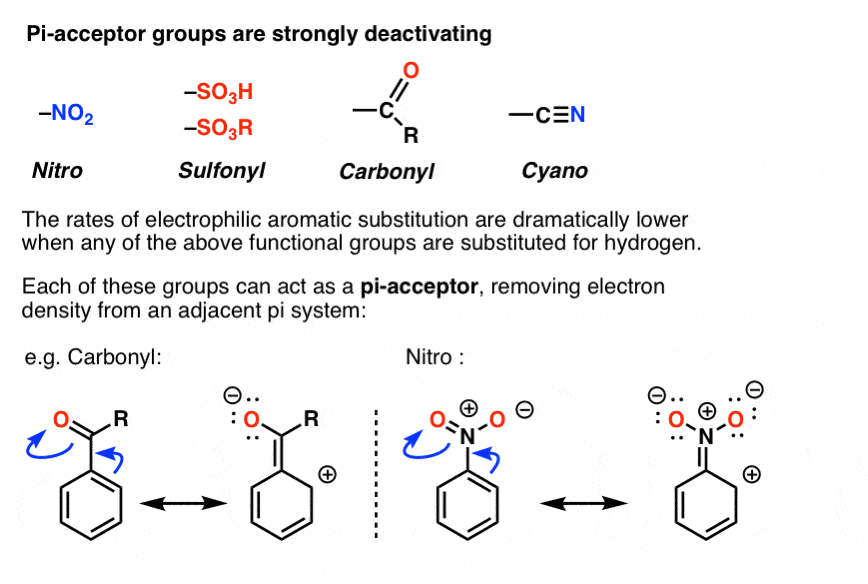
Is An Ester An Electron Withdrawing Group

Alright folks, gather ‘round! Let’s talk about something that sounds a bit fancy, a bit science-y, but I promise, it’s more like that quirky neighbor who always has a story to tell. We’re diving into the world of esters and whether they’re the type of group that likes to hog all the electrons. Think of it like this: have you ever had a friend who’s always borrowing your charger? Or maybe someone who’s just naturally a bit of a drama magnet? That’s kind of what we’re exploring today, but with molecules.
Now, before you start picturing tiny robots in lab coats arguing over a power outlet, let’s break it down. In the grand old scheme of chemistry, groups attached to a central molecule can have different personalities. Some are chill, some are energetic, and some, well, they’re like that one person who always seems to be subtly influencing the conversation. Esters are in this latter category, and the big question is: are they electron-withdrawing? Let’s find out!
The Big Kahuna: What Even IS an Ester?
First things first, what’s an ester? Imagine a molecule that’s like a neat little combination lock. It’s got a special arrangement of atoms: a carbon atom double-bonded to an oxygen atom, and also single-bonded to another oxygen atom. This second oxygen atom is then attached to another carbon-containing group. So, you’ve got your C=O, your C-O, and then your R' group hanging off the end. Pretty snazzy, right?
Must Read
Think of your favorite fruity smell, like a strawberry or a banana. Chances are, you’re smelling an ester! They’re the unsung heroes behind a lot of the delicious aromas and flavors in our world. They’re in perfumes, in food flavorings, and even in some plastics. They’re basically the flavor and fragrance folks of the molecular universe. So, when we talk about esters, we’re talking about things that often make life smell and taste a little bit better. Pretty cool gig, if you ask me.
But here’s where it gets interesting. Like any good flavor or fragrance, esters can also influence the behavior of the molecules they’re part of. And that’s where the electron-withdrawing idea comes in. It’s not about them physically stealing your headphones, it’s about them influencing the electron clouds. A bit like how a charismatic speaker can subtly sway an audience, an ester can subtly influence how electrons behave in its vicinity.
Electron Tug-of-War: The 'Withdrawing' Part
So, what does it mean for a group to be electron-withdrawing? Imagine a party where everyone’s got a slice of pizza. An electron-withdrawing group is like that one guest who, without even trying too hard, manages to get a few extra slices from other people’s plates. They don’t necessarily eat the extra pizza, but they’ve got this magnetic pull that makes others voluntarily (or maybe not so voluntarily!) share. They’re not starving, they just… collect.
