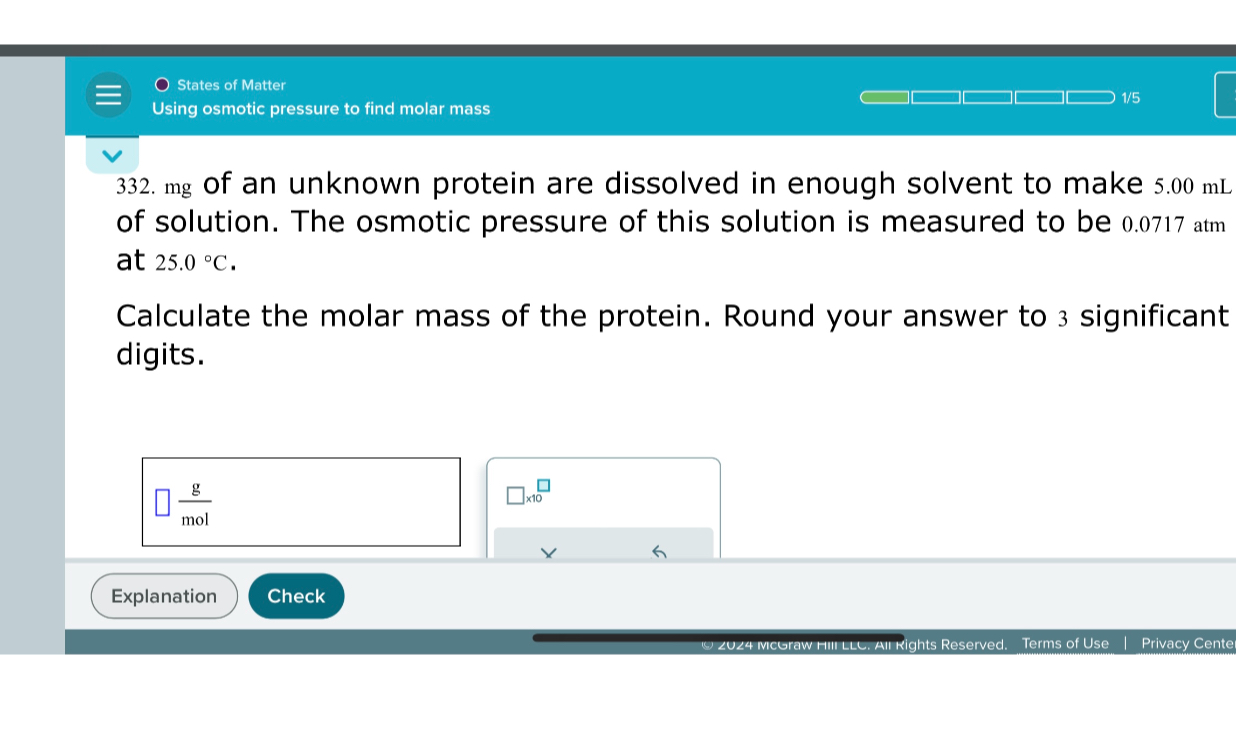
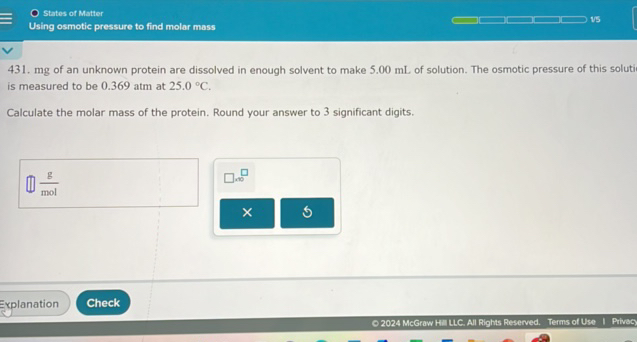
Using Osmotic Pressure To Find Molar Mass

Hey there, science explorers! Ever feel like some scientific concepts are locked away in a fancy lab, only to be understood by folks in white coats? Well, today, we’re going to unlock a pretty cool one together, and I promise it’s less about bubbling beakers and more about… well, let's just say something you might encounter with your morning toast!
We’re diving into the wonderfully weird world of osmotic pressure. Now, that might sound a bit intimidating, but stick with me. Think of it as nature’s way of balancing things out, a silent, invisible force that’s actually super important in our everyday lives. And the really neat part? We can use this fancy osmotic pressure to figure out the molar mass of things. Yep, we're talking about finding out how "heavy" a molecule is, in a chemical sense, just by watching how water moves!
