Total Number Of Valence Electrons In Co3 2-
So, I was at my niece's birthday party the other day, right? And she’s seven, and she’s really into, like, building forts. Not just any forts, though. These are architectural masterpieces. We’re talking elaborate tunnel systems, secret escape routes, and enough cushions to rival a cloud factory. I, being the designated “fun uncle,” was tasked with sourcing the primary building material: old blankets. Lots and lots of old blankets. And as I’m wrestling this Everest of patterned fabric, trying to make it fit into the back of my car, I’m thinking, “Man, this is a lot of… stuff. Like, how many individual threads are in this thing?”
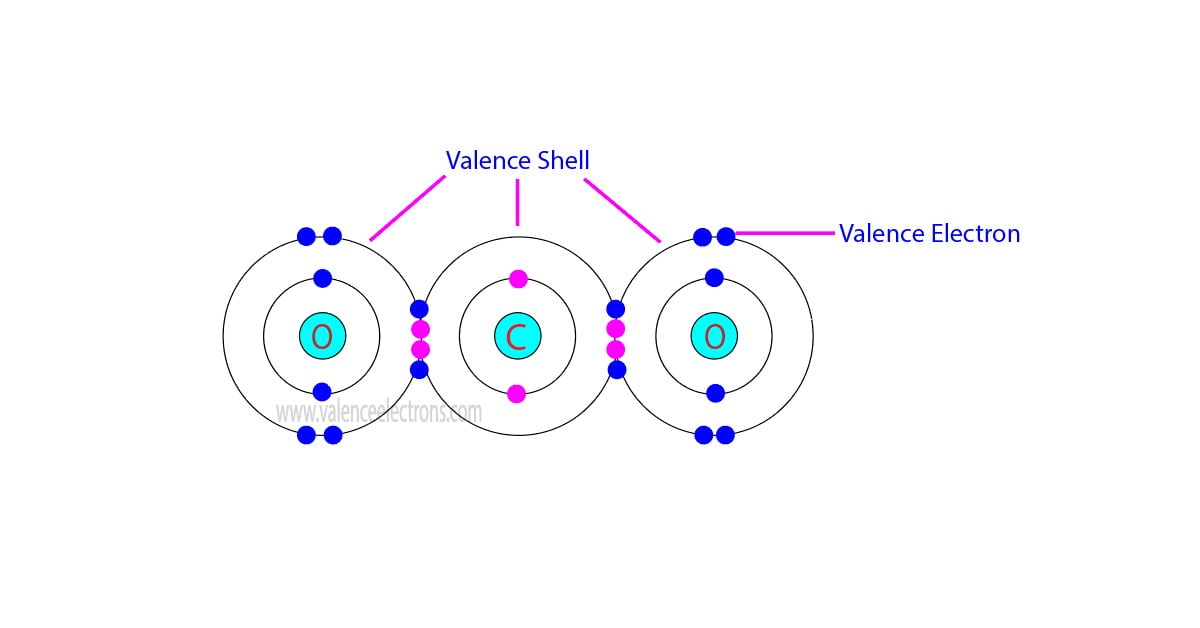
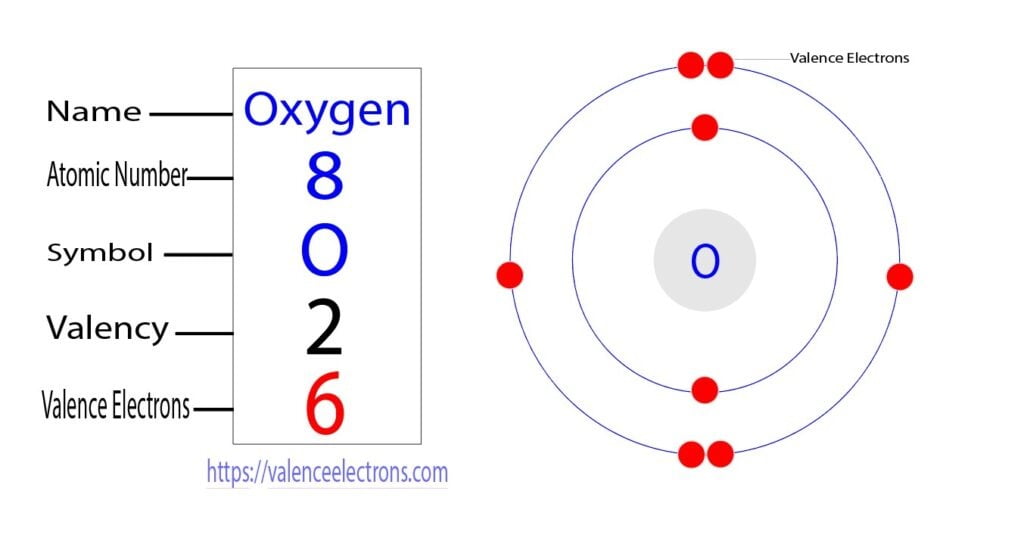
It got me pondering about the fundamental building blocks of everything around us. You know, the tiny bits that make up the big bits. And that, my friends, is a surprisingly similar feeling to figuring out the total number of valence electrons in, say, a rather dramatic-sounding ion like Co₃²⁻. Sounds fancy, right? But at its core, it’s just about counting up the "stuff" that matters for how things connect and behave. And like those blanket threads, valence electrons are the real MVPs of chemical bonding. They’re the outermost electrons, the ones that get all handsy and decide to hook up with other atoms. Pretty neat, huh?
Now, before we dive headfirst into the glorious world of cobalt and its merry band of electrons, let's have a quick refresher. What are valence electrons, exactly? Think of an atom like a little miniature solar system. You’ve got the nucleus (the sun) in the center, and electrons (the planets) orbiting it in different energy levels or shells. The valence electrons are the ones chilling in the outermost shell. They're the ones that interact with other atoms, forming bonds and creating all the cool molecules and compounds we see, from water (H₂O) to, well, the stuff that makes up my niece’s fort-building empire.
