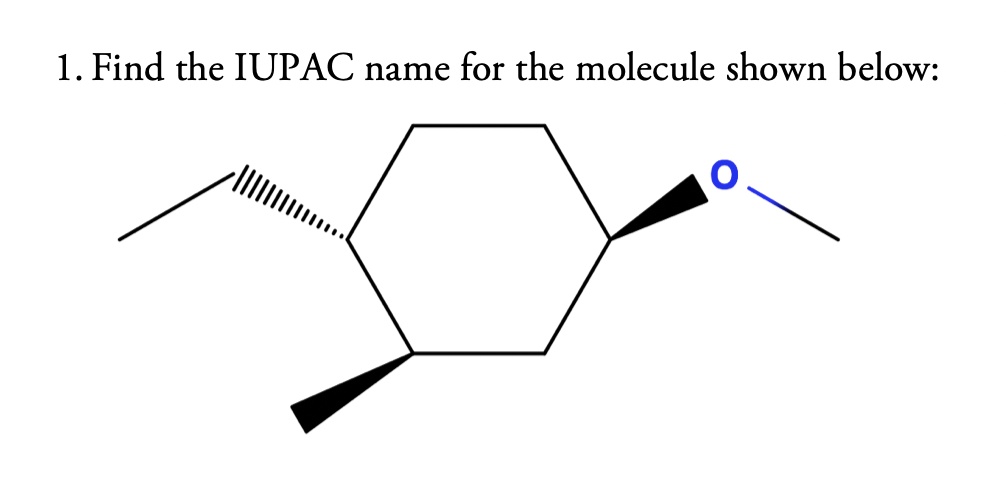
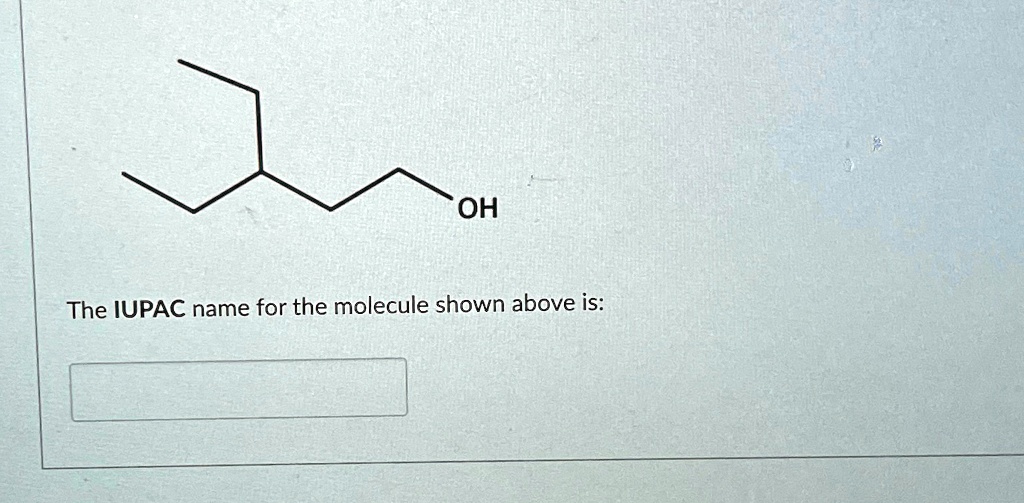
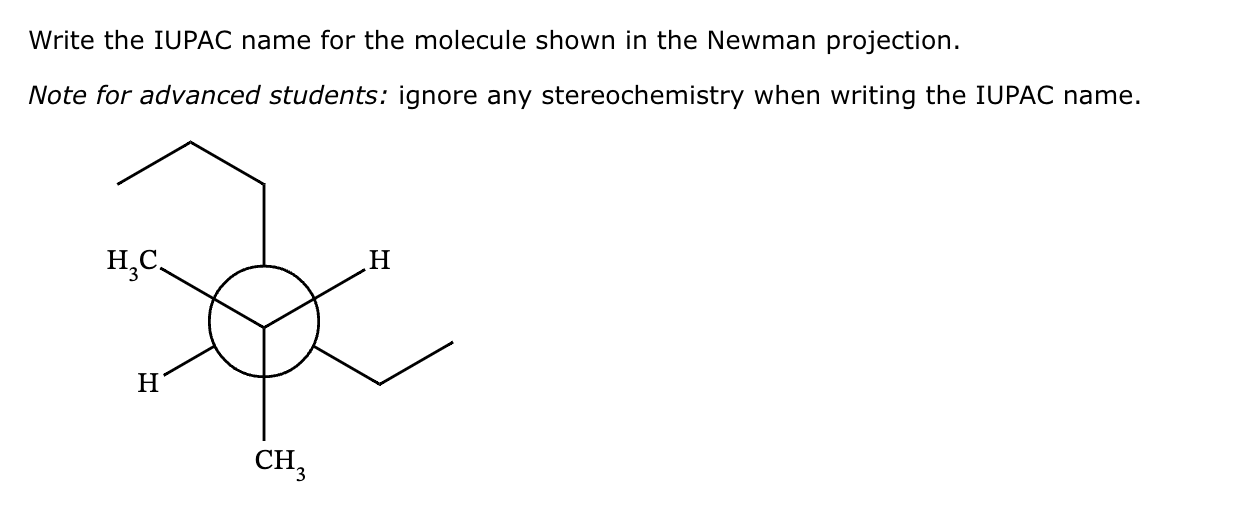
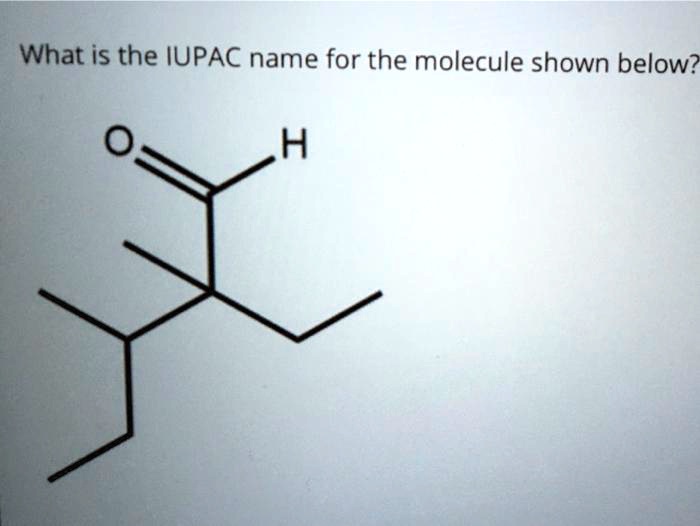
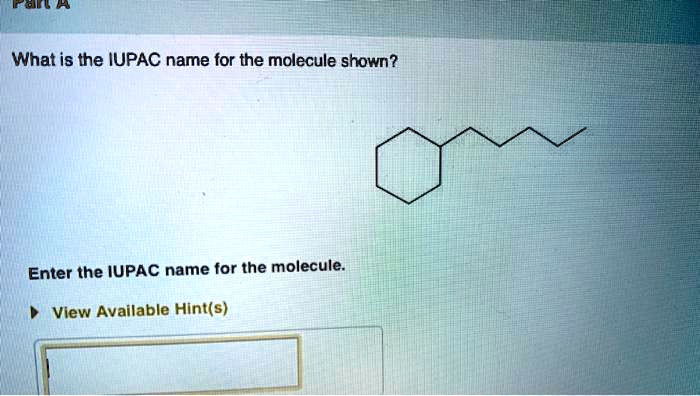
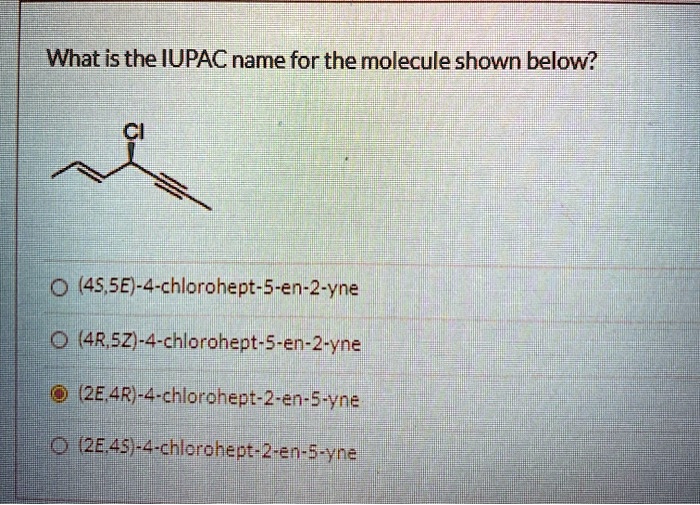
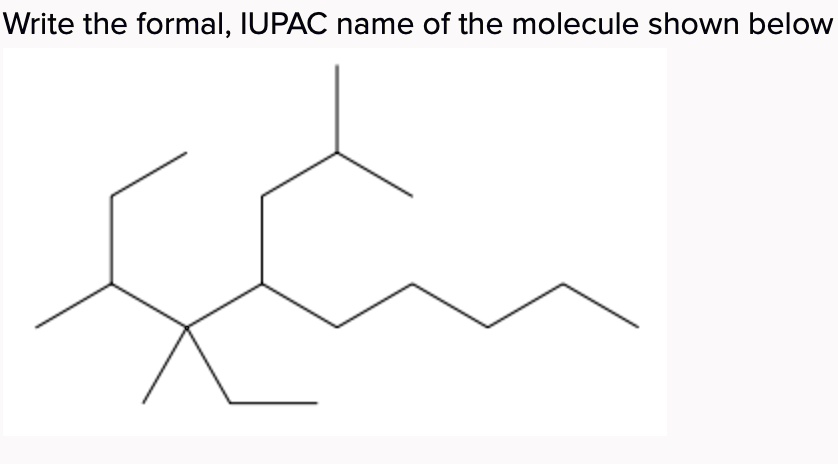
What Is The Iupac Name Of The Molecule Shown
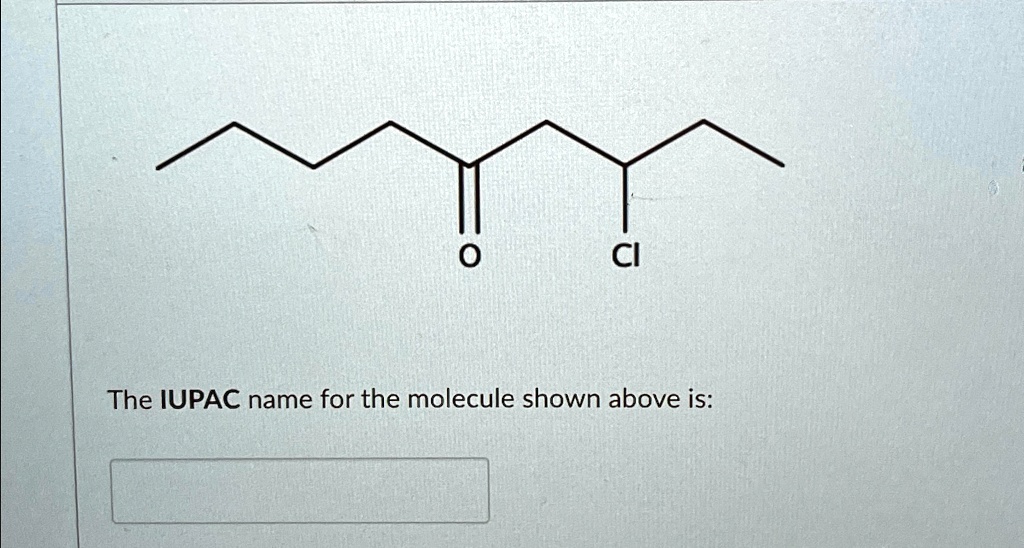
Ever looked at a complicated chemical formula and wondered what on earth it's called? It might seem intimidating, but understanding the secret language of molecules can actually be quite fun and surprisingly useful! Think of it like learning a secret code that unlocks the identity of all the stuff around us, from the water we drink to the air we breathe.
This secret code is called the International Union of Pure and Applied Chemistry (IUPAC) naming system. It's a set of rules that chemists all over the world use to give every single molecule a unique and descriptive name. Why is this so cool? Well, imagine trying to describe your friend to someone without their name – it would be a mess! The IUPAC system ensures everyone is talking about the exact same molecule. This is super helpful for scientists, of course, but it can be a fun brain-teaser for families too, and a fantastic pursuit for any budding hobbyist chemist!