The Unsung Hero of Water Movement
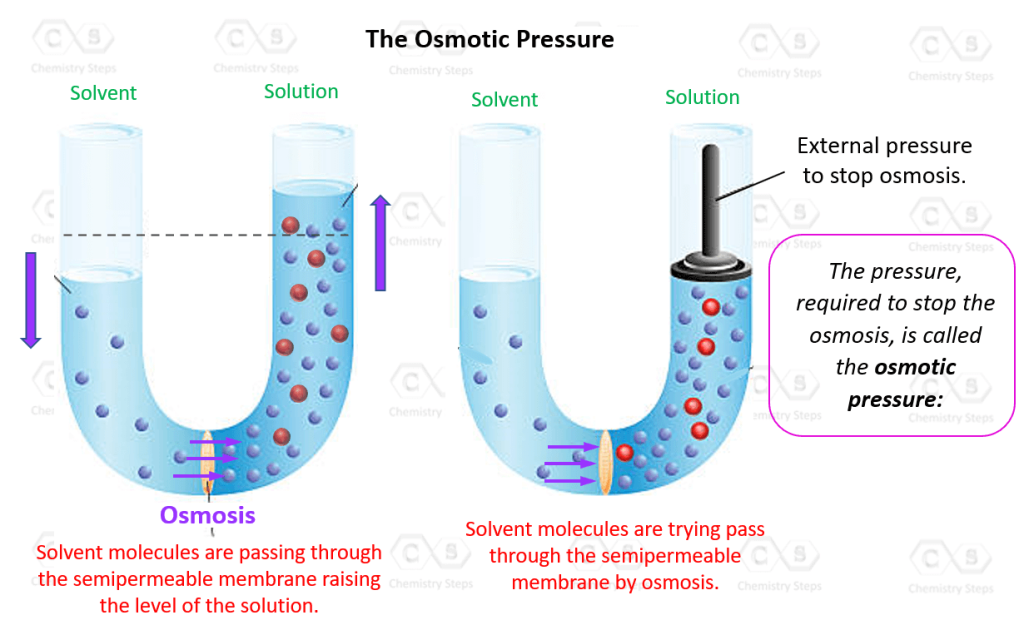
So, what exactly is osmotic pressure? Imagine you have two rooms, separated by a special wall. This wall is like a bouncer at a club, only letting certain guests through. In our case, this wall is a semipermeable membrane. Think of it as a super-selective sieve. It lets tiny things, like water molecules, zip through, but it’s a no-go zone for bigger guys, like sugar or salt molecules.
Must Read
Now, let’s say we have a room with just plain water and another room with sugary water. The semipermeable membrane is between them. What do you think will happen? The water molecules, being the social butterflies they are, will see the sugary side is a bit… diluted. They’ll want to even things out, to make the concentration of sugar (and water!) the same on both sides. So, they’ll start to move from the plain water side into the sugary water side.
This movement of water across a semipermeable membrane from an area of lower solute concentration to an area of higher solute concentration is called osmosis. And osmotic pressure? That’s the push, the force that makes this water movement happen. It's the effort water makes to try and dilute the more concentrated solution until both sides are happy and balanced.
Relatable Moments with Osmosis
You’ve experienced osmosis more times than you realize! Think about when you soak dried fruit, like raisins or prunes. You put them in water, and after a while, they plump up, right? That’s osmosis at work! The water molecules from the bowl move into the dried fruit (which has a higher concentration of sugars and other stuff inside) to try and even things out. The fruit cells act like mini semipermeable membranes.
Or consider your own body. Your cells are surrounded by membranes. When you drink a lot of water, your body can adjust the water balance. When you eat salty snacks, your cells can get a bit dehydrated as water moves out to balance the salt concentration in your bloodstream. It’s all about that osmotic pressure, keeping your internal environment just right.
Ever seen those little cucumbers you put in brine to make pickles? They start out a bit wrinkly. But as they sit in the salty brine, water moves out of the cucumber into the more concentrated salt solution. This is actually a slightly different example, where water moves out of the cucumber due to the high salt concentration outside, making them more firm. But the principle of water moving across a membrane to try and balance concentration is still the core idea!
Why Should We Care About This Invisible Push?
