What Forces Hold Ionic Solids Together Apex

Ever wondered why those sparkly salt crystals or chalky antacids stay so stubbornly solid? It’s all thanks to some seriously cool forces, and understanding them is surprisingly fun and useful! Think of it like unlocking a little secret about the everyday stuff around you. This isn't just for science buffs; it’s a neat peek into how the world works, and knowing a bit about it can make you feel a little more connected to the "why" behind things.
So, what’s the big deal about what holds ionic solids together? For absolute beginners, it’s a gentle introduction to the tiny building blocks of matter and the invisible forces that govern them. You’ll learn about atoms making friends (or, more accurately, exchanging best buddies!) and sticking together. For families exploring science at home, it's a fantastic opportunity for hands-on learning. Imagine using salt and sugar crystals to demonstrate these concepts – simple, visual, and engaging! Hobbyists, particularly those into things like rock collecting, geology, or even culinary arts (think salt structures!), will gain a deeper appreciation for the properties and formation of the materials they work with.
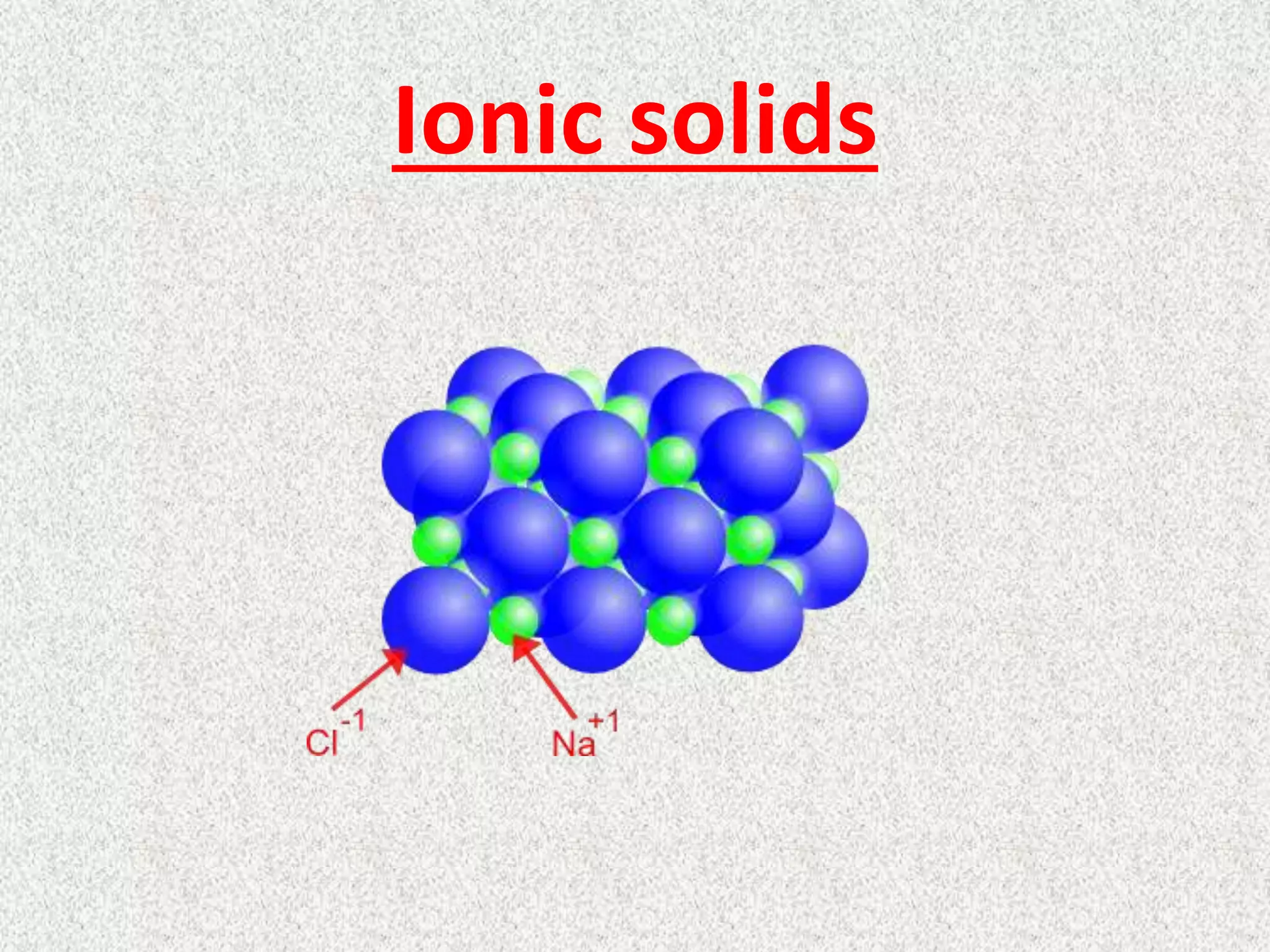
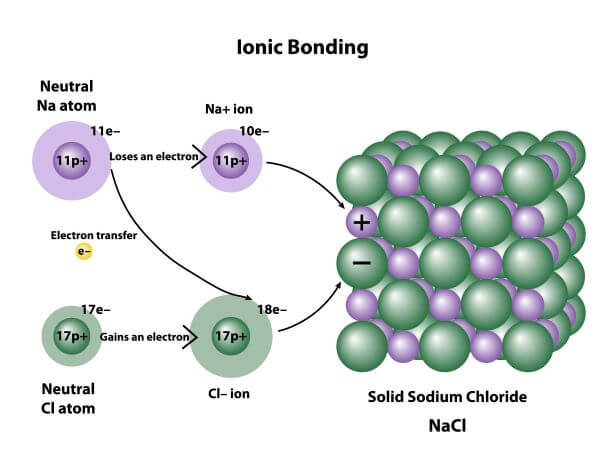
The main force at play in ionic solids is the electrostatic attraction. Imagine tiny magnets, but instead of magnetism, it's the opposite electrical charges that are doing the pulling. You've got positively charged ions and negatively charged ions, and they are just drawn to each other like super-strong glue! Think of table salt (sodium chloride, NaCl). Sodium atoms happily give up an electron to become positive (Na+), and chlorine atoms eagerly grab that electron to become negative (Cl-). These oppositely charged ions then arrange themselves in a repeating, crystal lattice structure, like perfectly stacked building blocks, to maximize these attractions and minimize repulsions. Other examples include sugar (though technically molecular, its crystal structure can show similar principles of orderly arrangement), calcium carbonate (chalk and seashells), and many common minerals.
Must Read
Getting started with this idea is easier than you might think! You don't need a fancy lab. Grab some table salt and a magnifying glass. Look closely at the crystals. See how they often have a regular, geometric shape? That’s a visual clue to the orderly arrangement of ions. You can also compare the texture of salt to something like sand or sugar. Notice the differences? Those relate to how the particles are held together. For a fun family experiment, try dissolving salt and sugar in water separately. Observe how quickly they dissolve – this relates to how the water molecules can interact with and pull apart the ions or molecules.
Understanding the forces that hold ionic solids together isn't just a science lesson; it's about appreciating the fundamental principles that create the solid world around us. From the salt in your food to the minerals in the earth, these invisible forces are hard at work, creating order and stability. It’s a small piece of knowledge that adds a big layer of understanding and, dare we say, enjoyment to the everyday!