Select All That Apply Tonicity Of A Solution
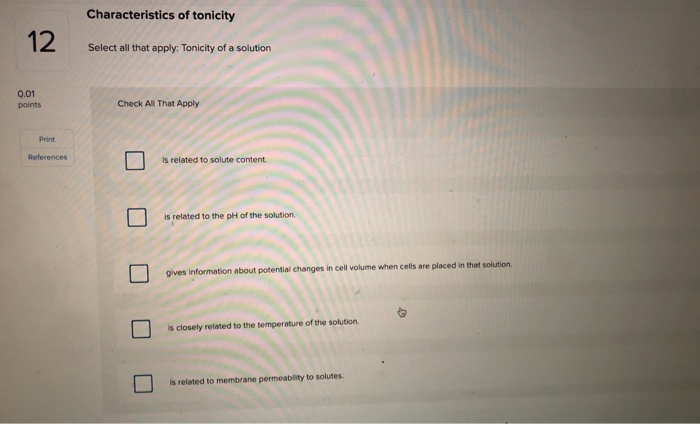
Hey there, science curious folks! Ever find yourself staring at a science textbook, or maybe just wondering about how things work at a super tiny level, and stumble upon some terms that sound… well, a little bit fancy? Today, we’re going to chat about one of those terms: Tonicity of a Solution. Sounds complex, right? But stick with me, because it's actually a really neat concept, and once you get it, you'll start seeing it everywhere!
So, what in the world is tonicity? Think of it like this: imagine you have a cell, and it’s floating around in some liquid, or a solution. This solution has stuff dissolved in it, like salt or sugar, right? Tonicity is basically a way of describing how the concentration of those dissolved things in the solution compares to the concentration of stuff inside the cell. It’s all about balance, or rather, the potential for imbalance!
Why Should We Even Care About This "Tonicity" Thing?
Okay, fair question. Why should you care about the water balance of a tiny cell? Because it’s super important for life! Cells are like little bags of jelly, and they need to maintain a specific internal environment to do their jobs. If they take in too much water, they might swell up and pop. If they lose too much water, they’ll shrivel up and get sad. Tonicity