For absolute beginners, learning a bit about IUPAC names can demystify science. It’s like learning the alphabet before you can read a book. You might not become a chemist overnight, but you'll start to understand the building blocks of the world. For families, it can turn everyday observations into learning moments. Pointing to a bottle of rubbing alcohol and knowing it’s called isopropanol (or more formally, propan-2-ol) can spark curiosity. For hobbyists, it’s an essential skill. If you're experimenting with making soaps, lotions, or even just trying to understand ingredient lists, knowing these names is key to safe and successful creation.
Must Read
Let's take a super simple example. You've probably heard of methane. That's the main component of natural gas. Its IUPAC name is simply methane. Now, if we add another carbon atom and some hydrogens, we get ethane. See the pattern? Meth- usually means one carbon, and eth- means two. It's like building with molecular LEGOs! You can also have variations like ethanol, which is the alcohol in your hand sanitizer and in alcoholic drinks, or ethanoic acid, which is the main ingredient in vinegar (acetic acid).

Getting started is easier than you think! You don't need to memorize a giant dictionary of names. Start with the basics:
- Learn the prefixes for the number of carbon atoms: meth- (1), eth- (2), prop- (3), but- (4), and so on.
- Familiarize yourself with common endings: -ane for simple, single-bonded molecules, -ene for double bonds, and -yne for triple bonds.
- Look up the names of common compounds you encounter every day. Water is technically dihydrogen monoxide, but that’s a name you’re unlikely to use! However, understanding the IUPAC naming for things like sugars or salts can be very rewarding.
So, the next time you see a complex chemical name, don't shy away from it! Embrace the challenge, and remember that behind every name is a story about a unique molecule. Learning the IUPAC system is a journey, and even a small step can open up a whole new world of understanding and appreciation for the chemistry that surrounds us. It’s a wonderfully practical and surprisingly enjoyable way to connect with the science of everyday life.