Heating Curve Of Water Lab Worksheet Answers
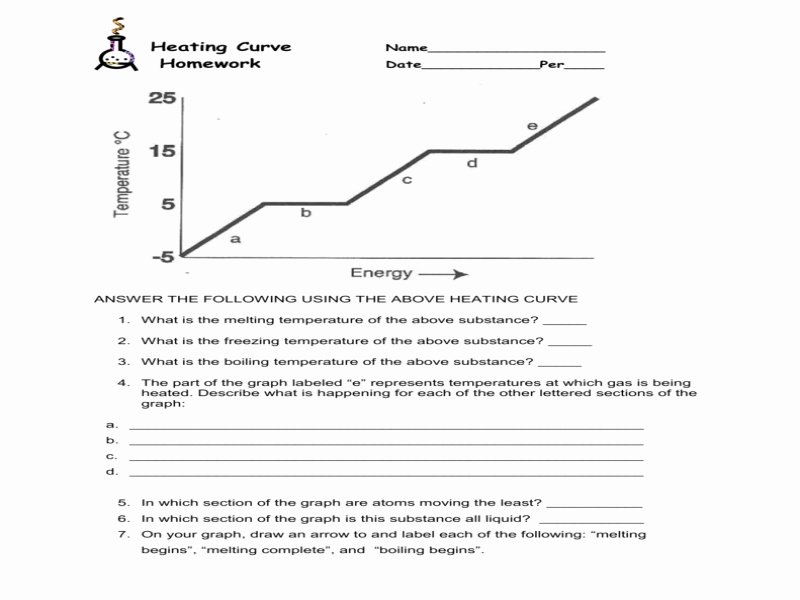
Hey there, fellow temperature tinkerers and kitchen wizards! Ever stared at a pot of water on the stove, willing it to boil faster so you can finally make that much-needed cup of tea or get those pasta noodles cooking? We’ve all been there, right? That’s pretty much the everyday adventure we’re diving into today, but with a fancy science-y twist: the Heating Curve of Water Lab Worksheet Answers.
Think of it like this: your water is on a journey. It starts out all chill, like a sleepy cat on a cool morning. Then, you crank up the heat, and it’s like waking up that cat with a gentle nudge. It starts to get a little antsy, then a bit more wiggly, and finally, whoosh! It’s doing the salsa in the pot. This lab is basically charting that entire dance, from its lazy start to its bubbly finale.
And those "worksheet answers"? Well, they're like the cheat codes to understanding your water's dance. Instead of just guessing why your water is doing what it's doing, these answers help you really get it. It’s like finally understanding why your toast pops up perfectly sometimes and is borderline charcoal other times. There’s a science to it, and today, we’re unlocking the secrets to water’s thermal tango.
Must Read
The Big Chill: Water in its Natural State
Before we even think about turning on the stove, let's talk about water when it's just… being water. We’re talking about ice. Remember building snowmen or having that ice cream headache from taking too big a bite? That’s water in its solid, frozen state. It’s all locked up, molecules holding hands tightly, not wanting to move much. Think of it like a bunch of toddlers holding onto each other at a playground – pretty stationary.
When you’re doing this lab, the starting point is usually ice. You’re measuring its temperature, and it’s probably hovering around 0 degrees Celsius (or 32 Fahrenheit, for those who prefer Fahrenheit like a comfortable old sweater). It’s cold, it’s solid, and it’s not going anywhere fast. This is the baseline, the initial setting on your water’s mood ring.
Imagine you’re trying to get a stubborn toddler to move. You can’t just shove them. You have to coax them, maybe offer a cookie. Similarly, you need to add a lot of energy to get those water molecules to even think about loosening up. This is where the heating curve starts its slow crawl upwards.
The Shivering Phase: Warming Up the Ice
So, you’ve got your ice, and you’ve started adding some heat. What happens first? The ice starts to warm up. It’s still ice, but it’s getting less… icy. Think of it like putting on a light jacket when it’s a bit chilly outside, but not yet freezing. You’re not changing your fundamental state (still wearing clothes!), but you’re definitely more comfortable.
