What Is The Importance Of Valence Electrons
Okay, confession time. When I first heard about valence electrons, my brain did that little buffering thing. It sounded like something you'd find in a super-fancy science fiction movie, not something that actually makes the world go 'round. But guess what? Turns out, these little guys are the unsung heroes of, well, everything. And in my humble, possibly unpopular opinion, they deserve way more fanfare.
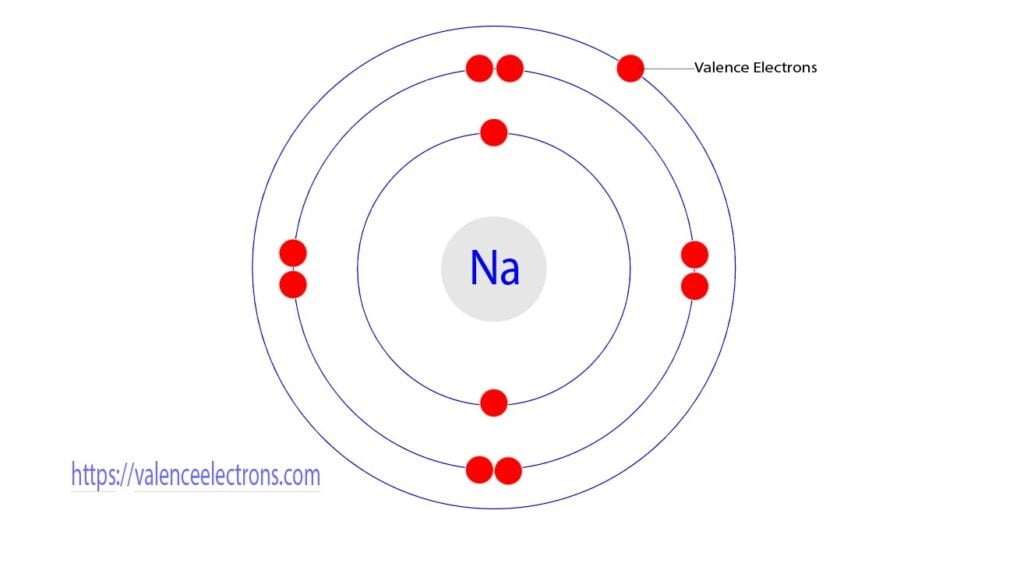
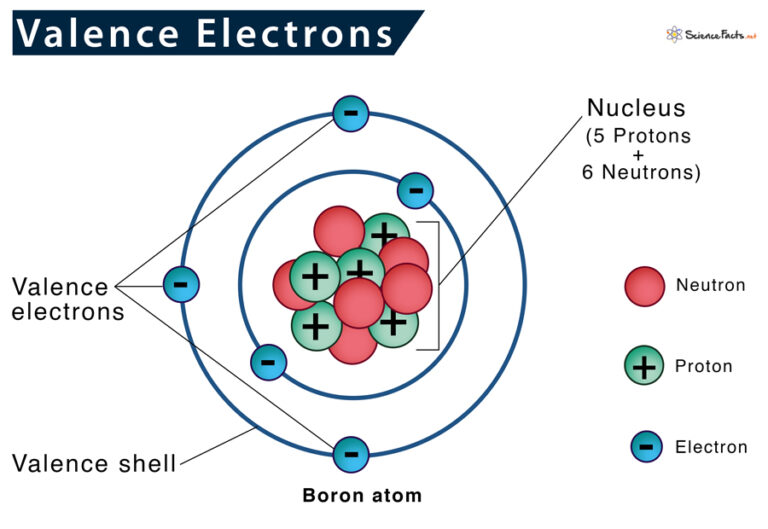
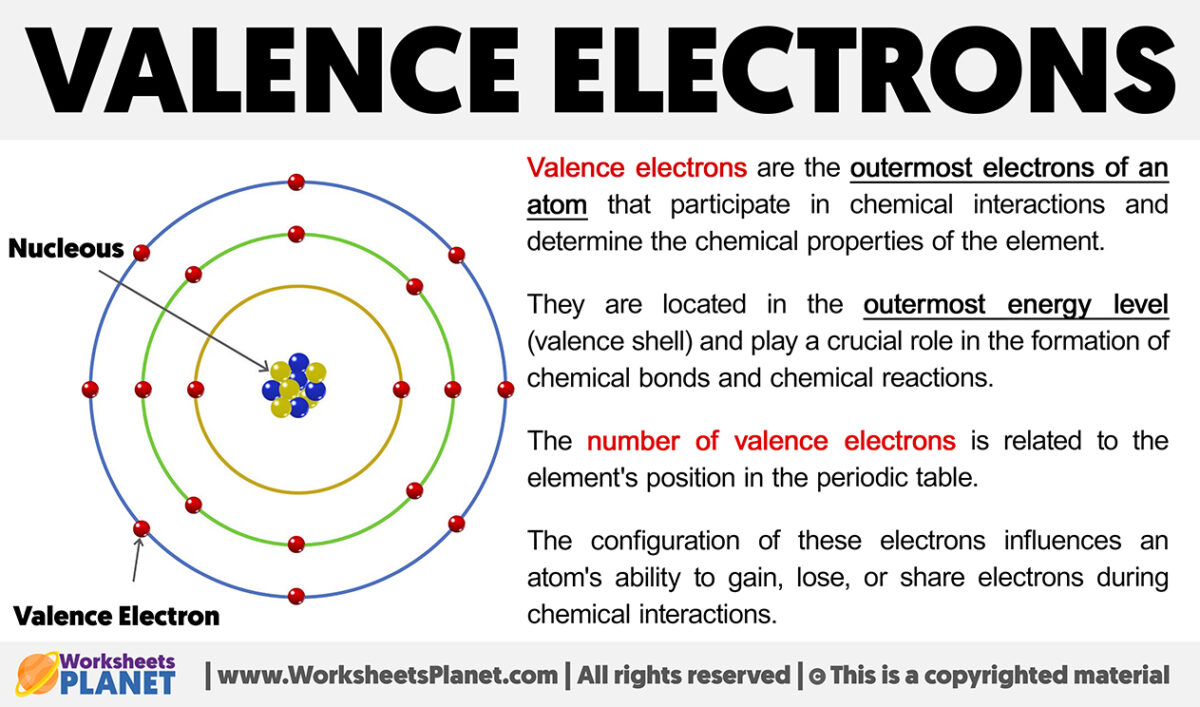
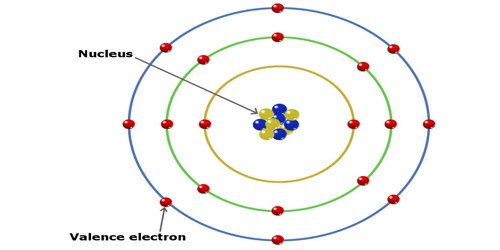
Think of atoms like tiny little apartment buildings. They have a core, like the building's foundation, where the important stuff like protons and neutrons hang out. Then they have these energy levels, kind of like floors. And on the very outermost floor, the one closest to the outside world, are the valence electrons. They're the tenants on the penthouse suite, the ones with the best view, and frankly, the ones who get all the action.
Why are they so important? Because they're the social butterflies of the atomic world. They're the ones who get to interact with other atoms. They're the ones who decide if an atom is going to be a loner, a best friend, or a clingy acquaintance. It's all about those valence electrons. They're the dating app profiles of the atom universe.
Must Read
Imagine you're at a party. You're not going to go around hugging the security guard or shaking hands with the potted plants, right? You're going to mingle with other guests. Valence electrons are the party guests of the atom. They're the ones who decide who to dance with, who to chat with, and who to form a permanent bond with.
