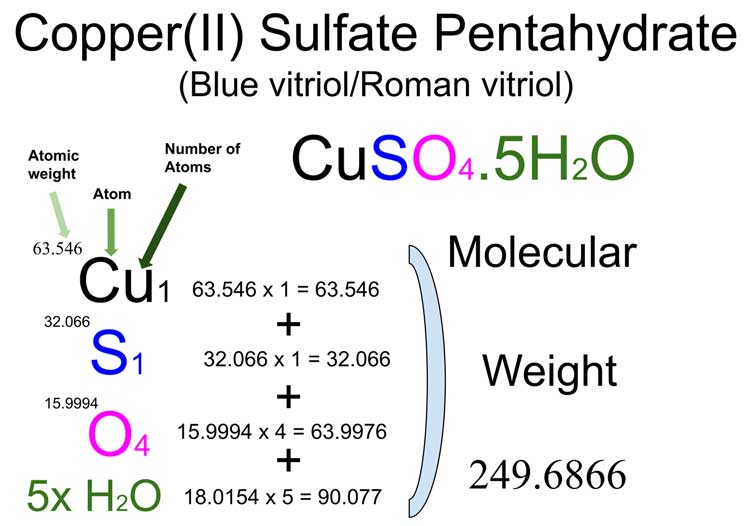
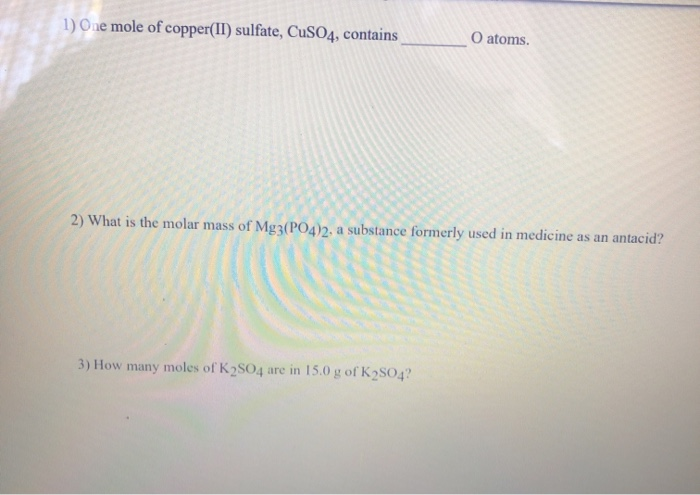
One Mole Of Copper Ii Sulfate Cuso4 Contains
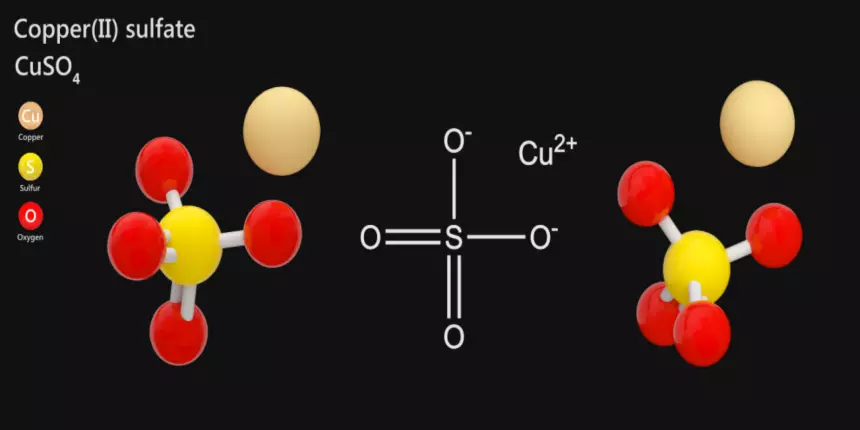
So, you wanna talk about copper(II) sulfate? Awesome choice! It's got this cool name, right? Copper(II) sulfate. Sounds a bit like a secret agent's codename. And honestly, it kind of is. This stuff is everywhere, and it does some seriously neat things. Think of it as a little chemical rockstar.
