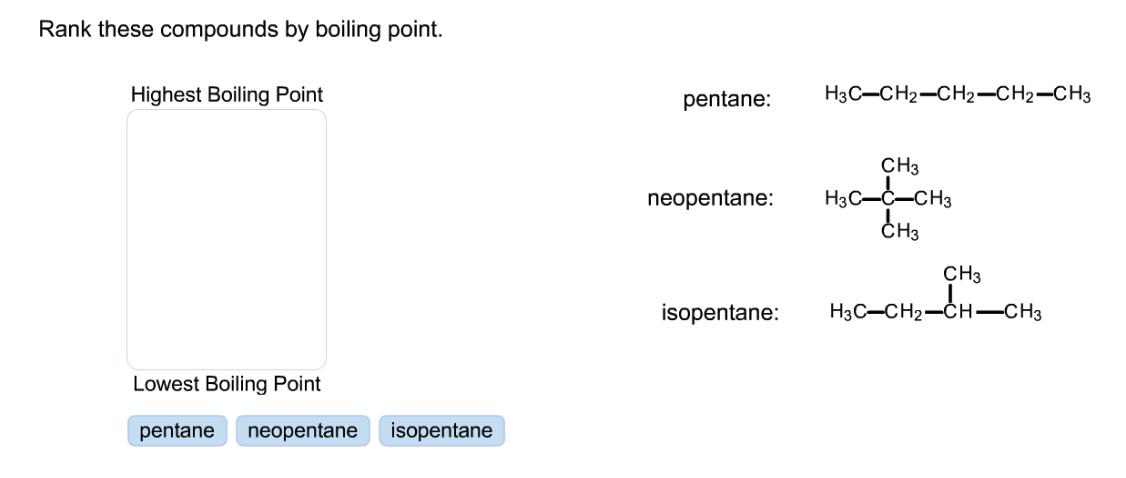
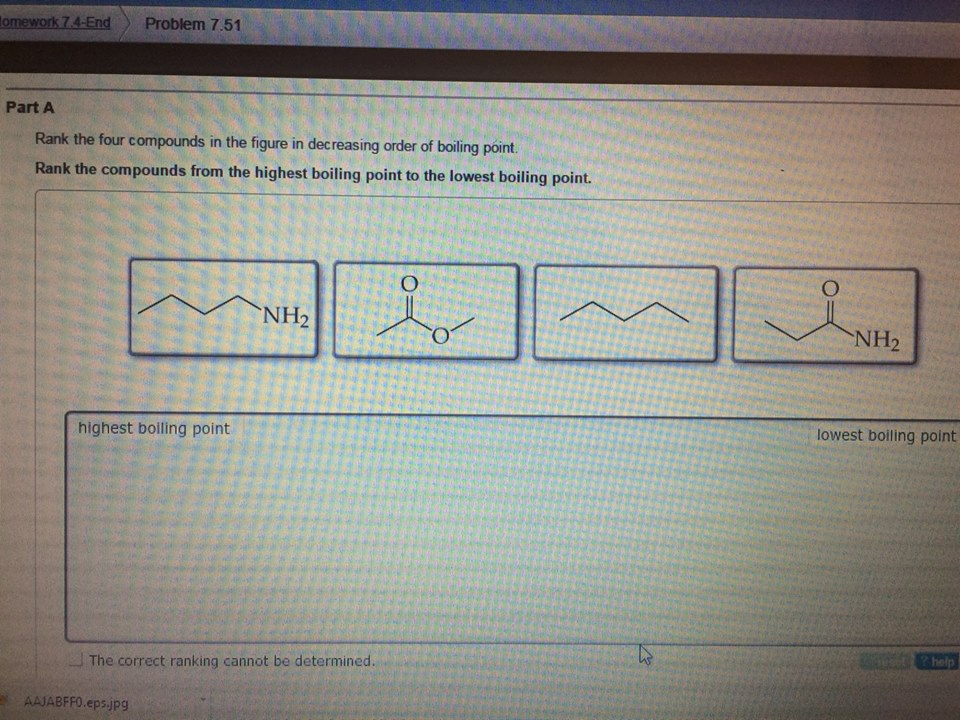
Rank The Shown Compounds By Boiling Point
So, imagine this: you've got a bunch of different substances, all chilling out, and you're wondering which one will be the last to pack its bags and turn into a gas. That's basically what "Rank The Shown Compounds By Boiling Point" is all about, and let me tell you, it's way more fun than it sounds!
