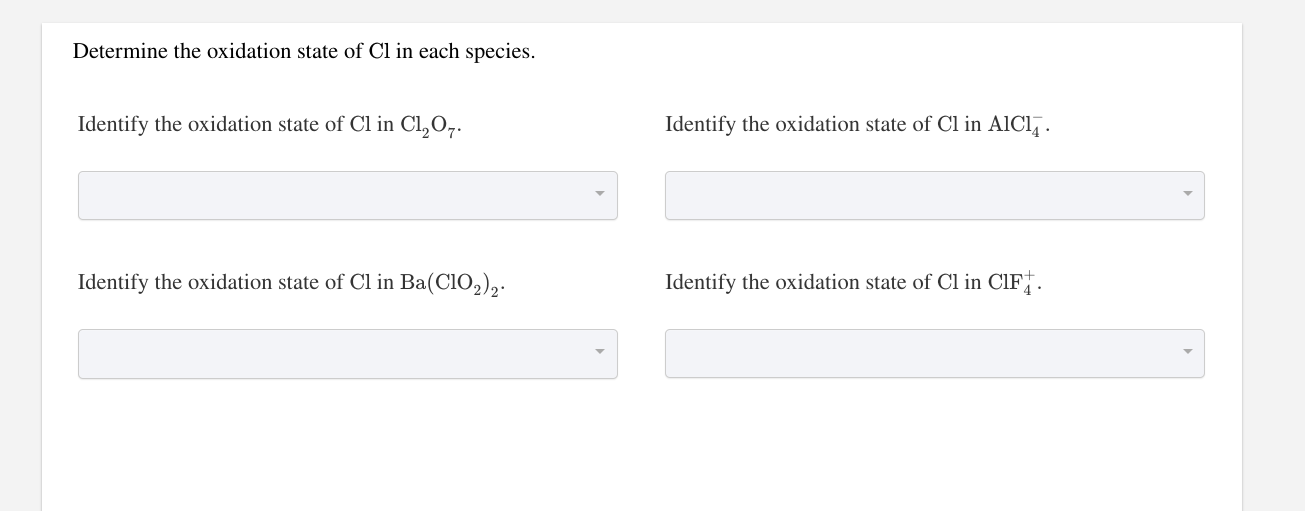
Ever wondered about the secret lives of molecules? It might sound like something out of a sci-fi movie, but understanding how atoms interact within compounds can be surprisingly fascinating! Think of it like detective work, where we're trying to figure out each atom's 'job' or 'charge' within a chemical family. Today, we're going to solve a little mystery surrounding chlorine, a common element that pops up in many everyday substances. Specifically, we're diving into the world of chloric acid, or HClO3, to uncover the hidden role of chlorine within this molecule.
Why is this important? Well, knowing the oxidation state of an atom like chlorine tells us a lot about its personality in a chemical reaction. It's like understanding if a character in a play is generally helpful, a bit mischievous, or takes on a neutral role. This knowledge is super useful for chemists, of course, as it helps them predict how molecules will behave, design new materials, and even develop life-saving medicines. But it's also a fun way for anyone to appreciate the intricate dance of atoms that makes up our world.
The purpose of determining an oxidation state is to assign a hypothetical charge to an atom in a compound. It’s a bookkeeping tool that helps us track electrons. Remember, atoms are constantly sharing or transferring electrons, and oxidation states help us keep tabs on who's losing electrons (getting oxidized) and who's gaining them (getting reduced). This concept is fundamental to understanding redox reactions, which are behind everything from batteries powering our phones to the way our bodies convert food into energy.
Must Read
The benefits of understanding oxidation states extend beyond just academic curiosity. For students learning chemistry, it’s a stepping stone to mastering more complex topics. For professionals in fields like environmental science, materials science, and medicine, it's an indispensable tool for problem-solving and innovation. Even for the curious home cook, it offers a glimpse into the chemical transformations happening when you bake a cake or ferment some kimchi!
So, let's get to the heart of our mystery: What is the oxidation state of Cl in HClO3? To figure this out, we rely on a set of well-established rules that chemists use as a guide. These rules are like the preamble to our chemical detective story, setting the stage for our investigation.
The Rules of the Game: Assigning Oxidation States
Before we solve for chlorine, let's quickly review some key rules we'll be using. These are pretty standard, and you'll see them applied again and again in chemistry:
- The oxidation state of an element in its elemental form (like O2, H2, or Cl2) is always 0.
- The oxidation state of oxygen in most compounds is -2. There are exceptions, like in peroxides, but for chloric acid, this rule holds true.
- The oxidation state of hydrogen is usually +1 when bonded to nonmetals.
- The sum of the oxidation states of all atoms in a neutral compound must equal 0.
These rules are our trusty magnifying glass and fingerprint kit as we examine HClO3.
Cracking the Case of HClO3
Let's break down chloric acid, HClO3, atom by atom, and apply our rules:
- We have one hydrogen (H) atom. According to our rules, hydrogen bonded to a nonmetal (and oxygen is definitely a nonmetal) has an oxidation state of +1.
- We have three oxygen (O) atoms. Oxygen typically has an oxidation state of -2. So, for our three oxygen atoms, the total contribution is 3 * (-2) = -6.
- We have one chlorine (Cl) atom. This is the one we're trying to find! Let's call its oxidation state 'x'.
Now, remember rule #4: the sum of all oxidation states in a neutral compound must be 0. Our compound, HClO3, is a neutral molecule.
So, we can set up an equation:
(Oxidation state of H) + (Oxidation state of Cl) + (Total oxidation state of O) = 0
Plugging in the values we know:
(+1) + (x) + (-6) = 0
Now, it's just a simple algebraic puzzle to solve for 'x':
x - 5 = 0
Adding 5 to both sides:
x = +5
And there you have it! The oxidation state of chlorine (Cl) in chloric acid (HClO3) is +5!
What Does +5 Mean for Chlorine?
An oxidation state of +5 for chlorine is quite high! This tells us that in chloric acid, chlorine has lost five of its electrons. This makes chlorine quite electron-deficient and a strong oxidizing agent, meaning it's very good at 'stealing' electrons from other substances. This property is what makes chloric acid and its salts, known as chlorates, useful in applications like matches, fireworks, and even some disinfectants.
So, the next time you encounter HClO3, you'll know that the chlorine atom within it is playing a rather significant role, having given up a considerable number of its electrons. It’s a small detail, but it reveals a lot about the molecule's character and its potential for chemical interactions. The world of chemistry is full of these little mysteries, and by learning to solve them, we unlock a deeper appreciation for the amazing substances all around us.