In our lab, this is the first part of the heating curve. The temperature of the ice goes up, up, up. You’re adding energy, and those tightly bound molecules are starting to vibrate a little faster. They’re still stuck in their ice fort, but they’re getting restless. They’re thinking, “Man, it’s getting a little warm in here. I wonder what’s going on outside?”

This phase is crucial because it shows that energy is being absorbed, and it's directly contributing to raising the temperature. It’s like the anticipation before a big event. You’re getting ready, building up energy, but the main show hasn’t started yet. And the answers on your worksheet will show this gradual temperature increase, a steady upward trend on your graph.
The Great Melting Caper: Ice Becomes Water
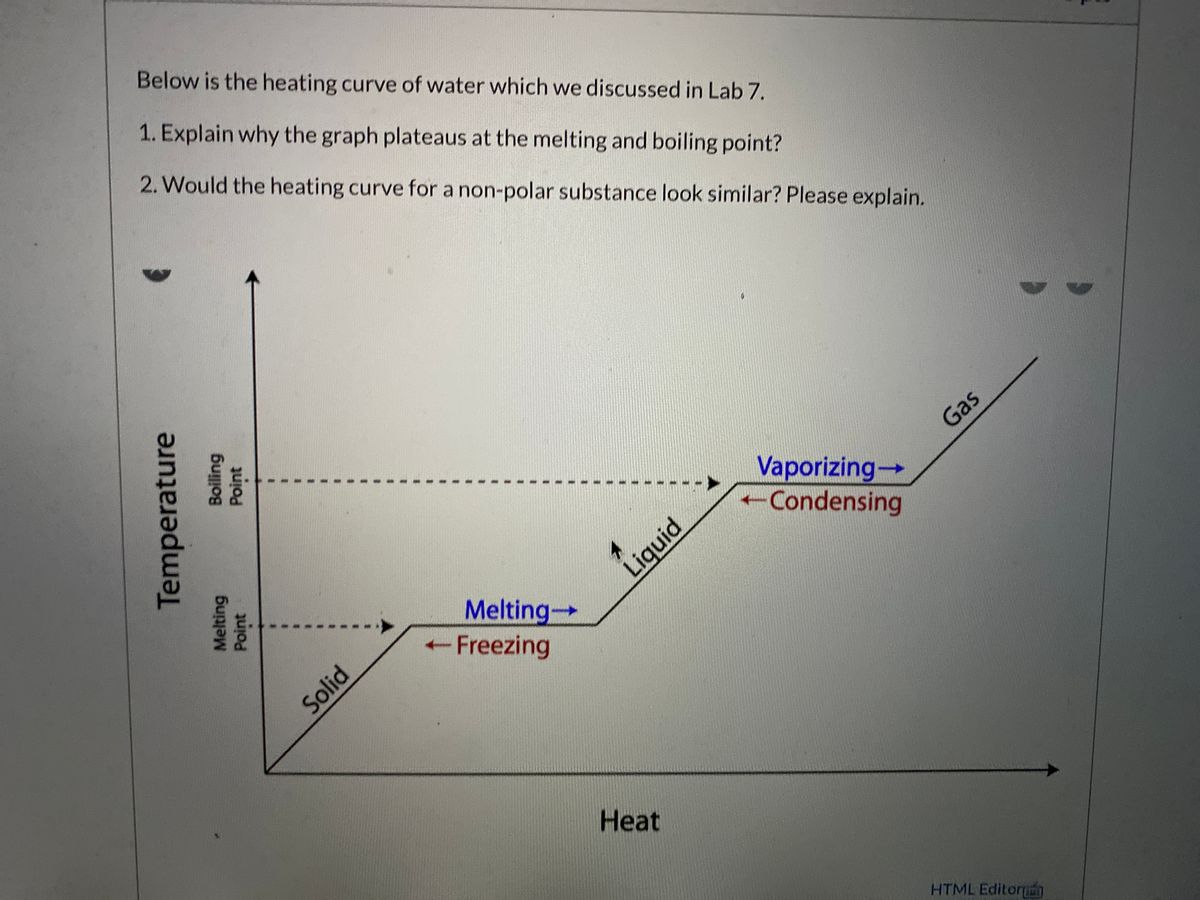
Now, here comes the magical part, the moment of truth: the melting point! This is where the ice finally says, “Okay, I’ve had enough of this shivering. I’m ready to let go!” And just like that, it transforms into liquid water. It’s a phase change, a complete metamorphosis.
