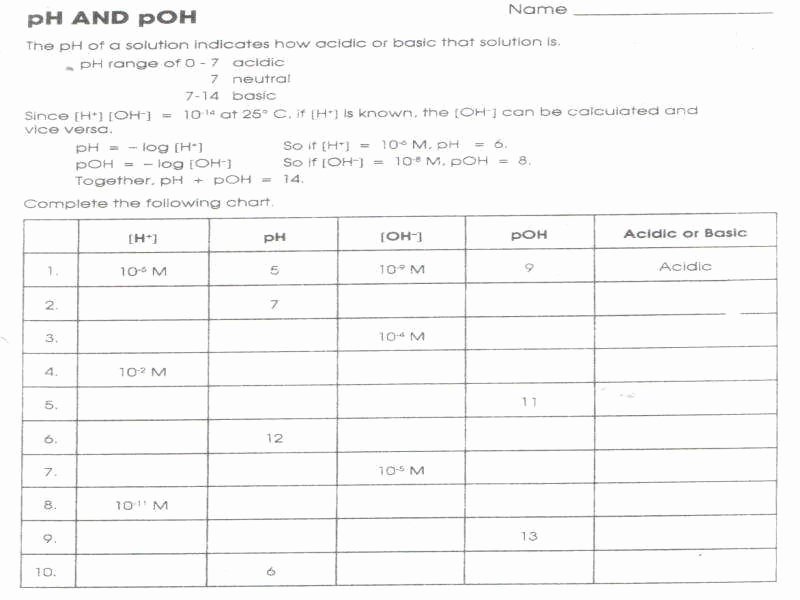
Ph And Poh Calculations Worksheet Answers

Hey there, fellow science enthusiast! Or maybe you're just trying to survive this chemistry class, eh? No judgment here. We've all been there, staring at a page of numbers and symbols, wondering if our brain is actually melting. Today, we're diving headfirst into the wonderfully weird world of pH and pOH calculations. And guess what? We're going to talk about the answers to those worksheets that have probably been giving you a bit of a headache.
So, grab your favorite mug, maybe a cookie (you deserve it!), and let's chat. Think of this as your friendly, no-stress guide to conquering those pH and pOH problems. We're not going to get bogged down in super-fancy jargon, just the good ol' practical stuff. You know, the stuff that helps you actually understand what’s going on, rather than just memorizing formulas until your eyes water.
First off, what even is pH and pOH? It's all about how acidic or basic something is. Like, is your coffee a little too tart, or is your soap, well, really soapy? That's the pH at play. And pOH is like its slightly less famous, but equally important, cousin. They're basically two sides of the same coin, telling us about the concentration of those little hydrogen (H+) and hydroxide (OH-) ions floating around in a solution.
Must Read
You might have seen a worksheet with a bunch of numbers and been asked to calculate the pH or pOH. And maybe you've even found the answer key, but it just feels like a magic trick, right? Poof! There's the answer. But where did it come from? That's what we're going to unpack today. We're going to demystify those answers and make you feel like a total chemistry whiz. Or at least, a slightly less confused one!
Let's start with the basics, because you can't build a house without a solid foundation, and you can't calculate pH without knowing the foundations of the pH scale. Remember that scale? It goes from 0 to 14. Super simple, right? Below 7 is acidic. Think lemon juice, vinegar – that sort of thing. Above 7 is basic, or alkaline. Think baking soda, drain cleaner (handle with care!). And right in the middle, at 7? That's neutral. Like pure water. So, when you see a pH of, say, 2.3, you instantly know, "Okay, that's pretty acidic!" See? You're already doing it.
Now, the real magic, or the calculation magic, comes into play when you're given information and need to find the pH or pOH. The most common scenario is when you're given the concentration of hydrogen ions ([H+]). This is usually expressed in moles per liter, or Molarity (M). So, if your worksheet says something like, "Calculate the pH of a solution with [H+] = 0.001 M," what do you do?
This is where the trusty formula comes in: pH = -log[H+]. Don't let the "log" scare you! It's just a mathematical function. On your calculator, it's usually labeled "log" or "log10." So, you take the concentration of H+ ions, punch it into your calculator after hitting the log button, and then hit the "negative" button. Easy peasy!