is the key player in making sure our cells stay happy and healthy. Think of it like keeping your phone charged – you don’t want it to run out of battery, and you don’t want to overcharge it and damage it either, right?
Must Read
This concept pops up in all sorts of cool places. Ever wonder why drinking a super sugary soda can make you feel a bit… off? Or why doctors give you IV fluids when you’re sick? Tonicity is at play!
The Three Main Flavors of Tonicity
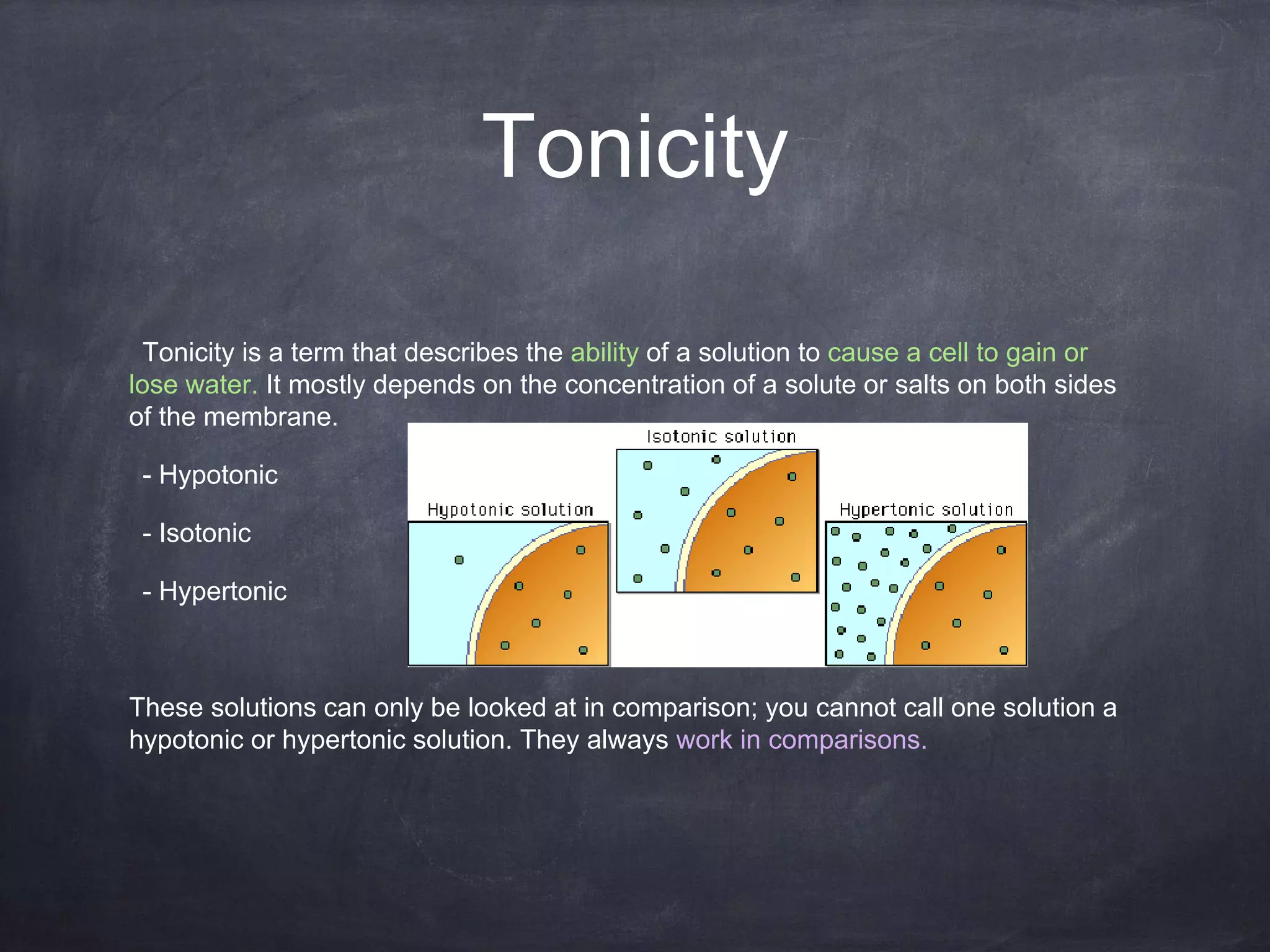
Scientists love to categorize things, and tonicity is no exception. We usually talk about three main types of solutions based on their tonicity: isotonic, hypotonic, and hypertonic. Let’s break them down, nice and easy.
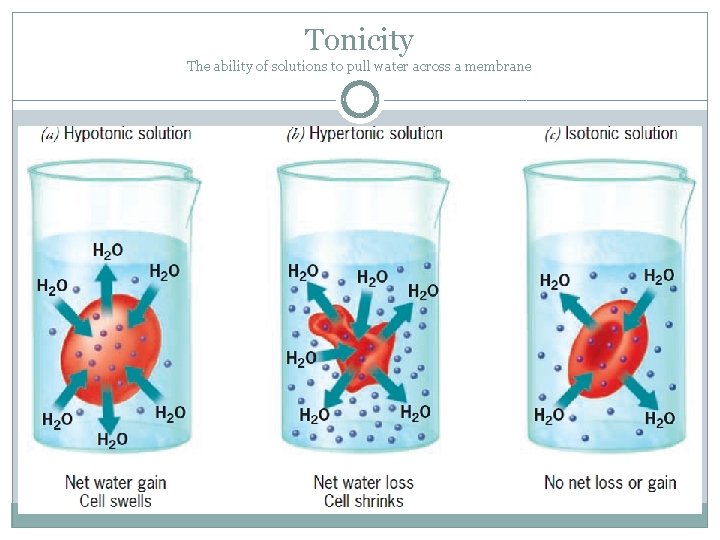
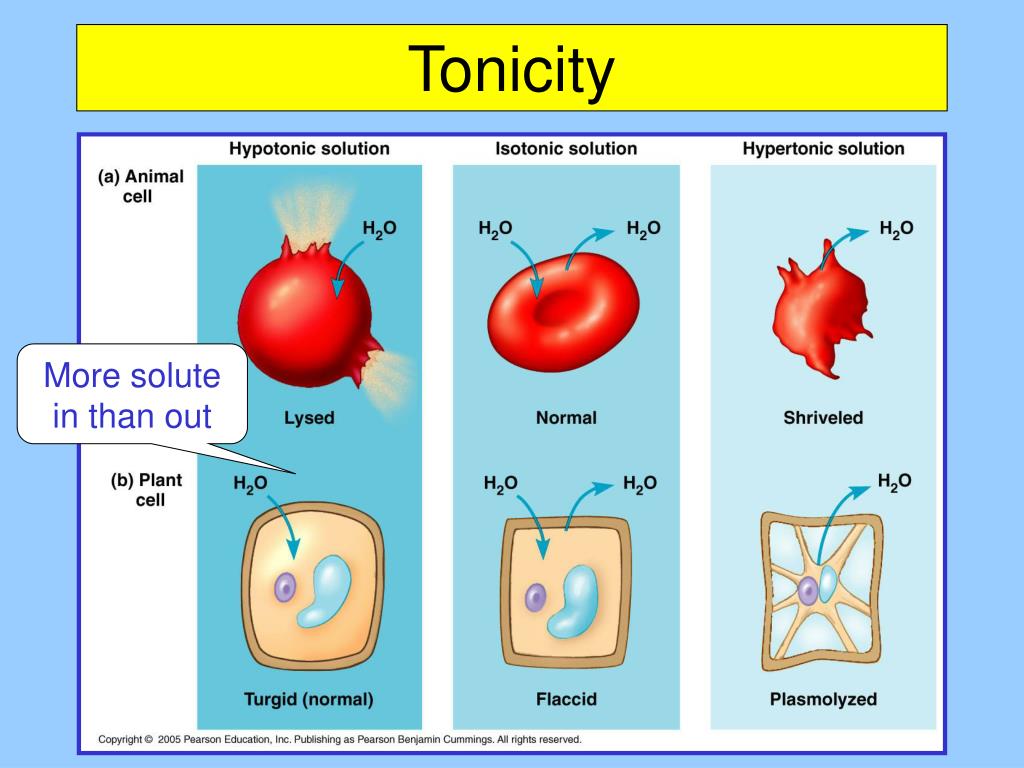
Isotonic: The Chill Zone
Imagine you’ve got a perfectly balanced seesaw. That’s kind of what happens in an isotonic solution. In this case, the concentration of dissolved stuff outside the cell is exactly the same as the concentration inside the cell. What does this mean for our cell? Well, water is pretty smart. It likes to move from areas where there's more of it to areas where there's less of it, trying to even things out. But in an isotonic solution, there's no real push for water to move in or out. It’s like a calm lake – things are just… chilling.