Okay, so water moves around. Big deal, right? Well, yes, it is a big deal! Osmotic pressure is crucial for life. Plants rely on it to draw water up from the soil into their roots. Your kidneys use it to filter waste from your blood. It’s involved in everything from how food is preserved (think of jams and cured meats, where high sugar or salt concentrations prevent bacterial growth by drawing water out of the microbes) to how our cells function.
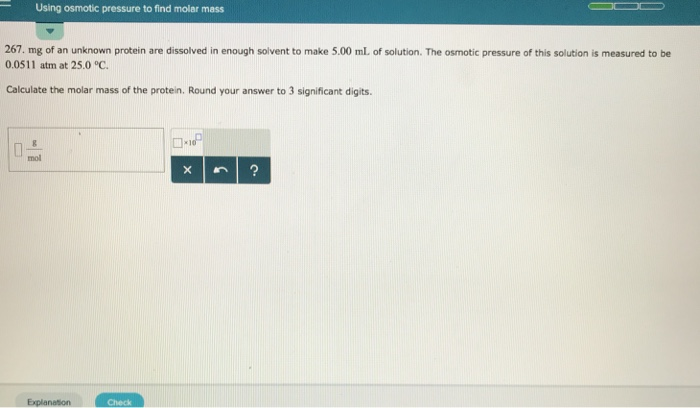
And here’s where it gets really cool for us budding chemists and curious minds: we can use this natural phenomenon to measure things! Specifically, we can use osmotic pressure to determine the molar mass of dissolved substances. Let’s call our dissolved substance our "mystery molecule."
Unmasking the Mystery Molecule: Molar Mass Detective Work
Imagine you have a mystery powder. You want to know what it is, and a key piece of information is its molar mass – essentially, how much "stuff" is in each of its molecules.
Here’s the general idea: you dissolve a known amount of your mystery molecule into a solvent, usually water. Then, you use a special setup with that semipermeable membrane we talked about. One side has pure solvent (like pure water), and the other side has your solution (water with your mystery molecule dissolved in it).
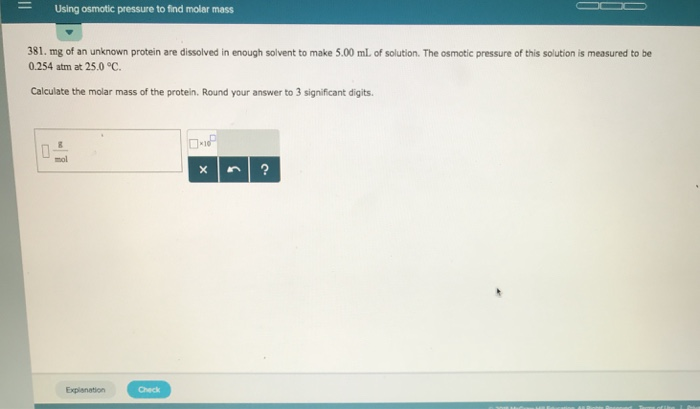
As we learned, water will move into the solution side to dilute it. This movement will create a difference in liquid levels, or a pressure difference. This pressure difference is directly related to how much "stuff" (how many solute molecules) is in the solution. The more solute molecules you have, the harder water has to push to dilute it, meaning a higher osmotic pressure.
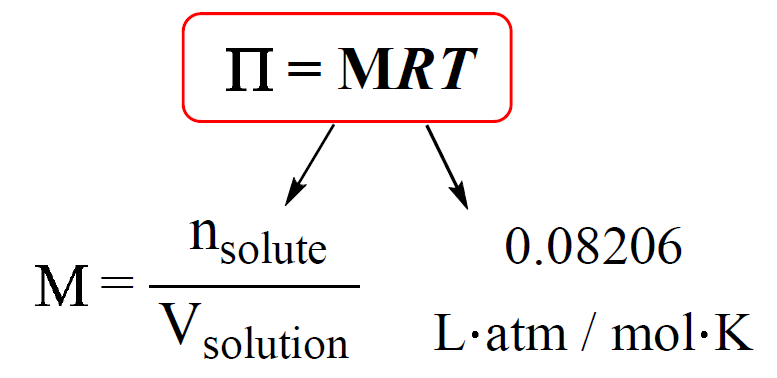
There’s a fantastic equation, often called the van't Hoff equation, that beautifully connects osmotic pressure ($\pi$) to the concentration of the solute (c), the gas constant (R), and the temperature (T). It looks like this: $\pi = iMRT$. For simplicity, let’s assume our mystery molecule doesn't break apart in water (so $i=1$), and we're often working at a constant temperature, so it simplifies even further in our minds to: osmotic pressure is proportional to concentration.
