The Sum Of Protons And Neutrons In An Atom

Hey there, curious minds! Ever wondered what makes up everything around us, from the air you breathe to the phone you're probably reading this on? Well, get ready to dive into a surprisingly fun and fundamental concept: the sum of protons and neutrons in an atom. Now, I know what you might be thinking – "Isn't that, like, super science-y and boring?" But trust me, understanding this basic building block of the universe can be incredibly satisfying and even a little bit mind-blowing. It's like unlocking a secret code to reality, and once you get it, you start seeing the world in a whole new light.
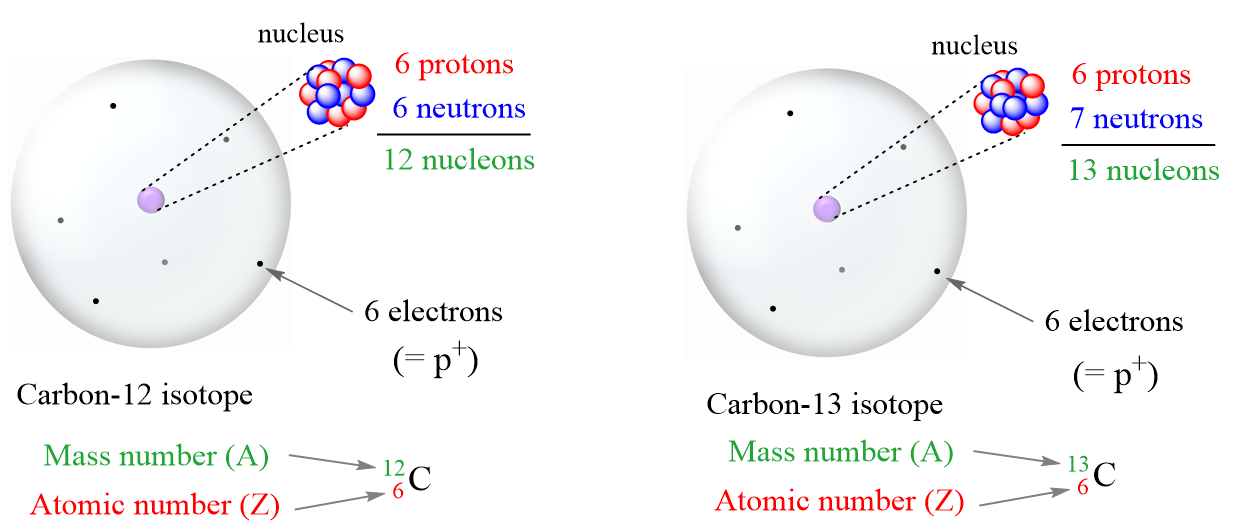
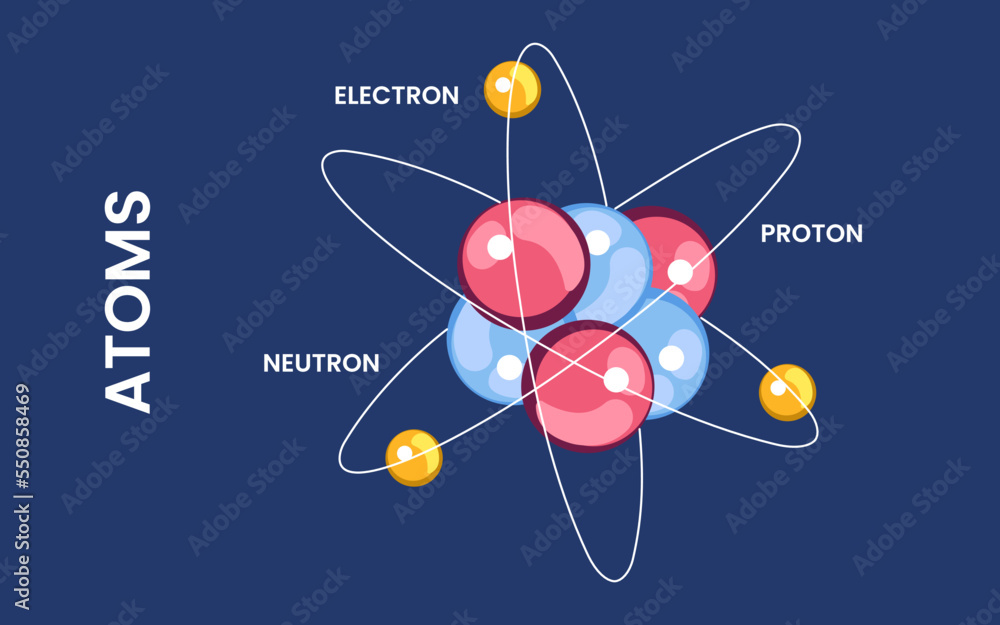
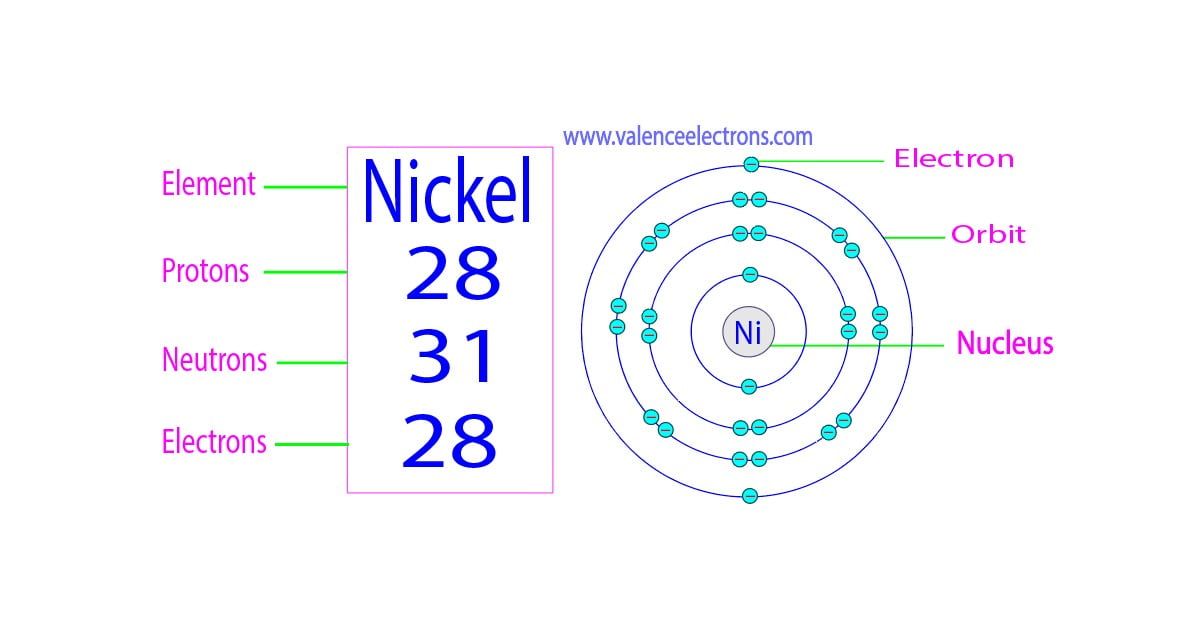
So, what exactly are we talking about? Think of an atom as a tiny solar system. In the center, you have the nucleus, which is like the sun. This nucleus is made up of two types of particles: protons (which have a positive charge) and neutrons (which have no charge). The sum of these two – the number of protons plus the number of neutrons – is known as the atom's mass number. Why is this so important? Because this number is essentially the weight of an atom, and it plays a crucial role in determining the properties and behavior of different elements. Without knowing this sum, we wouldn't be able to predict how atoms will interact, form molecules, or behave in chemical reactions.
The benefits of grasping this concept ripple through so many aspects of our everyday lives, even if we don't realize it. For instance, when you look at a nutritional label on your food, the presence and amounts of certain elements are directly tied to their atomic structure, including their mass numbers. Medicine relies