Let's try that 0.001 M example. So, you'd type in: -log(0.001). And what do you get? Drumroll please... you get 3! So, the pH is 3. See how that works? It's that simple. If your worksheet gave you the answer as pH = 3, now you know exactly how they got there. It’s like having the secret decoder ring!

What if the [H+] concentration is a bit more complex, like 1.5 x 10^-4 M? No sweat! You just do the same thing: pH = -log(1.5 x 10^-4). You'll need to use the scientific notation button on your calculator (usually labeled "EXP" or "EE"). So, you'd type in 1.5, then EXP, then -4, then hit the log button, and then the negative button. The answer you should get is approximately 3.82. Ta-da! The worksheet answer was probably 3.82, and now you're a pro at getting it.
But wait, there's more! Sometimes, instead of [H+], you're given the concentration of hydroxide ions ([OH-]). This is where pOH comes in. The formula is super similar: pOH = -log[OH-]. So, if a problem says, "Calculate the pOH of a solution with [OH-] = 0.01 M," you'd do pOH = -log(0.01). And guess what? That equals 2. So, the pOH is 2. Simple, right?
Now, here's where it gets really interesting, and where you might have seen some answers that involved both pH and pOH. There's a super handy relationship between them: pH + pOH = 14. This is like the golden rule of pH and pOH. It's always true for any aqueous solution at 25°C (which is usually the standard for these kinds of problems, so don't worry too much about the temperature unless they specify otherwise!).
So, let's say you calculated the pOH to be 2, like in our last example. And your worksheet also asks for the pH. You can just use that handy formula! pH = 14 - pOH. So, pH = 14 - 2, which means pH = 12. See? You can find one if you know the other. It's like a chemistry superpower!
What if you were given the [OH-] concentration and asked to find the pH directly? For example, if [OH-] = 5.0 x 10^-6 M. First, you'd calculate the pOH: pOH = -log(5.0 x 10^-6). That gives you a pOH of approximately 5.30.

Then, you use the golden rule: pH = 14 - pOH. So, pH = 14 - 5.30, which means pH = 8.70. If the answer on your worksheet was 8.70, you've just unlocked the mystery! It’s not just a number; it’s the result of these simple, elegant calculations.
Now, what about the other way around? What if you're given the pH and asked to find the [H+] concentration? This is where you use the inverse of the pH formula. Remember pH = -log[H+]? To get [H+], you need to undo the logarithm and the negative sign. So, you use the antilog function, which is usually 10 raised to the power of something. The formula is: [H+] = 10^-pH.
Let's say your worksheet gives you a pH of 4. You'd calculate [H+] = 10^-4 M. Your calculator will probably show this as 0.0001 M. If you're asked for scientific notation, it's 1 x 10^-4 M. Again, you've cracked the code!
And the same applies if you're given the pOH and asked for the [OH-] concentration. The formula is: [OH-] = 10^-pOH. So, if your pOH is 9, then [OH-] = 10^-9 M. See the pattern? It’s all about these inverse relationships.
Sometimes, worksheets get a little tricky. They might give you the concentration of a strong acid or a strong base and ask for pH or pOH. For strong acids, like HCl or H2SO4, the concentration of the acid is essentially equal to the concentration of H+ ions. So, if you have 0.05 M HCl, your [H+] is also 0.05 M. Then you just plug that into pH = -log[H+].
