The Total Number Of Lone Pairs In Ncl3 Is

Ever looked at a molecule and thought, "Man, that thing looks a little lonely"? It’s like when you’re at a party, and everyone’s in their little cliques, and there’s just one guy awkwardly standing by the snack table, trying to look busy. Well, in the world of chemistry, we’ve got our own versions of these slightly awkward, solo performers. Today, we're diving into the not-so-mysterious, actually quite chill world of NCl₃, and figuring out just how many of these "lone pairs" it’s rocking.
Think of lone pairs like the extra socks you find in the laundry. You know, the ones that have mysteriously lost their sole mates? They're there, they’re doing their thing, but they're not actively paired up with anyone. In the grand ballroom of a molecule, these lone pairs are the wallflowers, or maybe the super-chill, independent types who don’t need a dance partner to feel complete. They’ve got their own energy, their own vibe, and they’re just… there.
So, we’re talking about NCl₃. That’s Nitrogen (N) and three Chlorine (Cl) atoms chilling together. Picture it like a little family gathering. Nitrogen is the parent, and the three Chlorines are its kids. Now, just like in any family, everyone’s got their own stuff going on. They’re all sharing this molecular space, but how they’re interacting is what makes things interesting.
Must Read
In chemistry, when atoms get together to form a molecule, they do it by sharing or transferring electrons. It’s basically their way of making friends and staying stable. Think of it like sharing a pizza. You either split slices (covalent bonds) or someone just hands over a whole slice (ionic bonds, but let’s not get too complicated). For NCl₃, we’re mostly dealing with sharing. Nitrogen and Chlorine are pretty good at sharing, forming what we call covalent bonds.
Each atom wants to have a certain number of electrons around it to feel like it’s hit the molecular jackpot, the stable, happy number. For many atoms, especially in the second row of the periodic table (like Nitrogen and Chlorine), this magic number is eight. It's like having the perfect number of friends at your poker game – everyone’s got a role, and the game is just right. This is often referred to as the octet rule. It’s a rule, but like most rules, there are sometimes… exceptions. Or at least, ways of interpreting it.
Let’s start with our central character, Nitrogen. Nitrogen is sitting pretty in Group 15 of the periodic table. That means it usually comes with five valence electrons. Think of these valence electrons as Nitrogen’s "party favors" – the ones it brings to the molecular bash. These are the electrons that get to mingle and form connections.
Now, Nitrogen needs to make some friends, and it’s got three Chlorine atoms lined up, ready to be its pals. Chlorine, on the other hand, is in Group 17. That’s a whole different neighborhood. Chlorine has seven valence electrons. So, each Chlorine brings seven party favors to the gathering. And remember, we have three of these Chlorine characters.
When Nitrogen decides to bond with these three Chlorines, it’s going to share some of those party favors. Typically, Nitrogen will form a single covalent bond with each Chlorine atom. This means Nitrogen shares one of its electrons with a Chlorine, and that Chlorine shares one of its electrons with Nitrogen. Poof! A bond is formed. It’s like a handshake, but with electrons.
So, Nitrogen uses three of its five valence electrons to form those three N-Cl bonds. Each bond uses one electron from Nitrogen and one from a Chlorine. Now, remember Nitrogen started with five valence electrons. It’s used three for bonding. What’s left? Two electrons. These two electrons are like the ones Nitrogen kept in its pocket after handing out enough for handshakes. They didn’t get used in a bond.
And what do we call those leftover, un-bonded electrons? You guessed it: a lone pair! These are the electrons that just hang out around the Nitrogen atom, not actively involved in sharing with any particular Chlorine. They’re like the cool, quiet observer, just chilling on the sidelines.
So, right off the bat, we know Nitrogen contributes one lone pair to the party. That’s one pair of electrons that isn't holding hands with another atom in a bond. It’s just… a pair.
But wait, we’re talking about the total number of lone pairs in NCl₃. Does that only include the ones on Nitrogen? Well, technically, the question is a little ambiguous if you're thinking about all lone pairs within the entire molecule. If we're being super thorough, we should also consider the lone pairs on the Chlorine atoms. After all, they’re part of the NCl₃ crew!
Let’s go back to our Chlorine friends. Each Chlorine atom started with seven valence electrons. We know that one of those electrons from each Chlorine was used to form a bond with Nitrogen. So, each Chlorine atom used one electron for bonding. That leaves six electrons on each Chlorine atom that are not involved in bonding.
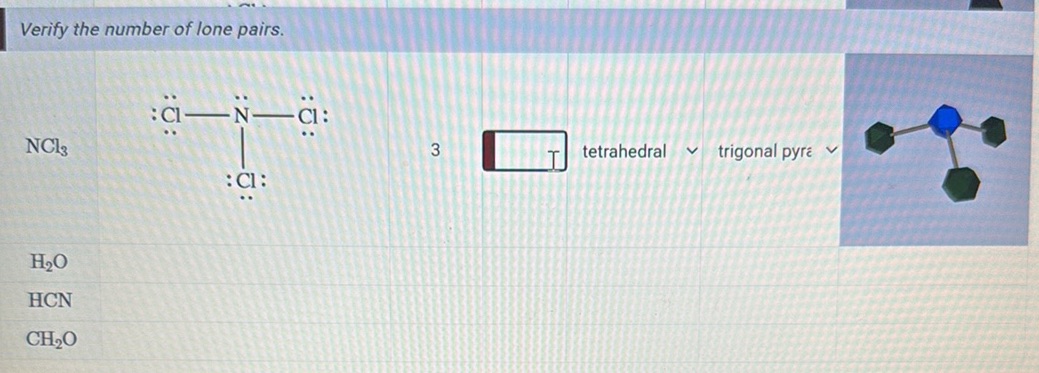
Now, six electrons, when you think about them in pairs, that makes three lone pairs per Chlorine atom. It’s like each Chlorine, after its friendly handshake with Nitrogen, has three little buddies (pairs of electrons) that just stick around with it. They’re not going anywhere, they’re just part of Chlorine’s personal space.
