Student Exploration Ionic Bonds Answer Key

Hey there, science explorers! Ever feel like chemistry is all, like, super serious and dry? Nah, not today! We're diving into something totally cool: ionic bonds. And guess what? I've got the inside scoop – the answer key to making it all make sense. Let's get this party started!
So, imagine tiny little particles, like atoms, throwing a party. They're not just chilling, though. They're trying to be super stable. Think of it like wanting the perfect outfit for a big event. Atoms want that full outer shell, that complete set of electrons. It’s their version of looking amazing.
The Great Electron Swap
Now, how do they get that perfect electron outfit? It’s all about a little give and take. Ionic bonds happen when one atom is like, "Ugh, I have way too many electrons in my outer shell. It's a total drag!" And another atom is like, "Oh my gosh, I'm just begging for one more electron! It would complete me!"
Must Read
So, they do a swap. One atom gives away an electron. Poof! It becomes a positive ion. Think of it like donating to a good cause – you feel good, and the recipient is super happy. The atom that takes the electron becomes a negative ion. They're like, "Yay! I got what I needed!"
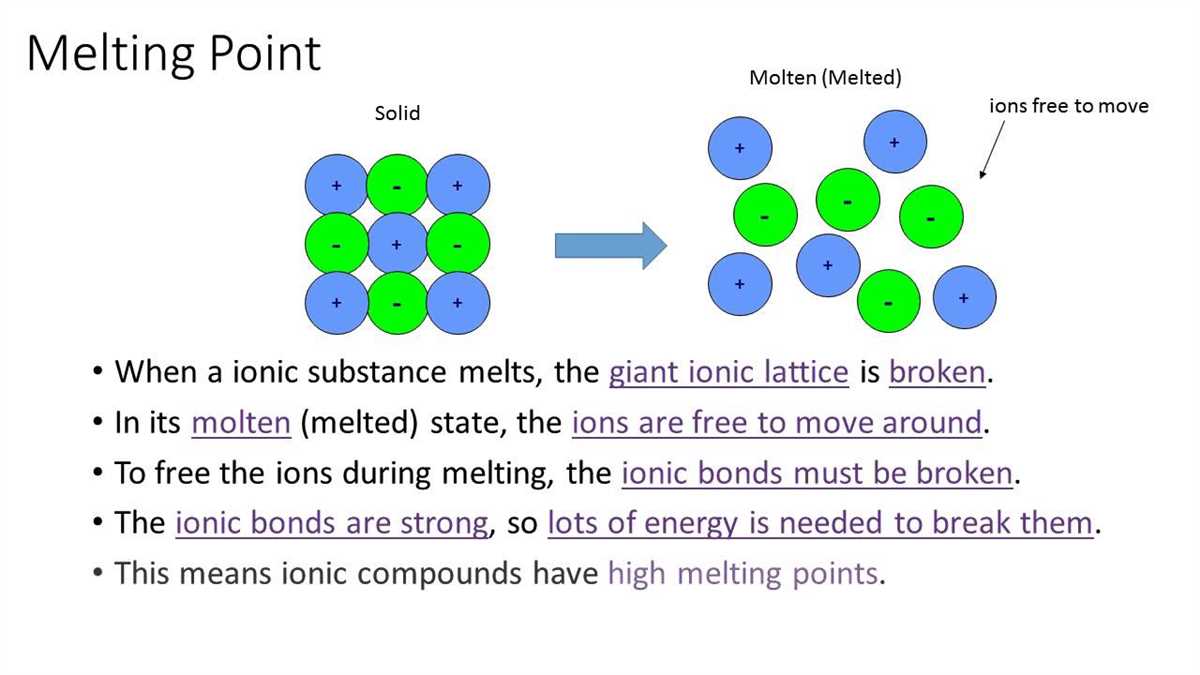
This is where the magic happens. Opposite charges, right? Positives and negatives. What do they do? They stick together! Like magnets, but with atoms. It's a super strong attraction. This is the ionic bond in action. It's like the universe's ultimate cosmic hug.
Salt? More Like Super-Bond!
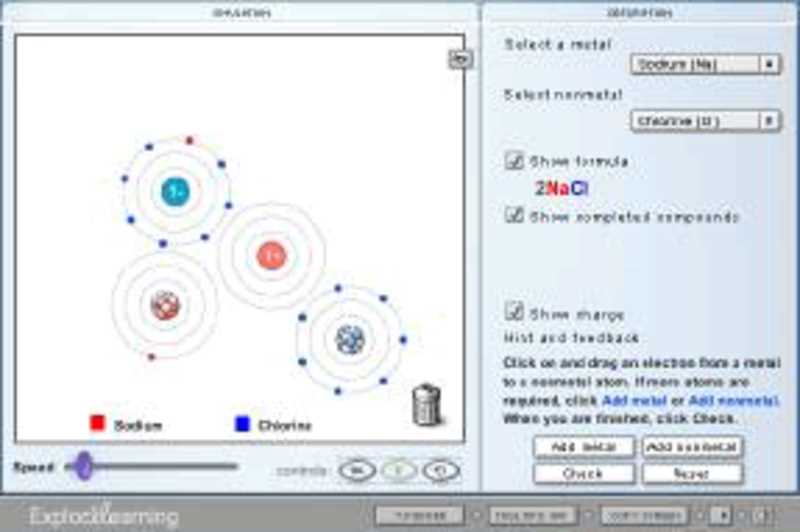
The most famous example? Table salt! Yep, that stuff you sprinkle on your fries? That's sodium chloride (NaCl). Sodium (Na) is the giver. It has one extra electron it's just itching to ditch. Chlorine (Cl) is the taker. It’s one electron short of a full house. So, sodium gives its electron to chlorine.
Sodium becomes Na⁺ (positive ion). Chlorine becomes Cl⁻ (negative ion). And bam! They’re stuck together, forming those shiny little salt crystals. Pretty neat, huh? It's like a tiny atomic romance leading to a delicious snack.
Think about it: the whole reason we have salt is because these two atoms decided to be generous and needy at just the right moment. It's a testament to the power of sharing… even if it's just electrons.
Why is this Fun? Let's Count the Quirks!
First off, the names! Cations (positive ions) sound like they’re yelling "Yay!" And anions (negative ions) sound a bit like they’re being put upon. It’s just fun to say. "Oh, that’s a sodium cation!" Sounds way cooler than "that’s a positively charged sodium atom," right?