Think of it like a friendly competition, a little science showdown. We're not talking about explosions or anything scary here. It's more like a gentle nudge, seeing how much heat each little molecule needs to get excited and float away as vapor.
It's like figuring out who's the most stubborn when it comes to changing their state!
What makes it so special is that it takes something that sounds super technical, like boiling points, and makes it totally accessible. You don't need a chemistry degree to get hooked. It's all about understanding how different things are built and how that affects their behavior.
Must Read
For example, you might have water, which we all know boils at a nice, predictable 100 degrees Celsius. But then you have other stuff, like maybe something called methane. And that's where the intrigue begins!
The "shown compounds" are the stars of this particular show. They're the contestants, lined up and ready for their moment in the heat. Each one has its own unique story, its own molecular makeup that dictates its boiling point.
You start looking at them, and you might notice patterns. Some are big and bulky, while others are sleek and small. Some have little sticky bits that like to hold onto each other, and others are more independent.
And that's the secret sauce, isn't it? It's about seeing the invisible forces at play. Those intermolecular forces are like the tiny hands that molecules use to hold onto their neighbors. The stronger those hands, the more heat you need to pry them apart.
So, when you're asked to rank them, you're essentially guessing which ones have the strongest grips. It's a puzzle, a delightful little brain teaser that you can solve with a bit of observation and a dash of scientific intuition.
The beauty of it is in its simplicity and its depth. On the surface, it's just about ordering things. But beneath that, it’s a window into the fundamental rules that govern the physical world around us.
Imagine you're at a party, and each compound is a guest. Some guests are super social and love to hug everyone (strong intermolecular forces). Others are more reserved and keep to themselves (weak intermolecular forces).

The "ranking" is like figuring out who needs the biggest introduction or the most encouragement to get up and dance (boil). The ones who are already mingling easily will need less of a push.
It's incredibly engaging because it taps into our natural curiosity about how things work. We see boiling water every day, but we don't always think about why it boils at that specific temperature.
This challenge invites you to ask those questions. It's like being a detective, examining clues – the structure of the molecules, the types of bonds – to solve the mystery of their boiling points.
And the best part? You don't need fancy equipment. You can do it in your head, or with a simple list. It’s a mental workout that’s surprisingly satisfying.
Let's say you’re shown ethanol and diethyl ether. You might know that ethanol has that little -OH group, which is like a super-glue for molecules. Diethyl ether, on the other hand, is a bit more spread out.
So, intuitively, you might guess that ethanol will have a higher boiling point because those -OH groups are going to be really clingy. And you'd be right!
It’s this kind of intuitive leap, this connecting of the dots, that makes the whole experience so rewarding. You start to see the hidden logic of chemistry in action.
