Oxidizing Agents Can Convert Co Into Co2 .

So, picture this: I was messing around in the kitchen the other day, trying to whip up some fancy homemade s'mores. You know, the kind where you toast the marshmallow until it's perfectly golden brown, not a charcoal briquette in sight. Anyway, I got a little too enthusiastic with the propane torch (don't judge!), and suddenly, there was this tiny puff of white smoke and a smell... well, let's just say it wasn't the sweet aroma of toasted sugar. It was a bit metallic, if that makes any sense. Turns out, I might have, in my amateur pyromania, slightly altered the state of some… well, let’s just say some things in my environment. Little did I know, I was getting a front-row seat to a fundamental chemical principle: oxidation!
Now, I’m no chemist, and if you’re picturing me in a pristine lab coat with bubbling beakers, you can stop that right now. My "lab" is usually a slightly cluttered desk, and my "experiments" often involve trying to figure out why my Wi-Fi is being a drama queen. But even us laypeople can grasp some pretty cool science concepts. And today, we’re diving into something that sounds a bit dramatic, but is actually happening all around us, all the time: how oxidizing agents can turn CO into CO2. Catchy title, right? (Okay, I admit, I’m working on the titles.)
Let's break down the villain of our story: CO. That’s carbon monoxide. Now, this stuff is a bit of a bad rap. And for good reason, honestly. It’s invisible, odorless, and can be seriously dangerous. Think of it as the ninja of toxic gases. It’s the byproduct of incomplete combustion – basically, when something burns, but there’s not enough oxygen around to make it burn completely. My overzealous marshmallow-toasting incident? Yeah, that’s a prime example of incomplete combustion. (Seriously, gotta work on that torch control.)
Must Read
So, what’s the deal with CO? Well, chemically speaking, a carbon atom is happily bonded to just one oxygen atom. Think of it as a cozy little diatomic molecule, feeling pretty content. But as we know, in the grand scheme of chemical reactions, contentment is often temporary. And in the case of CO, its relative stability is precisely what makes it so problematic when it gets into our bodies. It’s sneaky. It can bind to hemoglobin in our blood far more effectively than oxygen, effectively suffocating us from the inside out. Shivers. Not ideal, right?
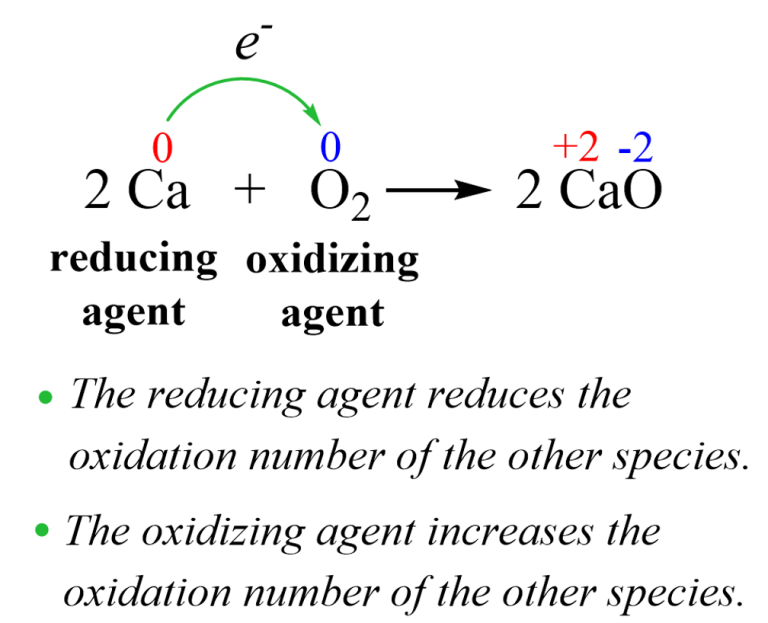
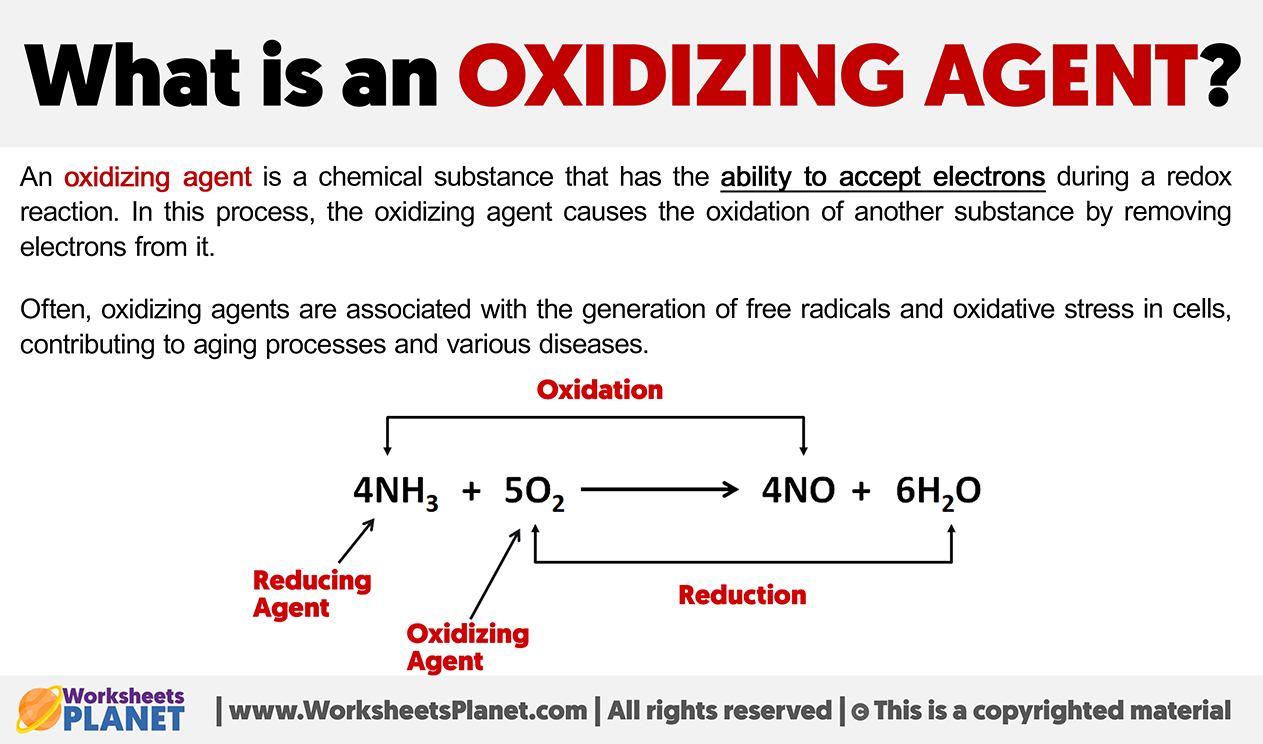
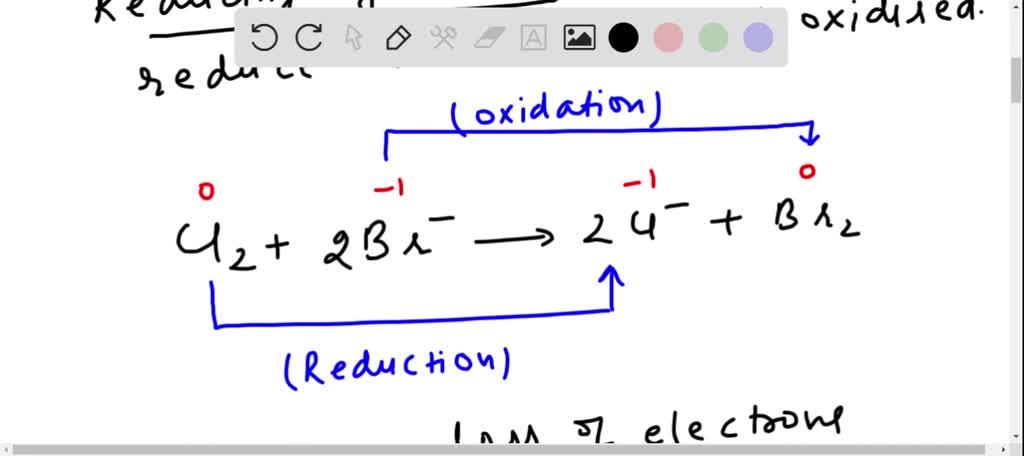
Now, enter the hero (or sometimes the instigator, depending on your perspective): the oxidizing agent. What even is that? Imagine a chemical that’s really, really good at taking electrons from other substances. It’s like a chemical vacuum cleaner for electrons. When an oxidizing agent encounters something, it essentially steals electrons from it, causing that other substance to become oxidized. It’s a bit like how a really popular influencer might “take” all the attention from everyone else at a party. You get what I mean?

