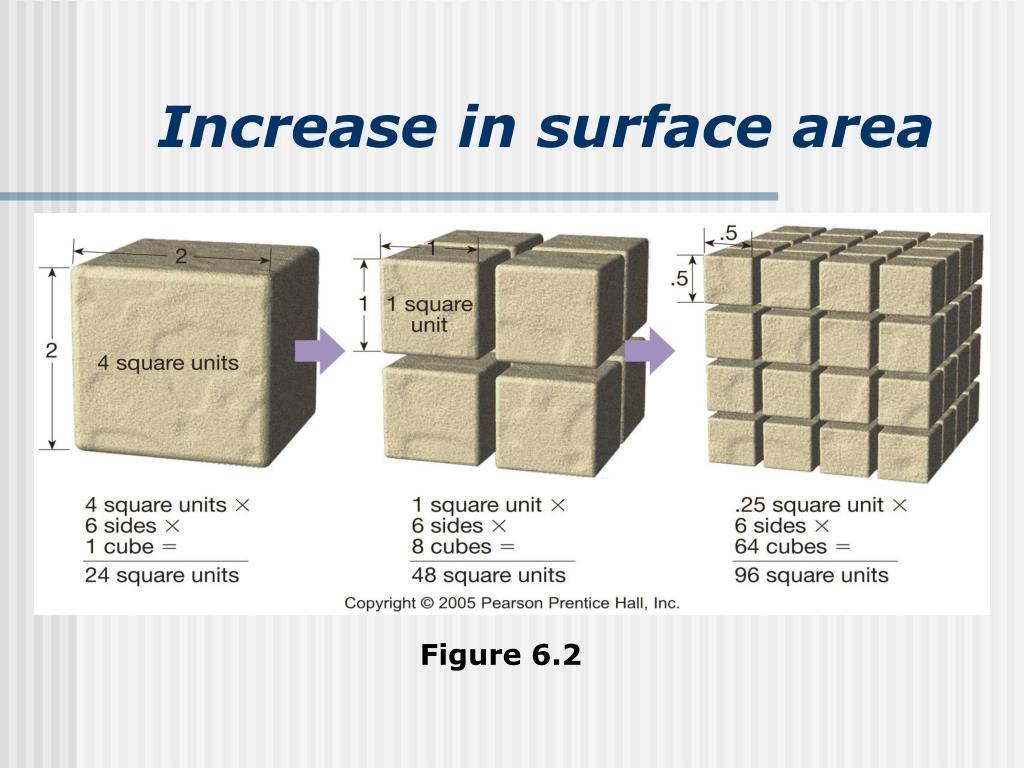
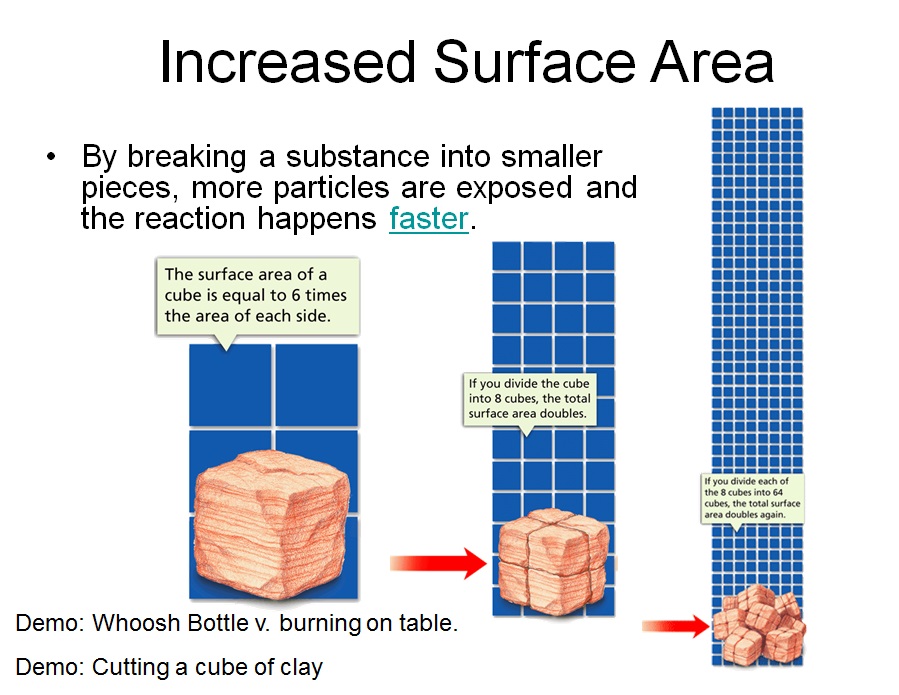
Increasing The Surface Area Of A Solid ____.