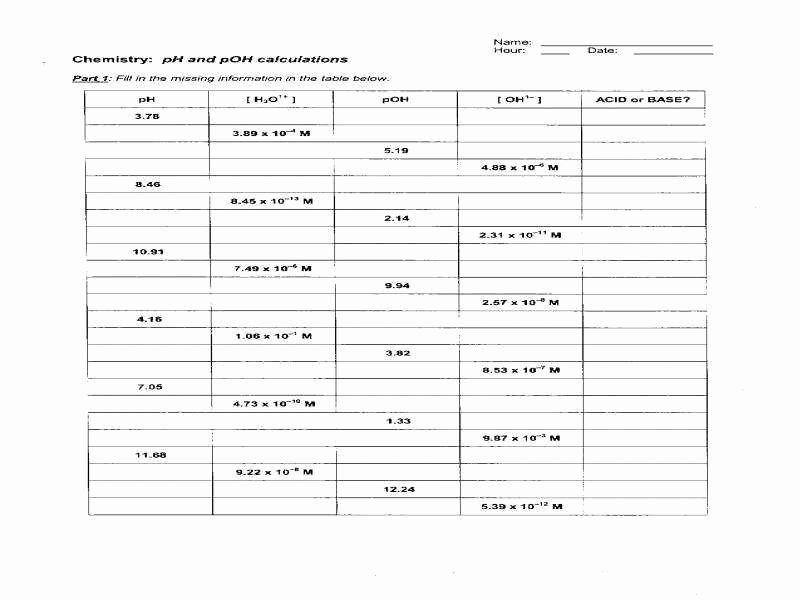
For strong bases, like NaOH or KOH, the concentration of the base is equal to the concentration of OH- ions. So, if you have 0.002 M NaOH, your [OH-] is 0.002 M. You can then calculate the pOH using pOH = -log[OH-], and then find the pH using pH + pOH = 14.
What if the problem involves a weak acid or a weak base? Whoa, hold up! That's a whole other level of complexity. Weak acids and bases don't fully dissociate, meaning they don't release all their ions. This involves something called an equilibrium constant (Ka or Kb), and it’s a bit more involved. If your worksheet had answers that involved weak acids or bases and you're just learning pH and pOH, it’s possible you might have been looking at answers for a more advanced topic. But for the typical introductory worksheets, it's usually strong acids and bases, or given ion concentrations.
Let's think about some common pitfalls that might lead to an incorrect answer on your worksheet. One of the biggest is forgetting the negative sign in the logarithm calculation. That little minus sign is crucial! If you forget it, you'll get a positive pH, which is almost always wrong unless you're dealing with extremely dilute solutions. Always double-check: pH = -log[H+]. That negative sign is your friend.
Another common mistake is using the wrong concentration. Make sure you're using the concentration of H+ for pH and the concentration of OH- for pOH. Don't mix them up! And remember that for strong acids/bases, the concentration of the acid/base is the concentration of the ions. Easy to forget, but important!
And then there's the issue of units. While pH and pOH are unitless, the concentrations of ions are almost always in Molarity (M). If you see something like "millimoles per liter" (mM), you'll need to convert it to Molarity by dividing by 1000. So, 5 mM is the same as 0.005 M. Small details, but they can make a big difference in your final answer!
Let's do a quick recap of the key formulas. You got this!
Key Formulas to Remember:
- pH = -log[H+]
- pOH = -log[OH-]
- pH + pOH = 14
- [H+] = 10^-pH
- [OH-] = 10^-pOH
See? Not so scary when you break it down. When you look at the answers on your worksheet, you can now trace them back to these formulas. That pH of 1.5? That came from a [H+] of 10^-1.5 M. That pOH of 10.2? That means the [OH-] was 10^-10.2 M. And you can probably figure out the corresponding pH too!
The beauty of these calculations is that they connect everything. You can go from [H+] to pH, then to pOH, then to [OH-], and back again. It’s a beautiful, interconnected system. And understanding these basic calculations is fundamental to so many other chemistry concepts.
So, the next time you see a pH or pOH worksheet, don't groan. Smile! You've got the tools. You've got the formulas. You've got the understanding. You can look at those answers and know exactly how they were derived. It's not magic; it's just good ol' chemistry. And you, my friend, are mastering it. Keep practicing, keep asking questions, and don't be afraid to play around with the numbers. You'll be a pH/pOH pro in no time!
And hey, if you ever need to check your work again, or you're just feeling a little rusty, come back to this. Think of this as your friendly chemistry coffee chat. We'll always be here to help you decipher those numbers. Now, go forth and conquer those calculations! You've earned that cookie, and maybe even a second cup of coffee. You're doing great!