Must Read
Why do they matter so much? Because atoms, bless their little energetic hearts, are often a bit greedy. They like to have their outermost shell completely full. It's like a universal rule of atomic happiness. When an atom has a full valence shell, it's stable and content. If it doesn't, it'll go out of its way to gain, lose, or share electrons to get to that blissful state. This whole electron-trading game is what chemistry is all about. It’s the cosmic dance of electron sharing and transfer.
So, let’s get to the star of our show today: Cobalt, or Co. This isn't just any old element; it's a transition metal, which means it’s got a bit of extra flair. Transition metals are known for their colorful compounds and their ability to exist in multiple oxidation states (which is basically a fancy way of saying they can be more or less electron-rich, depending on the situation). This makes them super interesting to chemists. Think of them as the chameleons of the periodic table.
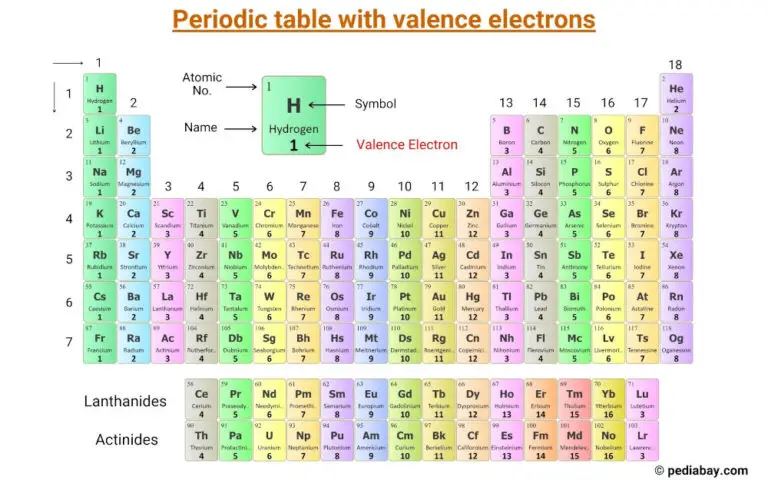
To figure out the valence electrons of a neutral cobalt atom, we need to look at its position on the periodic table. Cobalt has an atomic number of 27. This means a neutral cobalt atom has 27 protons and 27 electrons. Now, the electron configuration is a bit more complex than a simple 2-8-8-? setup because of those d-orbitals that transition metals love. However, for determining valence electrons in common scenarios, we generally focus on the outermost s and p orbitals. For cobalt, the electron configuration ends with 4s² 3d⁷. The 4s electrons are in the outermost shell, so we consider those as valence electrons. Typically, in transition metals, the outermost 's' electrons are the primary valence electrons, and sometimes the 'd' electrons in the adjacent shell also participate in bonding. For simplicity, and in many common contexts, we often count the 4s electrons as the core valence electrons for cobalt, which would be 2.