This is a super important part of the heating curve because, here’s the kicker: the temperature doesn’t change during melting. Yep, you read that right. You’re still pumping heat into the ice, but instead of getting hotter, all that energy is being used to break those bonds, to get those molecules to stop holding hands and start sliding past each other.
Think of it like a party where everyone is doing the macarena. They’re all energized, moving around, but their individual positions aren’t necessarily changing drastically. Or, even better, think about a really awkward family reunion. Everyone’s there, there’s a lot of energy, but nobody’s really going anywhere, just kind of mingling in the same space. That’s your melting ice!
The temperature stays stubbornly at 0 degrees Celsius (or 32 Fahrenheit) until all the ice is gone. This flat line on your graph is a big clue. It's telling you that the energy is busy doing a different job: breaking down the solid structure. The worksheet answers will reflect this plateau, this period of no temperature change despite continuous heating. It’s like hitting a pause button on the thermometer while the real work happens behind the scenes.

The Wiggly Phase: Warming Up the Liquid Water
Once all the ice has done its disappearing act and you’ve got good old liquid water, the temperature starts to climb again. This is where water gets its wiggle on. It’s like the party has really kicked off, and everyone’s doing their own thing. The molecules are now free to move around, slide past each other, and generally be more fluid.
This is the part of the heating curve where the temperature rises steadily. You’re adding heat, and the molecules are getting more kinetic energy, moving faster and bumping into each other more often. Imagine a bunch of kids who have just been let loose in a playground. They’re running, jumping, and generally having a blast. That’s your liquid water.
Your worksheet answers will show this upward slope again, but it might be a bit steeper than when you were heating the ice. This is because liquid water is generally more efficient at absorbing heat and converting it into temperature increase compared to breaking the solid bonds of ice. It’s like going from a slow shuffle to a lively jig!
The Big Boil-Off: Water Turns to Steam
And then, the moment we’ve all been waiting for (especially when you’re hungry!): the boiling point! This is when the liquid water decides it’s had enough of being confined to the pot and wants to float around as a gas. It’s another phase change, and this one is usually a bit more dramatic, with all those satisfying bubbles.
Just like melting, boiling is another one of those magical moments where the temperature holds steady. You’re still adding heat, but all that energy is being used to turn the liquid into gas, to overcome the forces holding the water molecules together in their liquid dance.

Think of it as the ultimate freedom for water molecules. They’re breaking free from the confines of the pot and are ready to explore the vastness of the kitchen air (or beyond!). This flat line on your graph, just like the melting plateau, signifies a phase change. The energy is working overtime to make this transformation happen.
The boiling point of water is a pretty constant value, 100 degrees Celsius (or 212 Fahrenheit) at standard atmospheric pressure. This is one of those facts that’s super handy to know. If your water isn’t boiling at that temperature, something might be up – maybe the atmospheric pressure is different, or maybe your stove isn’t as hot as you think!
The Hazy Future: Warming Up the Steam
Once all the liquid water has bravely ventured into the gaseous state as steam, the temperature can start to climb again. This is the final phase of our heating curve. The steam molecules are now free to zoom around at incredibly high speeds, bouncing off everything.
Imagine a bunch of hyperactive toddlers who have just discovered a trampoline. They’re going everywhere, at warp speed! That’s your steam. You’re still adding heat, and this energy is making those steam molecules move even faster, increasing their temperature.
This part of the graph will show another upward slope. The steam can get really hot, much hotter than the boiling point of water. This is why you need to be super careful around steam – it’s not just hot water, it’s really, really energetic hot water in a gaseous form, and it can cause serious burns. So, while it’s fun to think about the science, always remember safety first!