Today, we're diving into what happens when you grab a mole of this vibrant blue powder. Don't let the word "mole" scare you. It's not about furry little diggers. In chemistry, a mole is just a super-duper huge number. Like, ridiculously huge. It's 6.022 x 1023 individual particles. Imagine that many grains of sand. Now imagine that many copper(II) sulfate molecules. Mind-blowing, right?
So, What's In One Mole of CuSO4?
Alright, let's break it down. When you have one mole of copper(II) sulfate (that's CuSO4 for the shorthand lovers), you've got yourself a whole lot of atoms doing their thing.
Must Read
First off, you’ve got copper atoms. Loads of them. 6.022 x 1023 copper atoms, to be exact. These are the guys that give copper(II) sulfate its distinctive blue hue. Seriously, this is why those beautiful blue crystals sparkle. It’s all about the copper!
Then, you've got sulfur atoms. Another 6.022 x 1023 of them. Sulfur is a pretty important element, often associated with a… well, let’s just say a pungent smell in some of its forms. But in CuSO4, it’s playing nice.
And finally, the oxygen atoms. Oh boy, the oxygen! You get a whopping four times the number of copper or sulfur atoms. So that’s 4 x 6.022 x 1023 oxygen atoms. That’s a whole lot of O’s! Think of them as the busy bees, always ready to bond and make things happen.

The Amazing Blue Beauty
Let’s talk about that gorgeous blue color. It’s not just pretty; it’s a clue! That vibrant azure is thanks to how the copper ions interact with light. When copper(II) sulfate is in its solid crystalline form, or dissolved in water, it absorbs certain wavelengths of light and reflects others. The ones it reflects are mostly in the blue and violet range, which is what our eyes perceive. It’s like a tiny, natural light show happening in every crystal!
And here's a quirky fact: if you heat up copper(II) sulfate crystals, they can actually change color! They lose their water molecules and turn into a whitish-grey powder. It's like a magical color transformation. But add a bit of water back, and POOF! Blue again. It’s like a mood ring, but for a chemical compound. Talk about dramatic!
Why This Blue Stuff is Actually Super Useful
So, we’ve got this big ol' pile of atoms, all nice and blue. What’s the big deal? Well, copper(II) sulfate is way more than just a pretty face. It’s a workhorse!
For starters, it's a fantastic fungicide and algaecide. Ever seen those signs around ponds or pools telling you not to swim for a bit after treatment? Yep, that’s probably copper(II) sulfate at play. It’s tough on unwanted growths. Farmers use it to protect crops, and gardeners use it for their precious plants. It’s like a superhero for plants, zapping away the bad guys.
It’s also used in making pigments. That bright, cheerful blue you might see in some paints or inks? Sometimes, that’s thanks to copper(II) sulfate. Imagine your favorite blue crayon – there’s a good chance copper had a hand in its creation!
And get this, it’s used in electroplating. Ever seen something shiny and metallic that isn't actually made of pure gold or silver? Copper(II) sulfate helps lay down a thin layer of copper onto other metals, giving them a cool finish or preparing them for further coatings. It’s like giving everyday objects a fancy metallic makeover.
A Touch of History and Mystery
People have been messing with copper compounds for ages. Long before we knew about moles or atoms, ancient civilizations were using copper for all sorts of things. The vibrant blue of copper compounds would have definitely caught their eye. It’s kind of cool to think that we're still using and appreciating this element for its properties, just with a lot more scientific understanding now.