Water molecules will still be zipping around, of course, but for every water molecule that moves into the cell, another one moves out. So, the cell stays the same size. It's not gaining or losing water. This is the ideal state for many of our body cells. It's like wearing clothes that fit just right – comfortable and functional!
Hypotonic: The Water Party!
Now, let’s talk about hypotonic solutions. The prefix "hypo-" often means "less than" or "below." So, in a hypotonic solution, the concentration of dissolved stuff outside the cell is lower than the concentration inside the cell. What do you think happens then? You guessed it! Water is going to be more attracted to the inside of the cell, where there’s more "stuff" to dissolve. It’s like a magnet!
So, water starts rushing *into the cell. Imagine a balloon being filled with water. The cell will start to swell up. If it’s a plant cell, it’ll become nice and firm, which is called being turgid. Plants love this because it helps them stand up straight and keep their leaves perky. Think of a wilted plant suddenly perking up after a good watering – that’s the power of a hypotonic solution working its magic!
But for animal cells, like our red blood cells, this can be a problem. If they take in too much water, they can actually burst! This is called lysis. So, while hypotonic solutions can be great for plants, we have to be careful with them around our animal cells.
Hypertonic: The Great Escape!
Finally, we have hypertonic solutions. The prefix "hyper-" often means "more than" or "above." So, in a hypertonic solution, the concentration of dissolved stuff *outside the cell is higher than the concentration inside the cell. What’s water going to do now? It’s going to try and even things out by moving out of the cell and into the solution.
It’s like when you salt a slug – all the water gets pulled out, and it shrivels up. Our cells will do the same thing in a hypertonic solution. They’ll lose water and shrink. This process is called crenation. Imagine a raisin – it's basically a grape that’s lost a lot of its water and shriveled up. That's a cell in a hypertonic environment!

This is why drinking a super concentrated sugary drink might make you feel a bit dehydrated. The high sugar concentration in your digestive system can pull water out of your cells. Similarly, if you have a cut and you rinse it with very salty water, it might sting because the salt is pulling water out of the cells around the wound.
Real-World Tonicity Adventures
So, we’ve covered the basics. But where do we see this in action? Lots of places!
Medical IV Fluids: When someone is sick and needs fluids, doctors carefully choose the type of IV fluid. They usually use isotonic solutions, like normal saline (0.9% sodium chloride), because it won’t mess with the balance of their body’s cells. If they used a hypotonic solution, it could cause their red blood cells to burst. If they used a hypertonic solution, it could cause them to shrink. It’s all about maintaining that delicate balance!
Food Preservation: Ever wonder how jam stays good for so long, or how pickling works? High concentrations of sugar or salt are often used. These create a hypertonic environment for any bacteria or mold that might try to grow on the food. The high solute concentration pulls water out of the microbes, killing them or preventing them from multiplying. Pretty neat, huh? It’s a delicious way to use science!
Our Own Bodies: Even within our bodies, different environments have different tonicities. For example, the fluid in our blood plasma is generally isotonic to our cells. But think about your kidneys – they are masters at controlling water balance, adjusting the tonicity of urine to conserve or get rid of water as needed. It’s a complex but vital system!
The Takeaway: It's All About Balance
So, the next time you hear about the tonicity of a solution, don’t get intimidated! Just remember the three amigos: isotonic, hypotonic, and hypertonic. Think about whether water is likely to move into, out of, or stay put relative to a cell. It's all about that water-balance game, and understanding it helps us appreciate the incredible complexity and elegance of the living world around us, from the smallest microbe to our own amazing bodies. Pretty cool, right?



.jpg)