And here’s the kicker: when something gets oxidized, it’s essentially losing electrons. In the case of our friend CO, when it encounters a suitable oxidizing agent, it can be persuaded to give up some of its electron density. This might not sound like a big deal, but it has rather significant consequences for the CO molecule.
The most common and, frankly, the most important oxidizing agent we’re dealing with here is, you guessed it, oxygen (O2) itself. Yes, the very thing we need to breathe is also the stuff that can convert our stealthy CO into something a little less sinister. It’s like nature’s own cleanup crew! When there’s plenty of oxygen around, CO has a much better chance of reacting with it to form… you guessed it again… CO2, or carbon dioxide.
Now, CO2. That's a different beast altogether. It's the stuff we exhale, it's the fizz in your soda, and, yes, it's a greenhouse gas. But compared to CO, it's relatively harmless to us directly. A carbon atom in CO2 is happily bonded to two oxygen atoms. It’s like the CO molecule decided to upgrade its social life and found two more friends. This extra oxygen atom makes the molecule more stable and, crucially, less likely to interfere with our precious hemoglobin.

So, the reaction looks something like this, in a very simplified way: 2CO + O2 → 2CO2. See? Two molecules of carbon monoxide, plus one molecule of oxygen, come together to form two molecules of carbon dioxide. It’s a chemical marriage, where the extra oxygen atom from the O2 molecule essentially bridges the gap and forms a new bond with the carbon atom in each CO molecule. Pretty neat, huh?
This is why good ventilation is so crucial, especially in situations where incomplete combustion might be happening, like near car exhaust fumes or poorly maintained furnaces. When there’s ample oxygen flowing, the CO that’s produced has a much higher chance of being converted into CO2 before it can do any harm. It’s like opening all the windows during a smoky barbecue – letting in fresh air to dilute and, in a chemical sense, transform the nastier stuff.
But it's not just oxygen, is it? Oxidizing agents are a whole category of chemicals. You have things like halogens (think chlorine, bromine), peroxides (like hydrogen peroxide, which you probably have in your bathroom cabinet for cuts), and even certain metal oxides. These guys are all electron-snatchers extraordinaire. And while oxygen is the most common player in atmospheric CO oxidation, other oxidizing agents can be used in industrial processes or specific chemical reactions to achieve the same transformation.
For instance, in some catalytic converters in cars, there are precious metals like platinum and palladium. These metals act as catalysts, which means they speed up chemical reactions without being used up themselves. They are brilliant at helping CO react with oxygen to form CO2, effectively cleaning up the exhaust. So, next time you see a car driving by, you can think of that little box under the car as a miniature chemical powerhouse, diligently working to turn CO into CO2. It’s a silent guardian of our air quality. (Though, let's be honest, some exhaust fumes still smell pretty questionable.)
The concept of oxidation isn't just limited to gases, though. Think about rust! When iron rusts, it’s reacting with oxygen in the air and water. The iron is getting oxidized, forming iron oxides – basically, rust. Or what about when you cut an apple, and the flesh turns brown? That’s oxidation too! Enzymes in the apple react with oxygen in the air, changing the molecules and causing that browning effect. It’s oxidation in action, making things look… less appealing, in the case of the apple. But hey, at least it’s not CO!
What’s really interesting is the idea of controlling these reactions. Scientists are constantly looking for more efficient ways to convert harmful pollutants like CO into less harmful substances. It’s all about understanding the nuances of electron transfer and finding the right oxidizing agents and conditions to make these transformations happen effectively. It’s like being a chemical matchmaker, trying to get the right molecules together for a happy, non-toxic union.

And it’s not just about cleaning up pollution. Oxidation plays a role in so many things. It’s fundamental to how our bodies produce energy (cellular respiration is essentially a controlled oxidation process). It’s how we generate electricity in batteries. It’s even involved in how some medicines work!
So, the next time you hear about "oxidation," don't just think of your grandma's silver tarnishing. Think about the complex dance of electrons, the power of oxidizing agents, and the fascinating transformation of a silent killer like CO into the more manageable CO2. It’s a process happening all around us, silently shaping our world. And who knows, maybe one day, my marshmallow-toasting skills will improve, and I'll have a better understanding of how to manage my own little "incomplete combustion" events. Until then, I'll stick to admiring the chemists who make the big stuff happen!
It’s a reminder that even seemingly simple chemical conversions can have huge implications. The subtle addition of an oxygen atom can change a gas from a deadly threat to a component of our atmosphere. It's a testament to the power and elegance of chemical reactions, and how they’re constantly at work, both in the grand scale of our planet and in the tiny details of our everyday lives. Pretty cool, right? Makes you wonder what else is happening that we're not even aware of!

.webp)