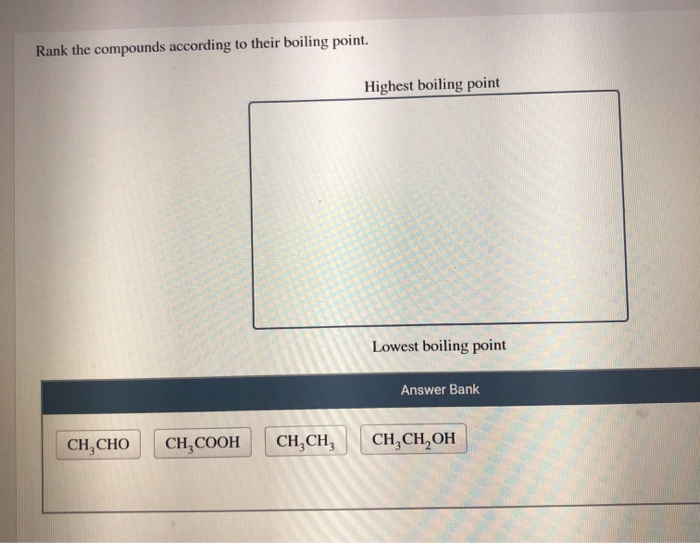
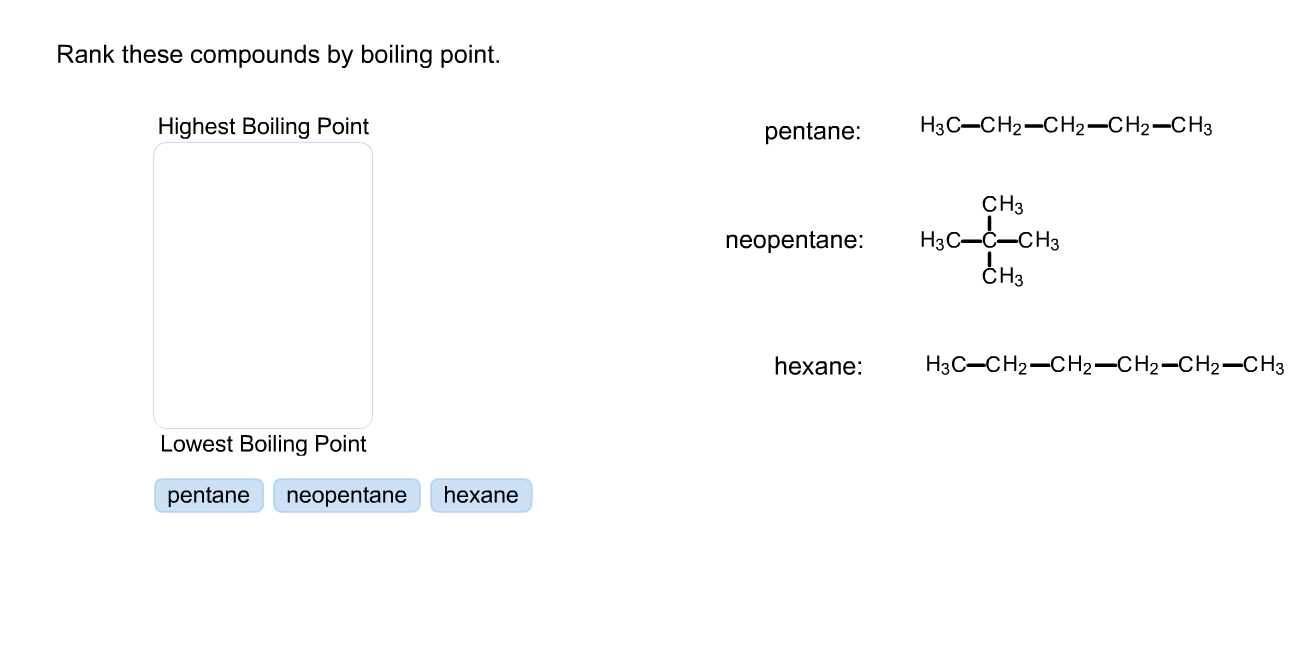
The compounds themselves are often interesting. They're not just abstract names; they're the building blocks of the world. You might encounter things that are familiar, like alcohol (ethanol), or things that are part of everyday life in unexpected ways.
The "shown" aspect is crucial too. It means you're given a specific set to focus on. It narrows down the field and makes the challenge manageable and fun.
Think of it as a curated collection of chemical curiosities. Each one is a little experiment waiting to be understood, a puzzle piece in the grander scheme of molecular behavior.
What makes it particularly entertaining is the element of surprise. You might think one compound will be hotter than another, only to discover it's the other way around. That’s when you learn something new!
It's a gentle way to encounter scientific principles without feeling overwhelmed. The ranking is the playful goal, but the real win is the understanding you gain along the way.
You start to develop a feel for it. You look at a molecule, and you can almost sense its boiling point. It's like developing a superpower, a little chemistry intuition.
The language used is usually straightforward, avoiding jargon where possible. It’s designed to be inviting, to make you feel like you’re part of the discovery process.

It’s like a friendly challenge from a knowledgeable friend who wants to share a cool science secret with you.