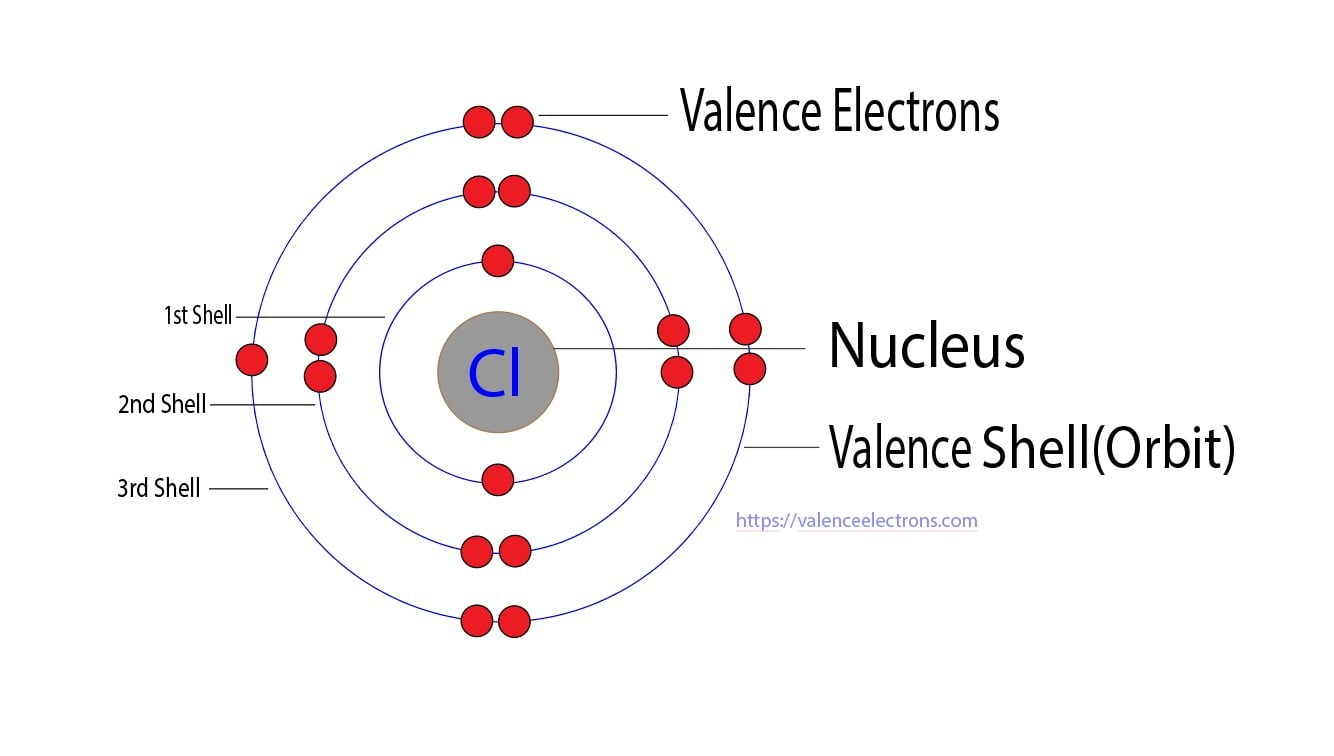
However, cobalt is famous for its ability to lose electrons and form positive ions. The most common ions are Co²⁺ and Co³⁺. When cobalt forms an ion, it loses electrons from its outermost shell. For Co²⁺, it loses two electrons, typically from the 4s orbital, leaving it with a configuration that often involves the 3d orbitals. For Co³⁺, it loses three electrons. This is where it gets a little more nuanced with transition metals. For Co³⁺, it's generally understood to lose the two 4s electrons and one electron from the 3d orbital, resulting in a 3d⁶ configuration. This ability to lose electrons, especially forming the stable Co³⁺ ion, is crucial for understanding its chemistry.
