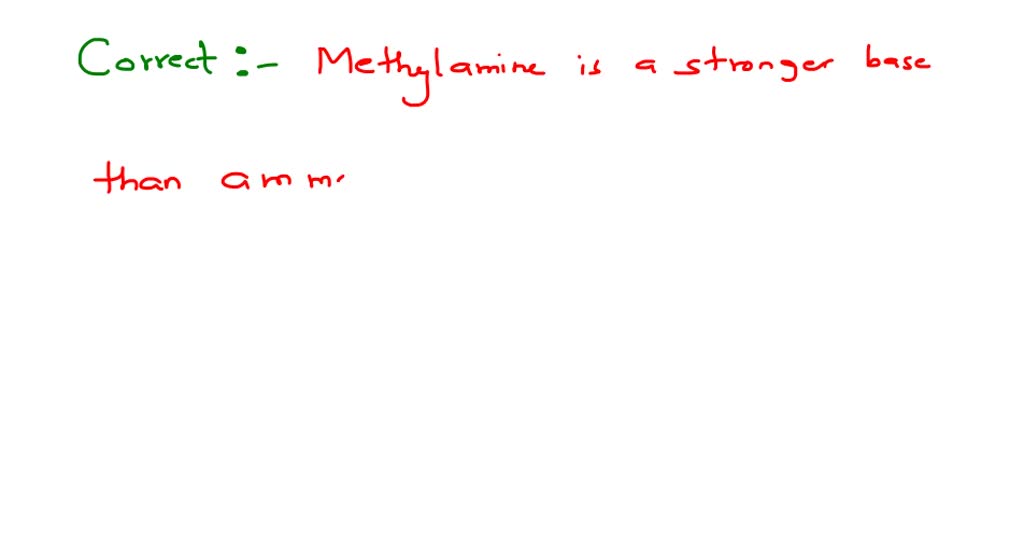Methylamine Is Stronger Base Than Ammonia

Hey there, science explorers! Ever wonder about the invisible world of molecules and how they interact? It’s a pretty fascinating place, full of tiny building blocks that make up… well, everything. Today, we’re going to peek at a couple of these little guys, specifically ammonia and its slightly more robust cousin, methylamine. You see, there’s this whole concept of bases in chemistry, and it turns out, methylamine is a bit of a rockstar when it comes to being a base compared to good ol’ ammonia. Intriguing, right?
So, what exactly is a base in this context? Think of it like a chemical greeter. Bases are really good at donating or, more accurately in many cases, accepting a specific little particle called a proton. Protons are basically just a positive charge floating around. When a molecule can easily grab onto one of these protons, we call it a base. The easier it is for them to snag a proton, the stronger that base is. Pretty straightforward, huh?
Now, let’s bring out our contenders: ammonia (NH₃) and methylamine (CH₃NH₂). Ammonia is a pretty common molecule. It’s what gives some cleaning products that… distinct smell. We’ve all probably encountered it. It’s got a nitrogen atom in the middle, all huggled up with three hydrogen atoms. This nitrogen atom has a lone pair of electrons, a little bit of extra "stuff" that’s key to its basic nature. It’s like a little magnet, ready to attract a proton.
Must Read
Methylamine, on the other hand, is like ammonia’s slightly fancier sibling. It’s got the same basic structure – a nitrogen atom with three attachments – but one of those attachments isn't a hydrogen atom. Instead, it’s a methyl group. What’s a methyl group? It’s just a carbon atom with three hydrogen atoms attached to it (CH₃). So, instead of NH₃, we have CH₃NH₂. It’s like taking one of the hydrogens off ammonia and swapping it out for a tiny carbon chain.
And here’s where the cool part comes in: this tiny swap makes methylamine a stronger base than ammonia. How can such a small change have such a big effect? It’s all about how willing that nitrogen atom is to grab onto a proton. Think of it like this: Imagine you’re trying to pick up a little bouncy ball. Ammonia is pretty good at it, but methylamine has a little something extra that makes it even better.

The methyl group (CH₃) attached to the nitrogen in methylamine is what chemists call an electron-donating group. These groups are like little cheerleaders for the electrons. They gently push more electron density towards the nitrogen atom. Remember that lone pair of electrons on the nitrogen? This electron-donating effect makes that lone pair even more available, even more eager to snatch up a proton. It’s like giving the bouncy ball grabber a little extra grip!
So, when ammonia encounters a proton, it’s like, "Okay, I can take that." But when methylamine encounters a proton, it’s like, "Oh, yes please! This is even easier!" This means that in the same chemical environment, methylamine will grab onto more protons than ammonia will. It’s more efficient at its job as a base.
Let’s try a fun analogy. Imagine you have two friends, Alex (ammonia) and Ben (methylamine), who are both really good at collecting stray Lego bricks (protons). Alex is pretty decent at scooping them up. But Ben has a special, slightly larger scoop (the methyl group’s electron-donating effect) that makes it even easier and faster for him to grab all the Lego bricks scattered around. If there’s a pile of Lego bricks, Ben will end up with more than Alex in the same amount of time because his scoop is just a bit better suited for the job.
This difference in basicity might seem small in the grand scheme of things, but it has real implications in chemistry. For instance, in chemical reactions, the strength of a base can determine how quickly and efficiently a reaction proceeds, or even if it proceeds at all. Sometimes, you need a milder base like ammonia, and other times, you need the extra punch of a stronger base like methylamine to get the job done.
It’s also why these molecules behave differently in solutions. When dissolved in water, methylamine will produce a higher concentration of hydroxide ions (OH⁻) compared to ammonia. Hydroxide ions are the hallmark of basic solutions. Think of it like a party: ammonia might let in a steady stream of guests, but methylamine throws open the doors and lets in a bit of a crowd, making the party (the solution) more "basic."
The nitrogen atom in both molecules is the key player, with its lone pair of electrons acting as the proton magnet. But the methyl group’s subtle electron-pushing talent gives methylamine that extra edge. It’s a beautiful example of how small structural changes in molecules can lead to significant differences in their properties and reactivity. It's like adding a tiny spoiler to a car – it might not look like much, but it can change how it handles on the road!
So, next time you hear about ammonia or methylamine, you can impress your friends (or just yourself!) with the knowledge that methylamine is a bit of a superhero when it comes to being a base. It’s all thanks to that little methyl group, making its nitrogen atom just a tad more enthusiastic about grabbing protons. Pretty cool, right? The molecular world is full of these subtle yet powerful differences, and exploring them is half the fun!