What Is The Iupac Name For Isobutyl Alcohol

Ever wonder what all those weird, long names are that chemists use? It might sound intimidating, but understanding the IUPAC name for something like isobutyl alcohol is actually kind of fun and super useful! Think of it like having a secret code that unlocks the identity of chemicals. This isn't just for scientists in lab coats; knowing these names can make you feel a little more in-the-know, whether you're a curious beginner, a family exploring the world around you, or a hobbyist dabbling in home chemistry or craft projects.
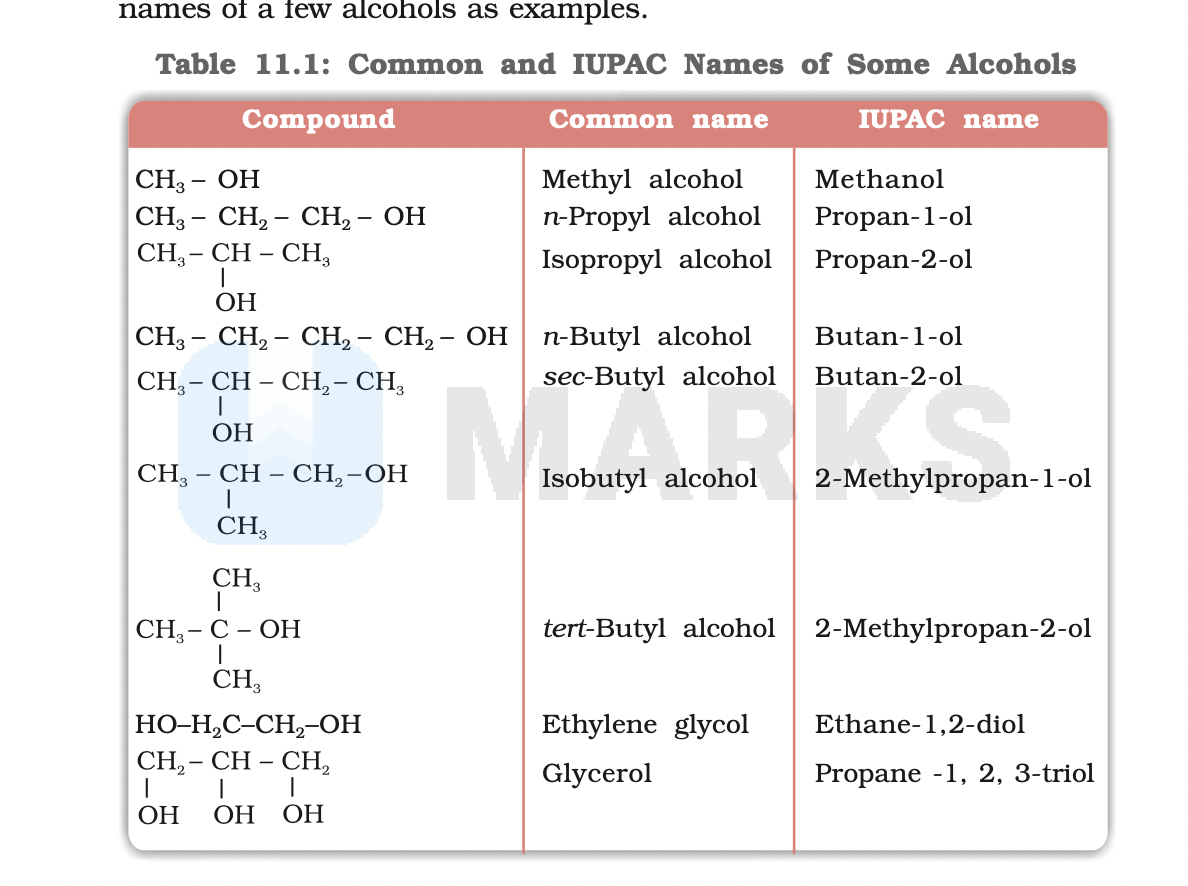
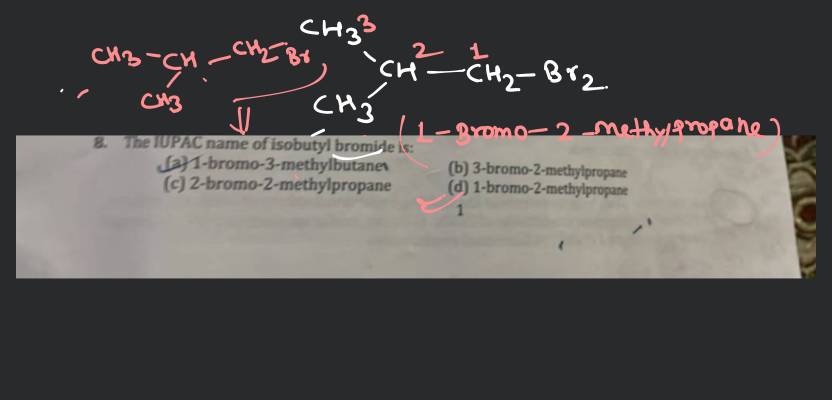
So, what exactly is the IUPAC name for isobutyl alcohol? Drumroll please... it's 2-methylpropan-1-ol. Pretty neat, right? The IUPAC, which stands for the International Union of Pure and Applied Chemistry, has a standardized system for naming chemical compounds. This system ensures that no matter where you are in the world, or who you're talking to, there's only one correct name for a specific chemical. This is incredibly helpful to avoid confusion and ensure clear communication, especially when discussing things like ingredients in products or the components of substances.
For beginners, learning this name is a fantastic first step into the world of organic chemistry. It demystifies chemical nomenclature and shows you that it's not all rocket science. Families can use this as a fun learning opportunity. Imagine pointing to a label on a product (if it lists ingredients) and saying, "Hey, that's a fancy way of saying it contains isobutyl alcohol!" It sparks curiosity and encourages learning. For hobbyists, especially those into making soaps, candles, or even certain DIY cleaning solutions, knowing the correct chemical names can be important for understanding what you're working with and ensuring safety and efficacy.
Must Read
Let's break down 2-methylpropan-1-ol a little. The 'propan' part tells us it's a three-carbon chain. The '-1-ol' indicates an alcohol group (an -OH) attached to the first carbon. The '2-methyl' tells us there's a methyl group (a -CH₃) attached to the second carbon in that chain. This is the precise description of the molecule's structure, making it unique and identifiable. Think of it like giving a house an address – it tells you exactly where it is.
To get started with this kind of naming, the best tip is to start small. Focus on common chemicals and their IUPAC names. Look up the IUPAC name for water (H₂O – dihydrogen monoxide, though that’s rarely used outside of humor!), or sugar (sucrose). Websites dedicated to chemistry or even some educational apps can be great resources. Don't be afraid to jot down notes or draw out the structures as you learn. It's all about building familiarity.
Understanding the IUPAC name for isobutyl alcohol, or 2-methylpropan-1-ol, is more than just memorizing a string of words. It’s about appreciating the elegant logic behind chemical naming and gaining a little more confidence in understanding the world of chemistry. It's a small step that can lead to a greater appreciation for the science that shapes our everyday lives, and that's pretty darn cool!