In chemistry, these “pizza slices” are actually electrons, which are tiny, negatively charged particles that zip around atoms. They’re crucial for how molecules bond and interact. An electron-withdrawing group is an atom or a collection of atoms that has a strong enough pull to draw electron density towards itself from the rest of the molecule. It’s like they’re saying, “Hey, mind if I borrow some of that electron-y goodness?”
This electron-pulling can have a big impact. It can make certain parts of the molecule more positive (because they’ve lost some of their negative electron charge) and can influence how easily other reactions can happen. It’s like changing the vibe of the party – suddenly, there’s a bit more tension, a bit more focus on the electron-hoarder.

The Ester's Subtle Influence
Now, back to our ester. Does it play the electron-withdrawing game? The short answer is, yes, generally it does. But it’s not always the most aggressive player in town. Think of it as a moderately popular kid at school. They can definitely get people to notice them and perhaps even influence their choices, but they’re not exactly the school bully.
The main culprit for the ester’s electron-withdrawing nature is that double-bonded oxygen (the C=O part). Oxygen is a super electronegative element. That’s a fancy way of saying it’s really, really good at attracting electrons. It’s like a vacuum cleaner for electrons. Because it’s double-bonded to the carbon, it has a strong grip on the electrons in that bond, pulling them closer to itself.
This also affects the carbon atom it’s attached to. That carbon atom, in turn, pulls electrons from other parts of the molecule. It's a bit of a chain reaction. Imagine a line of people holding hands, and the person at the end with the really strong grip starts pulling. The person next to them pulls a little, and so on down the line. The ester group, especially that C=O, is like that strong-handed person at the end.
The single-bonded oxygen in the ester also plays a role. Oxygen atoms, in general, are electron-lovers. So, having not one, but two oxygen atoms in the ester group gives it a definite electron-seeking tendency. It’s like having two slightly demanding friends who both want the best seat in the house. They’re going to make sure they get their way with the electron-sharing situation.
The Nuances: It's Not Always Black and White
But here’s where it gets a little more interesting, and perhaps a bit like trying to predict your teenager’s mood. The extent to which an ester acts as an electron-withdrawing group can depend on what else is attached to it. Think of it like this: if you’ve got that popular kid at school, their influence might be amplified if they’re also wearing the coolest sneakers. Similarly, if the groups attached to the ester are themselves electron-donating, they might counteract some of the ester’s pull. It’s a bit of a molecular balancing act.
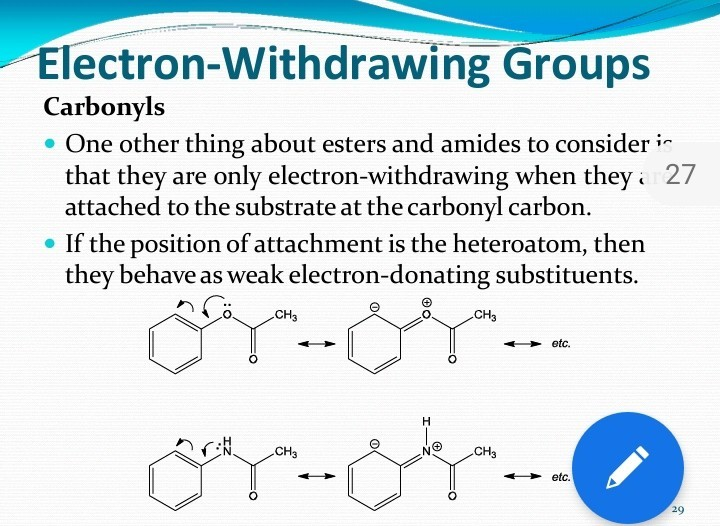
For example, if the R' group attached to the second oxygen is something that likes to give up electrons (an electron-donating group), it can somewhat lessen the overall electron-withdrawing power of the ester. It’s like the friend with the extra pizza slices giving a small slice back to someone else. There’s still a pull, but it’s not as dominant.