Then there's the sheer electrifying nature of it all. We're talking about charged particles holding hands. It’s like a tiny, invisible dance party where everyone’s got a charge. And sometimes, these bonds can be so strong, they form these amazing crystal structures. Think of those sparkly geodes you see? Some of that sparkle is thanks to ionic bonds!
And get this: not all ionic compounds are boring white powders. Some are brilliantly colored! For example, copper(II) sulfate is a gorgeous blue. That color? It’s the electrons in the copper ions getting excited by light and giving off specific wavelengths. So, even colors in minerals are often tied to ionic bonding!
The "Who Gives What?" Game
One of the coolest parts of exploring ionic bonds is figuring out who gives and who takes. It’s like a game of chemical matchmaking. You look at the periodic table, and you can practically see which elements are destined to be electron donors and which are electron receivers.
![Ionic Bonds Gizmo Answer Key 2025 [FREE ACCESS] - AnswerKeyFinder](https://answerkeyfinder.com/wp-content/uploads/2022/07/Ionic-Bonds-Gizmo-Answer-Key.jpg)
Generally, metals are the givers. They’re on the left side of the periodic table. They’re like, "Here, take it, I've got plenty!" Nonmetals, on the right, are usually the takers. They’re always looking for that last electron to feel complete. It's a beautiful, predictable dance.
So, when you see a compound like magnesium oxide (MgO), you know magnesium (a metal) is going to give, and oxygen (a nonmetal) is going to take. Magnesium gives two electrons (it’s super generous!), and oxygen happily snags them. Voila! Another stable ionic compound.
Beyond the Basics: The Answer Key Unlocked!
Okay, so the "answer key" isn't really a sheet of answers, it's more like understanding the rules of the game. The key principles are: electronegativity (how much an atom wants electrons), electron transfer, and the resulting electrostatic attraction.

When you're working through problems, ask yourself: 1. Is this a metal and a nonmetal? (Likely ionic!) 2. How many electrons does each atom need to gain or lose to get a full outer shell? 3. What will the resulting charges be? 4. Opposite charges attract! They form the bond.
It's like solving a little chemical puzzle. And once you get it, it’s super satisfying. You start seeing these bonds everywhere! In batteries, in ceramics, even in your own bones (calcium phosphate is ionic!).
The Fun Never Stops!
The truly fun part is realizing that these tiny, invisible forces are responsible for so much of the world around us. The hardness of rocks, the ability of certain salts to dissolve in water, the conductivity of some materials – it all ties back to how atoms decide to share (or, more accurately, transfer) their electrons.
So, next time you're munching on some salt, or admiring a piece of crystal, give a little nod to the amazing world of ionic bonds. It’s not just chemistry; it’s a fundamental force that builds our universe, one electron transfer at a time. And understanding it is like having a secret superpower for understanding how things work. Pretty cool, right?