heavily on understanding atomic behavior; think about medical imaging techniques like X-rays or PET scans, which involve manipulating atoms and their properties. Even the technology that powers your home appliances, from your refrigerator to your smartphone, is a testament to our deep understanding of how atoms, with their specific proton and neutron counts, interact.
Must Read
Common examples abound! The element carbon, the backbone of all organic life, has atoms with different numbers of neutrons, leading to isotopes like carbon-12 and carbon-14. This difference in neutron count is what allows us to use carbon dating to determine the age of ancient artifacts. Water (H₂O) is formed from hydrogen and oxygen atoms, and their mass numbers dictate how these elements combine to create this essential substance. Even the metals in your jewelry or the silicon in your computer chips owe their unique characteristics to the specific combination of protons and neutrons within their atoms.
So, how can you enjoy this exploration even more? Start by visualizing! Grab some LEGO bricks – different colored ones for protons and neutrons – and build yourself some atoms. It's a fun, tactile way to grasp the concept. Secondly, don't be afraid to look up periodic tables! They’re a treasure trove of information about elements, including their atomic numbers (which tells you the number of protons) and often their average atomic masses. Comparing the mass numbers of different elements can be quite fascinating. Finally, remember that this is just the tip of the iceberg! The more you learn about atoms, the more you'll appreciate the incredible complexity and beauty of the universe that surrounds us. So, go forth and count those protons and neutrons – your mind will thank you for it!