Since there are three Chlorine atoms in NCl₃, and each of them has three lone pairs, that means we have a total of 3 Chlorine atoms * 3 lone pairs/Chlorine atom = 9 lone pairs on the Chlorine atoms. These are the lone pairs that belong to the Chlorine kids, so to speak.
So, to get the total number of lone pairs in the entire NCl₃ molecule, we need to add up the lone pairs on the Nitrogen and the lone pairs on all the Chlorines. We found one lone pair on Nitrogen, and a whopping nine lone pairs spread across the three Chlorines.
That brings our grand total to: 1 lone pair (on Nitrogen) + 9 lone pairs (on the Chlorines) = 10 lone pairs.
Ten lone pairs! That’s quite a collection. It’s like finding a whole drawer full of those single socks. They're not doing much in terms of bonding, but they’re definitely there, taking up space and influencing how the molecule behaves. These lone pairs are super important, even if they’re not actively "holding hands" with other atoms.
Think about it this way: these lone pairs, especially the ones on the central Nitrogen atom, have a bit of a reputation. They’re often described as being a bit "pushy." They’ve got negative charge, and negative charges don’t really like being too close to each other. So, these lone pairs tend to push the bonding pairs away, influencing the molecule's shape. It’s like when you’re trying to arrange a bunch of balloons in a small room – the balloons (lone pairs and bonding pairs) are going to spread out as much as possible.
In the case of NCl₃, the lone pair on Nitrogen, along with the three bonding pairs to the Chlorines, makes for a pyramid-like shape. It’s not flat like a pancake; it’s more like a little three-sided tent. That lone pair is doing its best to stay out of the way of the N-Cl bonds, but its presence definitely shapes the whole structure. It’s like the sibling who’s always in the middle of the couch, making everyone else squish together a bit.
So, while the question might seem a bit technical, at its heart, it’s about counting electrons that are just hanging out. These aren’t the electrons doing the heavy lifting in the bonds, but they’re crucial players in the molecular drama. They contribute to the molecule’s overall electron cloud, affecting its reactivity and how it interacts with other molecules. It’s like knowing who’s at the party, even if they’re just standing in the corner.
The "lone pairs" are basically those electrons that don't participate in any chemical bonds within the molecule. They're just floating around the atom's nucleus, looking a bit like they're waiting for a dance partner that never shows up. In chemistry, we have a systematic way of figuring this out, using the concept of valence electrons and the octet rule. It's like a detective story, but with electrons as the suspects.
For NCl₃, we have one Nitrogen atom and three Chlorine atoms. Nitrogen, being in the fifth group of the periodic table, has five valence electrons. Chlorine, in the seventh group, has seven valence electrons. When they form bonds, they try to achieve a stable configuration, usually with eight electrons around them.

Nitrogen will use three of its five valence electrons to form single bonds with each of the three Chlorine atoms. This means Nitrogen is now sharing three pairs of electrons with the Chlorines. However, Nitrogen started with five electrons and only used three for bonding, leaving two electrons remaining. These two electrons form one lone pair on the Nitrogen atom. This lone pair is like the single headphone you find after unpacking a new gadget – it’s there, but it’s not part of the main setup.
Now, let's not forget the Chlorine atoms. Each Chlorine atom brings seven valence electrons to the table. When it forms a bond with Nitrogen, it uses one of its electrons. So, each Chlorine is left with 7 - 1 = 6 valence electrons that are not involved in bonding. These six electrons form three lone pairs around each Chlorine atom. Think of these as the little side dishes that come with the main meal – they’re not the star, but they’re definitely part of the spread.
Since there are three Chlorine atoms, and each has three lone pairs, we have a total of 3 Chlorine atoms * 3 lone pairs/Chlorine atom = 9 lone pairs on the Chlorine atoms. These are the little clusters of electrons that are just chilling with their respective Chlorine buddies.
So, if we're looking for the total number of lone pairs in NCl₃, we add up the lone pairs on Nitrogen and the lone pairs on all the Chlorines. That's 1 lone pair (on Nitrogen) + 9 lone pairs (on the three Chlorines) = 10 lone pairs.
It’s like a molecular treasure hunt, and the treasure is these non-bonding electrons. They're crucial for understanding how the molecule acts, its geometry, and its overall personality. Even though they’re not actively forming bonds, they’re influencing the molecule’s behavior in significant ways. They’re the quiet strategists, the unseen influencers. They give NCl₃ its particular character, making it behave the way it does in chemical reactions.
So next time you encounter a molecule, remember to look beyond the obvious bonds. Those lone pairs are there, like the unsung heroes or the quirky characters, adding depth and complexity to the molecular world. They’re not just random electrons; they’re essential components that contribute to the molecule’s unique identity and function. It's all about counting the little extras that make the whole picture complete. And in the case of NCl₃, those little extras add up to a grand total of ten!