On the other hand, if the R' group is something that’s also electron-hungry, then the ester can become an even stronger electron-withdrawing group. It’s like that popular kid teaming up with their equally popular friend – their combined influence on the electron distribution is even greater. They’re a formidable duo, making sure the electron party is happening exactly where they want it.
Why Should We Care About Electron Sharing?
Okay, so why is this whole electron-pulling thing important? Well, it affects how molecules react. Think of reactions as tiny molecular dances. The way electrons are distributed dictates the rhythm and steps of that dance.
When an ester is electron-withdrawing, it can make adjacent atoms more susceptible to attack by other molecules that are looking for electrons. Conversely, it can make parts of the molecule less likely to give up their electrons. It’s like a spotlight being shone on certain areas, making them more reactive or less reactive.
For instance, consider the carbonyl carbon (the C=O carbon) in an ester. Because it’s bonded to that electron-greedy oxygen, it develops a partial positive charge. This makes it a prime target for nucleophiles – molecules or parts of molecules that are electron-rich and looking for a positive spot to latch onto. This is why esters can undergo reactions like hydrolysis (breaking apart with water) or react with Grignard reagents (fancy chemical tools). The electron-withdrawing nature of the ester group is a key player in making these reactions happen.

It’s like having a designated dance floor. The electron-withdrawing ester essentially sets up the conditions, creating an area where certain types of molecular partners are more likely to meet and mingle. Without that influence, the dance might not happen, or it might happen in a completely different way.
Analogy Time: The Popular Kid at the Mall
Let’s try another analogy. Imagine an ester as that one student who always seems to be surrounded by a group of friends at the mall. They’re not necessarily the loudest, but people tend to gravitate towards them. They have a certain… presence.
The double-bonded oxygen is like their really cool outfit – it’s eye-catching and draws attention. The single-bonded oxygen is like their stylish accessory, adding to their overall appeal. Together, the ester group creates a center of influence.
Now, are they actively trying to take things from people? Not necessarily. They’re not walking around with a shopping bag, snatching hoodies. But their presence, their aura, might make other shoppers (other molecules) feel a little self-conscious about their own style (electron distribution) and subconsciously adjust their position or behavior. They might pull their own belongings (electrons) a little closer, or perhaps be more inclined to approach the popular group. It's a subtle influence, a redirection of attention and energy.
If the popular kid is wearing even more eye-catching trends (stronger electron-withdrawing groups elsewhere), their influence might be even more pronounced. If they’re hanging out with someone who’s a bit of a wallflower (electron-donating group), the overall magnetic pull might be a little less intense.
The Verdict: Yes, But With a Wink
So, to circle back to our original question: is an ester an electron-withdrawing group? The general consensus, the scientific nod of approval, is yes. The presence of those electronegative oxygen atoms, especially the double-bonded one, creates a significant pull on electron density.
However, it’s important to remember that chemistry, like life, is full of nuances. The exact strength of this electron-withdrawing effect can vary depending on the molecular neighborhood. It’s not always a dramatic, forceful snatching of electrons, but more of a persuasive, influential draw.
Think of it as an ester saying, with a charming smile, “Could you perhaps shift those electrons a little closer? It just feels right.” And often, the rest of the molecule, or the atoms around it, oblige. They’re not losing out entirely, they’re just redistributing their wealth, so to speak, under the subtle guidance of the ester.
So, the next time you enjoy a delightful fruity scent, or perhaps encounter an ester in a chemical reaction, remember its subtle power. It’s not just about the smell; it’s about the way it artfully influences the electron party, making the molecular world a little more interesting, a little more reactive, and a whole lot more fragrant.
And that, my friends, is the easy-going, slightly whimsical, but ultimately accurate take on whether esters are electron-withdrawing. They are indeed, and they do it with a certain molecular flair!