Now, we're dealing with a polyatomic ion: Co₃²⁻. This isn’t just a single cobalt atom floating around; it’s a cluster of three cobalt atoms that have collectively gained a charge of -2. This is a bit unusual for cobalt, which typically forms positive ions. This compound is likely an organometallic species, where cobalt atoms are bonded to organic ligands and the entire complex carries a negative charge. In these complex ions, the concept of "valence electrons" can become a bit more intricate because we're looking at the total number of electrons involved in bonding within the entire ion, not just for a single atom in isolation.
So, how do we tackle Co₃²⁻? We need to break it down. First, we need to consider the contribution of each cobalt atom. A neutral cobalt atom (Co) has 27 electrons. If we consider the valence electrons specifically for bonding in a typical context, and acknowledging the complexity of transition metals, we often think about the electrons in the outermost shells. For simplicity in many introductory contexts, we might consider the electrons that are readily available for bonding. However, for transition metals, it's more accurate to consider the total number of electrons outside of the core noble gas configuration. The noble gas preceding cobalt is Argon (Ar), which has 18 electrons. So, cobalt has 27 - 18 = 9 electrons beyond the Argon core. These are the 4s² and 3d⁷ electrons.
Let’s take a slightly different approach that’s often used for these types of complex ions. We'll count the total number of valence electrons contributed by all the atoms, and then adjust for the charge. A neutral cobalt atom (Co) has an electron configuration of [Ar] 4s² 3d⁷. This means it has 2 + 7 = 9 electrons in its outermost shells (4s and 3d) that are typically considered valence electrons for bonding purposes. So, for three cobalt atoms, the total contribution from cobalt would be 3 * 9 = 27 electrons. Now, here’s the crucial part: the ion has a charge of -2. This negative charge means the ion has gained 2 extra electrons. Therefore, the total number of valence electrons in Co₃²⁻ is the sum of the valence electrons from the three cobalt atoms plus the two extra electrons from the negative charge: 27 + 2 = 29 electrons.
Wait, 29? That sounds… a bit odd for a typical number. Is there something I’m missing? This is where the nature of these complex ions, especially those involving transition metals and negative charges, can really bend our brains. The bonding in such species can be quite delocalized, meaning the electrons aren’t neatly confined to individual atoms or bonds in the way they are in simpler molecules.
Let’s pause for a moment. When we talk about "valence electrons" in polyatomic ions, especially ones with transition metals and negative charges, it's often about the total number of electrons available for bonding and contributing to the overall electronic structure of the ion. This is different from just adding up the valence electrons of neutral atoms and adjusting for charge. The actual bonding in Co₃²⁻ is likely to be quite sophisticated, involving delocalized bonding where electrons are shared across the entire Co₃ unit.