The visual aspect of seeing the compounds can also be helpful. Sometimes, seeing the structures helps solidify the understanding of why one behaves differently from another.
It’s not just about memorizing numbers; it’s about understanding the why behind those numbers. And that’s where the real magic happens.
You might be presented with a list, or even pictures of the molecules. Then, you're tasked with putting them in order, from the one that boils at the lowest temperature to the one that needs the most heat.
It's a bit like a race, where the finish line is boiling. The slowest starter, the one that takes the longest to reach the boiling point, is at the top of the ranking.
And the compound that boils first, the one that’s quickest to vaporize, will be at the bottom of the list.
This whole exercise is designed to be approachable and even exciting. It demystifies chemistry, making it a subject that anyone can engage with and enjoy.
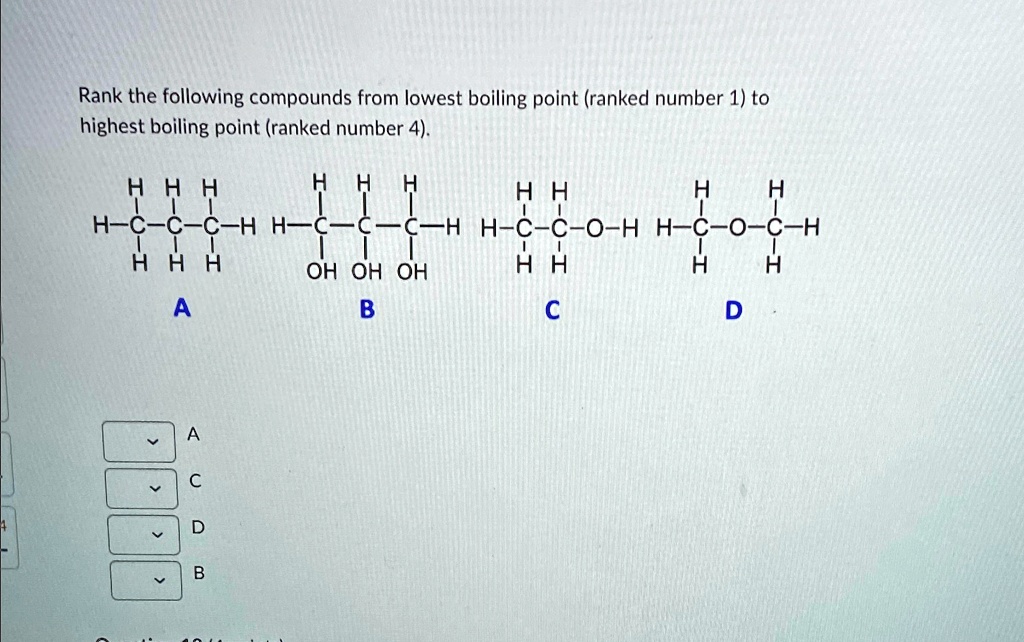
It encourages critical thinking and problem-solving skills in a fun and low-stakes environment. You’re not being tested; you’re being invited to explore.
The compounds you’re shown could be anything from simple gases to more complex liquids. The variety keeps things interesting and exposes you to different molecular structures.
For instance, you might see something like hydrogen, which is incredibly light and has very weak intermolecular forces. Then, you might see something like glycerol, which is a big, sticky molecule with lots of opportunities to form strong bonds.
The difference in their boiling points would be huge, and understanding why is the rewarding part.
It’s this sense of discovery, of peeling back the layers of how matter behaves, that makes "Rank The Shown Compounds By Boiling Point" so special and engaging.
It’s a gentle invitation to play with science, to tinker with knowledge, and to have a little fun while you’re at it.
So, if you ever stumble upon a challenge like this, don't be intimidated. Embrace it! It’s a fantastic opportunity to learn something new and to appreciate the subtle, yet powerful, forces that shape our world.
You might just find yourself surprisingly captivated by the boiling point adventures of these chemical characters!



![SOLVED: 14. [1 pts] Draw - the Lewis structure for each of the](https://cdn.numerade.com/previews/8c543499-45cf-4441-b080-491ebcd1e425_large.jpg)