What Is A Measure Of Average Kinetic Energy

Ever wondered what makes things wiggle? Like, really wiggle?
We’re talking about the tiny, invisible dance of atoms and molecules. It’s the secret life of stuff! And there’s a super cool way to measure how much they’re busting a move. It’s called average kinetic energy.
Get Ready to Vibrate!
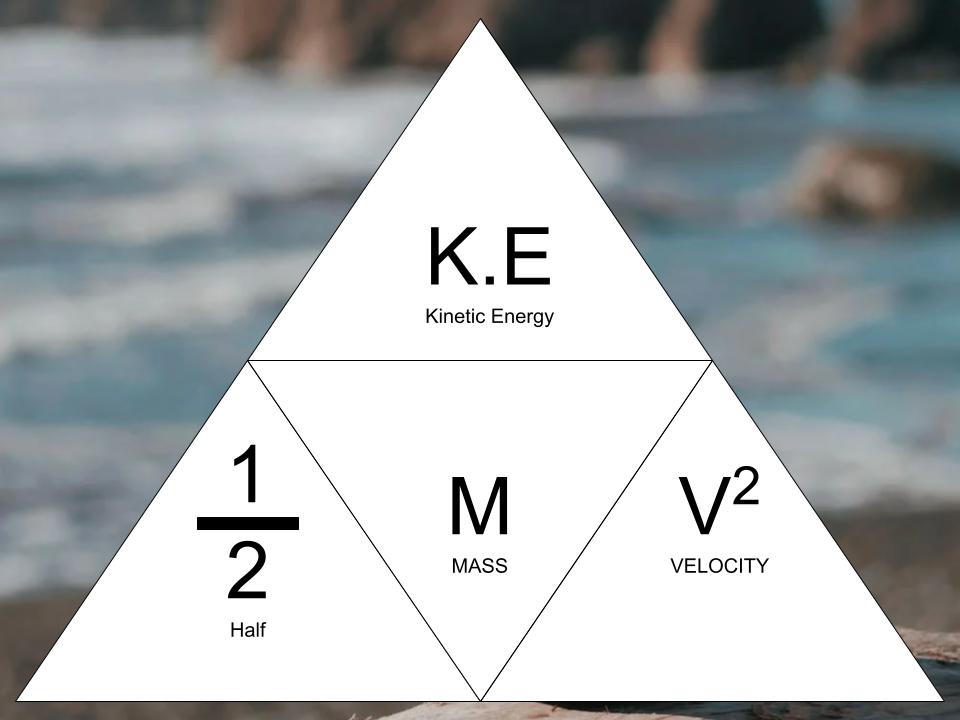
So, what exactly is this "average kinetic energy" thing? Imagine a room full of hyperactive toddlers. Some are zooming, some are bouncing, some are just jiggling in place. Kinetic energy is basically the energy of motion. The faster something moves, the more kinetic energy it has. Makes sense, right?
Must Read
But here’s the fun part. When we talk about average kinetic energy, we’re not looking at just one super-speedy molecule. We’re looking at the whole bunch. It's like taking all those toddlers and figuring out their average zoominess level. Not too serious, just a fun way to understand the overall vibe of a substance.
It's All About the Jiggle
Think about ice. It's pretty chill, right? The molecules are mostly just vibrating. They're not going anywhere fast. Now, imagine that ice melting into water. Suddenly, the molecules have a bit more freedom to slide and swirl. They're definitely jiggling more!
And then, BAM! Steam. Those water molecules are going absolutely bonkers. They're zipping around at lightning speed. That's a huge jump in their average kinetic energy. It’s like the toddlers went from a gentle sway to a full-on disco party.
Temperature: The Ultimate Vibe Check
Here’s where it gets really interesting. Guess what’s directly related to this average kinetic energy? Yep, you guessed it: temperature!
Hot things have molecules that are zooming and zapping with lots of energy. Cold things have molecules that are… well, a bit more sluggish. They’re still moving, mind you. Nothing ever truly stops moving at the molecular level (unless you get to absolute zero, which is a whole other wild story for another day!).
So, when you feel a hot cup of coffee, you’re not just feeling heat. You’re feeling the intense wiggling of all those coffee molecules. It’s a molecular mosh pit!

The Quirky Truth About "Hot"
It’s kind of funny to think that "hot" is just a measure of how much tiny things are freaking out. That glowing ember? It’s just a bunch of atoms doing a frantic jig. The sun? A colossal ball of super-heated, wildly energetic particles. It's all about the motion, baby!
And "cold" isn't about stillness; it's about a slightly less frantic dance. Even in the deepest, darkest reaches of space, there’s still some residual molecular jiggle. It’s a universe that never truly sleeps. Kind of inspiring, if you think about it!
Why Should You Care About Molecular Jiggles?
Okay, okay, you might be thinking, "This is neat, but why does it matter to me?" Well, it explains SO much!
Why does a balloon deflate when it gets cold? Because the air molecules inside slow down and don't push out as much. Why does cooking make food softer? Because the heat (remember, the jiggling!) breaks down the molecules in the food.
It’s the secret sauce behind so many everyday phenomena. It’s the hidden engine that powers everything from a perfectly baked cake to the wind in your hair.
It's a Universal Language
And here’s a really cool tidbit: the average kinetic energy of a substance is the same regardless of what that substance is, as long as it’s at the same temperature. So, a mole of hydrogen gas at 20°C has the same average kinetic energy as a mole of gold at 20°C.
Mind. Blown. It’s like a universal handshake for molecules. They might be different types of dancers, but if they’re at the same party temperature, they’re all doing that average dance with the same energy.
The Fun Doesn't Stop There!
We're not even scratching the surface here. This concept of average kinetic energy is a cornerstone of thermodynamics, which is basically the science of heat and energy. It helps us understand how engines work, how refrigerators keep things cool, and even how stars shine.
It’s a big deal, but we can still think about it in a fun, relatable way. It's all about the microscopic ballet happening all around us, all the time.
Think of it as a Tiny Dance-Off
So next time you feel the warmth of the sun or the chill of a winter breeze, remember the microscopic dance party. It’s the ultimate testament to the fact that everything, absolutely everything, is in motion.
The average kinetic energy of molecules. It’s not just a science term; it’s the secret rhythm of the universe. And isn't that just a wonderfully peculiar thought?