So, if you measure the osmotic pressure ($\pi$) of your solution, and you know the temperature (T) and the gas constant (R), you can figure out the concentration (c) of your mystery molecule. And here’s the best part: if you know how much mass of your mystery molecule you dissolved to get that concentration, you can then calculate its molar mass! It’s like a chemical fingerprint!
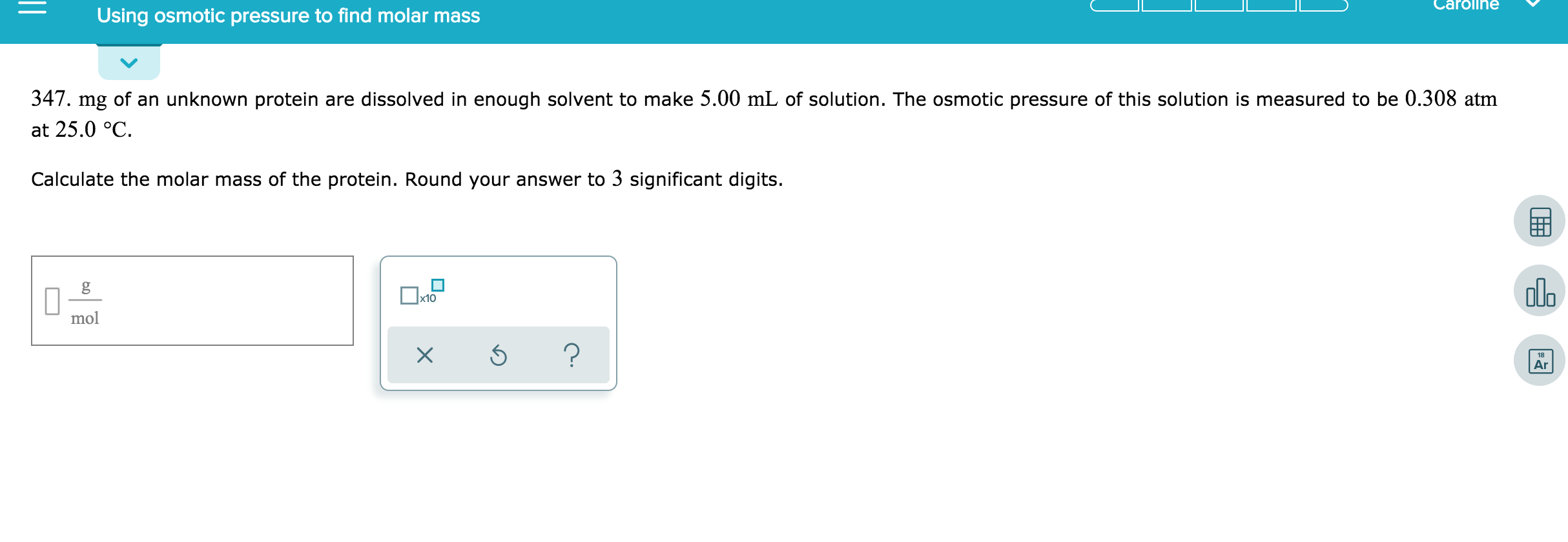
Why This is Cooler Than You Think
Why bother with this method when we have other ways to find molar mass? Well, for some molecules, especially very large ones like polymers (think plastics!) or large biological molecules (like proteins!), other methods can be tricky or downright impossible. Osmotic pressure offers a gentler approach. It's especially useful for colligative properties, which are properties of a solution that depend only on the number of solute particles, not on their identity. Molar mass determination is a prime example of this.
Imagine you’re a baker trying to figure out the precise amount of sugar in a new recipe. If the sugar molecules were too tiny to see or weigh directly, but you knew that a certain sugar concentration would make your dough rise just so high (due to osmotic pressure within the yeast cells!), you could work backward to figure out how much sugar you’d added. It’s that kind of indirect, clever detective work!
So, the next time you see a raisin plump up, or feel that slight thirst after a salty meal, remember the silent, powerful force of osmotic pressure. It’s not just a concept in a textbook; it’s an everyday phenomenon that keeps our world, and even our bodies, in balance. And it’s a pretty neat trick for uncovering the secrets of the molecular world, one water molecule at a time!