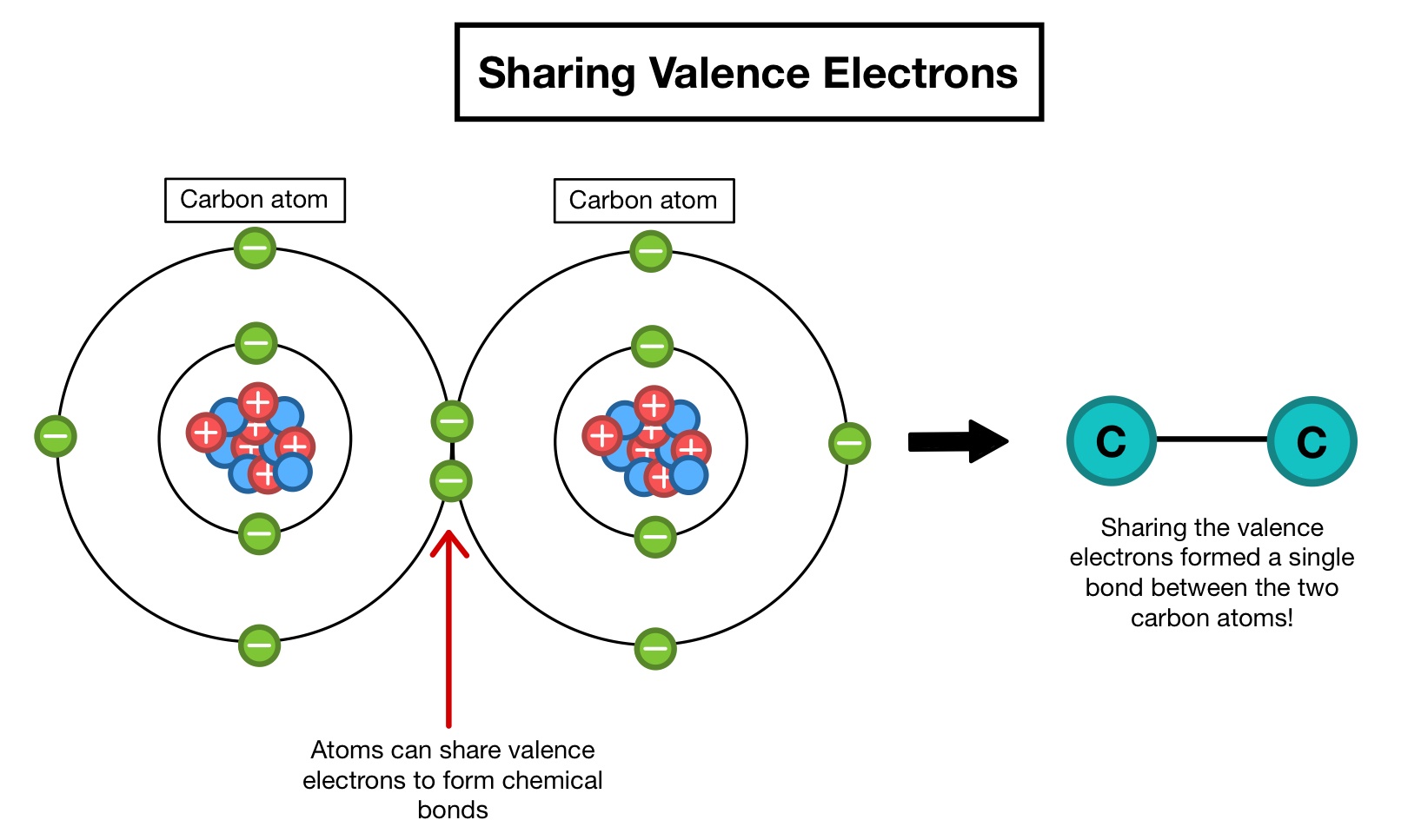
This is where all the cool stuff happens. When atoms decide to share their valence electrons, you get covalent bonds. Think of it like a potluck dinner. Everyone brings a dish, and they all share. This is how molecules are born. Water? That's hydrogen and oxygen playing nice with their valence electrons. Sugar? Same deal, just with more players.
Then there are atoms that are a bit more generous, or perhaps a bit desperate for company. They'll straight-up give away a valence electron. This creates ions, which are like atoms with an extra bit of energy or a missing piece, looking for something to complete them. It's like someone at the party who keeps offering you their chips, even when you're full.
And the ones who take those electrons? They're equally important! They're the ones who complete the circuit, who make the connection. This is how ionic bonds form, like when salt (sodium chloride) comes to be. It's a whole drama of giving and taking, all thanks to those handy-dandy valence electrons.

Seriously, without these guys, we wouldn't have anything solid. No tables, no chairs, no your favorite comfy couch. Even your phone, which you're probably using to read this, relies on the intricate dance of valence electrons to function. Think about that for a second. Your ability to scroll endlessly is powered by tiny electrons making friendship bracelets with other electrons.
It's kind of mind-blowing, isn't it? These little bits of energy, clinging to the edge of existence, are responsible for the very fabric of reality as we know it. They're the reason why a tiny seed can grow into a giant tree. They're the reason why that piece of chocolate melts perfectly in your mouth. It's all about their desire to be stable, to have a full outer shell. It's like they're all striving for that ultimate atomic chill.
So, next time you look at anything, anything at all, take a moment to appreciate the valence electrons. They’re the ultimate matchmakers. They’re the secret ingredient in the universe's recipe book. They’re the unsung heroes, the quiet achievers, the reason why atoms don't just float around like a bunch of awkward strangers at a cosmic speed dating event. They're the reason for connections, for bonds, for stuff. And honestly, I think that's pretty darn cool. Maybe it’s an unpopular opinion, but I’m going to go ahead and say it: Valence electrons are the real MVPs.