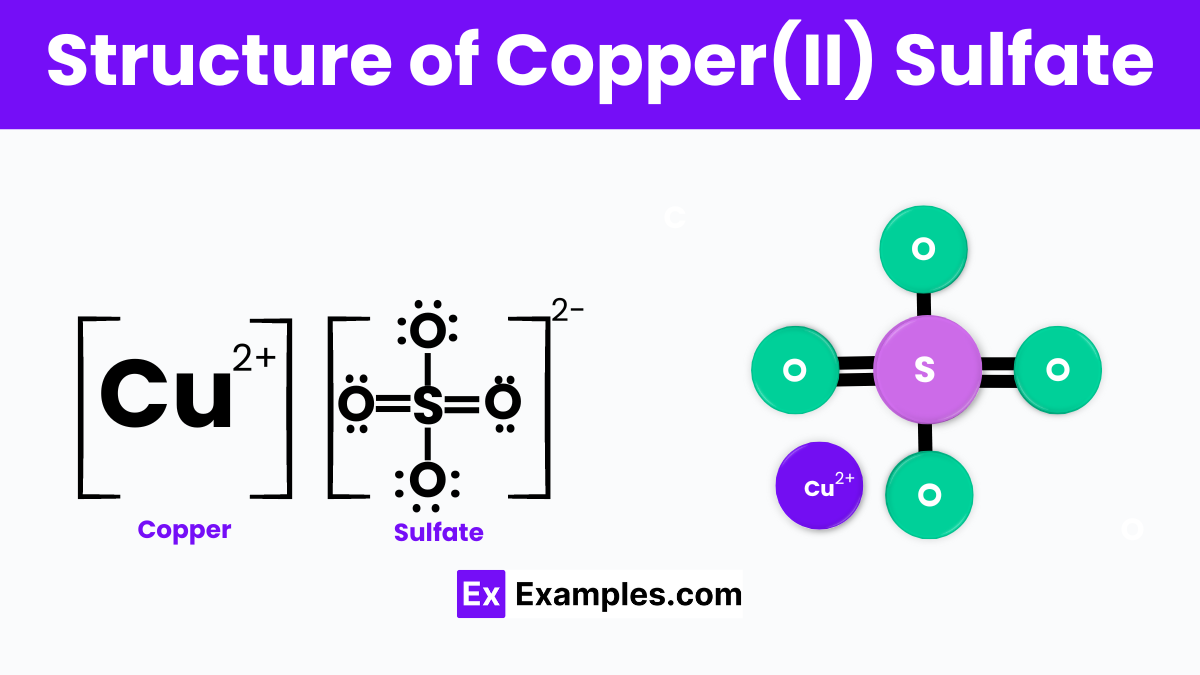
There's a historical anecdote about copper(II) sulfate being used as an antidote for certain poisons. While it’s not recommended to go around self-medicating with chemicals, it highlights how people observed and experimented with substances, trying to unlock their secrets.
The Mole: The Ultimate Chemical Party Planner
Back to our mole. This isn't just a static pile of atoms. It’s a potential party waiting to happen! When you dissolve CuSO4 in water, those copper ions and sulfate ions get separated and go swimming around. They’re free to interact with other things, which is where all its useful properties come from.
Think of the mole as the key to understanding how much of this stuff you actually have. If you need to, say, kill algae in a pool, you don't just throw in a pinch. You calculate how many moles you need. It’s all about precision, and the mole is your trusty measuring stick.
And if you ever get the chance to see large, perfect crystals of copper(II) sulfate, grab it! They are absolutely stunning. They form beautiful, geometric shapes, and the color is just mesmerizing. It’s a little piece of chemistry you can hold in your hand (carefully, of course!).
A Little Bit of Caution (Because, You Know, Science)
Now, while copper(II) sulfate is super cool, it's also a chemical. So, it’s important to be a little careful. Don’t go munching on it or playing with it without adult supervision. It can be irritating to skin and eyes, and ingesting it isn't a good idea. Safety first, always!
But don't let that dampen your enthusiasm! The fact that a simple compound like CuSO4 has such a vibrant color, such a wide range of uses, and a history of being admired and utilized makes it incredibly fascinating. It’s a reminder that the world around us, even the stuff in a little blue crystal, is full of wonder and scientific marvels.
So next time you see that bright blue copper(II) sulfate, remember the mole. Remember the billions upon billions of copper, sulfur, and oxygen atoms all working together. It’s a tiny, dazzling testament to the amazing world of chemistry. Pretty neat, huh?