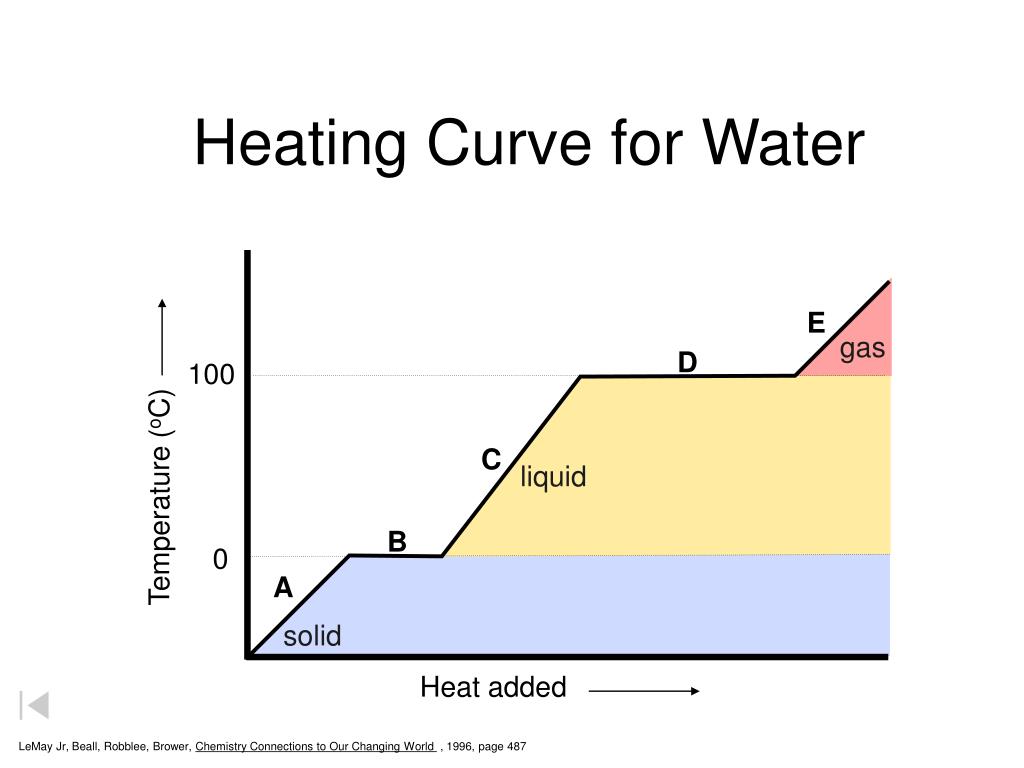
Putting It All Together: The Grand Heating Curve Spectacle
So, when you look at your heating curve graph from the lab, you’re basically seeing a visual representation of water’s journey. It starts low and steady, goes through a flat line where it’s changing form, then climbs again, hits another flat line for a different form change, and finally climbs again as a gas.
The worksheet answers are your guide to interpreting these different segments. They’ll help you identify the melting point, the boiling point, and the temperature changes within each state. They confirm what you observed and learned through the experiment.
Think of it like baking. You have different stages: mixing the batter, baking, and decorating. Each stage uses energy and results in a different outcome. The heating curve is just like that, but for water and its temperature. It’s a fundamental concept in chemistry and physics, but it’s also something you see in action every single day, from your morning coffee to a steaming hot shower.
Why Does This Even Matter, Besides Passing My Science Class?
Honestly? Because understanding this helps you understand so much more about the world around you. It’s why ice melts in your drink, why steam rises from a hot pavement after rain, and why we have things like clouds and rain in the first place. It’s the science behind why your soup takes so long to cool down, or why a kettle whistles.
The heating curve of water is a beautiful illustration of how energy affects matter. It shows us that changing temperature isn't always just about getting hotter or colder; sometimes, it’s about a fundamental transformation. These lab answers aren't just numbers on a page; they’re clues to the amazing molecular dance that water performs every single day.
So next time you’re waiting for water to boil, take a moment to appreciate the science happening in that pot. You’re witnessing a heating curve in action, and with those worksheet answers, you’ve got the power to understand it all. Pretty cool, right? Now go forth and impress your friends with your newfound water-warming wisdom!