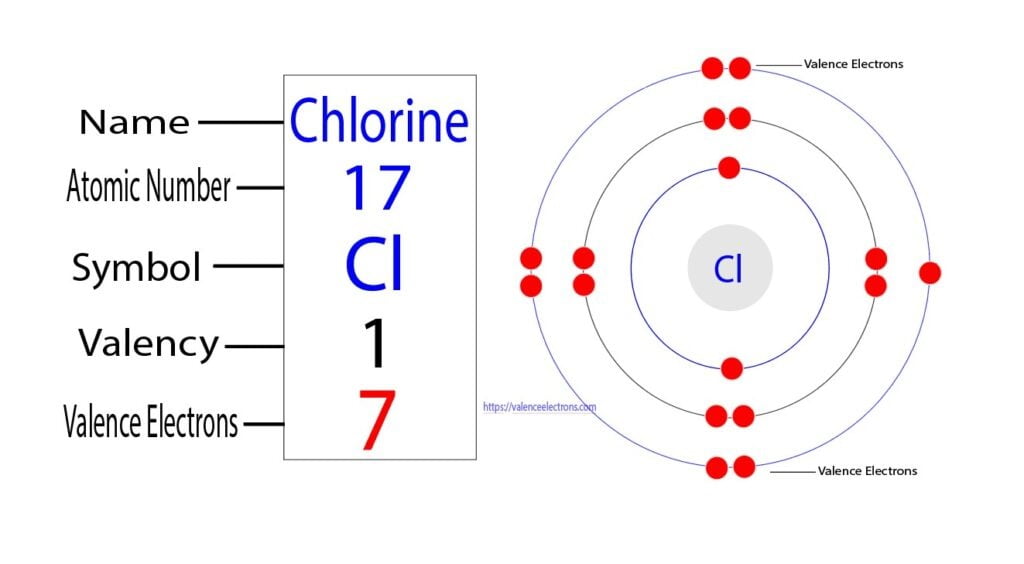
Let's try a more rigorous approach that accounts for the specific electronic configurations and bonding. For transition metals, it's often more useful to consider all electrons outside the preceding noble gas configuration as potential valence electrons. For Cobalt, this is the [Ar] 4s² 3d⁷ configuration, so 9 electrons outside of Argon. For Co₃²⁻: Each Co contributes 9 electrons (from 4s and 3d orbitals). Total from 3 Co atoms = 3 * 9 = 27 electrons. The -2 charge means we add 2 electrons. Total valence electrons = 27 + 2 = 29 electrons. Okay, 29 electrons. It's still 29. Why does this number feel a bit… unexpected? In many common molecules, you end up with even numbers of electrons, especially when considering total electrons in bonds (pairs). But when you have unpaired electrons or complex delocalized systems, odd numbers can absolutely happen. Think about radicals in organic chemistry; they have an odd number of valence electrons. And these complex cobalt ions can have quite intricate electronic structures.
Perhaps the interpretation of "valence electrons" here needs to be precise. If we are strictly talking about the sum of electrons in the outermost 's' and 'd' orbitals for each neutral cobalt atom, plus the extra electrons from the charge, then 29 is the result. It highlights that this is not a simple ionic compound where you have discrete cations and anions. This is a covalently bonded cluster of cobalt atoms with an overall negative charge.
Let’s consider an alternative perspective. Sometimes, for transition metals in these complex anions, the focus shifts to the electrons that are most involved in the bonding, which can be a subset of the total valence electrons. However, without knowing the specific ligands attached to the cobalt atoms in Co₃²⁻, it's hard to say definitively. The formula "Co₃²⁻" suggests a cobalt cluster anion, and its precise structure and bonding would depend on what it's bonded to.
However, if the question is simply about the total number of valence electrons as calculated by summing up the valence electrons of the neutral atoms and then accounting for the charge, then the calculation leading to 29 is the standard method for this type of problem. It's the number of electrons that the entire Co₃²⁻ unit possesses for its electronic structure and bonding.
It’s like if you were trying to count all the LEGO bricks to build a massive spaceship. You count all the red bricks, all the blue bricks, all the yellow bricks, and then you realize you bought an extra pack of green bricks, so you add those in. You end up with a big, potentially uneven number of total bricks. The electrons are similar; we're accounting for all the available ones that make up this specific chemical entity.
So, to wrap this up in a neat little bow, the total number of valence electrons in Co₃²⁻ is calculated by taking the number of valence electrons in three neutral cobalt atoms and adding the two extra electrons due to the -2 charge. A neutral cobalt atom has 9 valence electrons (4s² 3d⁷). Therefore, three cobalt atoms contribute 3 * 9 = 27 electrons. Adding the 2 extra electrons from the charge gives us a grand total of 29 valence electrons. It’s a number that speaks to the complexity and interesting bonding behavior of such cobalt clusters!