Imagine you have a big, smooth rock. It looks nice, but it’s kind of… boring, right? Now, what if we could give that rock a super-powered makeover? We can do something that makes it way more interesting, and it all comes down to something called surface area.
Think of it like this: the surface area is all the "outside bits" of something. It's the skin, the crust, the stuff you can actually touch. When we talk about increasing the surface area of a solid, we’re essentially giving it more accessible outsides. It’s like taking a plain t-shirt and adding a bunch of cool pockets and patches. Suddenly, it’s got more going on!
Why is this so much fun? Well, it unlocks a whole world of cool interactions. Things that were once slow or shy start to become incredibly active. It’s like they suddenly got a megaphone!
Must Read
Let’s take a humble piece of sugar, for example. A big lump of sugar dissolves in your tea, but it takes its sweet time. It’s like one person slowly telling a story in a quiet room.
But if you take that same sugar and crush it into fine powder, like fairy dust, it’s a whole different ballgame. Each tiny grain has its own little surface, its own little place to say hello to the tea. It’s like a hundred tiny people all shouting the story at once!
That powder sugar dissolves almost instantly. It’s a dazzling display of speed and efficiency. This is the magic of increasing surface area at play. It’s not just about making things dissolve faster, though. Oh no, that’s just the appetizer!
Think about baking. When you bake a cake, you want all those lovely ingredients to mix and react. If you used a giant, uncrushed walnut in your batter, it would barely get involved. It would be like a grumpy guest hiding in the corner at a party.
But if you chop that walnut into tiny little pieces, or even grind it into a fine flour, it spreads its nutty goodness everywhere. It gets to mingle with all the other ingredients. The whole cake benefits from its presence.

This is a fantastic example of how we can make a solid, like that walnut, much more effective. By breaking it down, we expose more of its deliciousness. It’s a culinary superpower, really!
And it’s not just about food. Let’s talk about science experiments that are just plain cool. Have you ever seen how quickly a tiny bit of powder can catch fire? It’s mesmerizing, and a little bit scary!
This is often because the material has a massive surface area. When it hits the air, there are so many tiny particles ready to react with the oxygen. It’s like a chain reaction of exciting events. This is what happens with things like dust explosions, which can be quite dramatic.
Of course, it's important to be careful with these kinds of things. But the sheer power and speed are undeniably fascinating. It shows you how much more is happening "under the hood," or rather, on the outside, than we might think.
Consider how we breathe. Our lungs are incredibly clever. They are designed to have a huge surface area. They are packed with millions of tiny little air sacs called alveoli.

Each alveolus is like a tiny balloon. And there are so many of them! This massive collection of tiny surfaces allows us to absorb oxygen from the air really, really efficiently. It’s how we get the energy to run and play and do all the things we love.
If our lungs were just a couple of big, smooth bags, we wouldn’t get enough oxygen. We'd be feeling pretty sluggish, that's for sure. The complex, wrinkly, vast surface of our lungs is a marvel of engineering.
Think about how a sponge works. A simple, solid block of material wouldn't soak up water very well. It would just sit on top. But a sponge is full of tiny holes and channels.
These holes create a massive network of surfaces that can interact with the water. The water gets drawn in, pulled into all those nooks and crannies. It’s a visual representation of increased surface area in action. It’s a very satisfying process to watch.
This is why things like activated charcoal are so special. It looks like a simple black powder, but it has been treated to have an incredibly porous structure. This means it has a mind-bogglingly large surface area.
Because of this huge surface area, activated charcoal is amazing at trapping impurities. It’s used in water filters, air purifiers, and even in medicine to absorb poisons. It’s like a super-sponge for unwanted molecules.

The science behind it is all about that extra "skin" it has. More skin means more opportunities for things to stick to it. It's a simple concept with profound effects.
What’s truly captivating is how this principle pops up everywhere. It’s not just in chemistry labs or fancy kitchens. It’s in nature, in our bodies, and in everyday objects.
Imagine a tiny seed. It needs to absorb water to sprout and grow. Its outer shell has to be permeable enough to let that water in. And often, seeds have intricate textures that increase their ability to interact with their environment.
Even the way paint works involves surface area. When you paint a wall, the paint needs to spread out and stick. The tiny particles of pigment in the paint have a surface area that allows them to bind to the surface of the wall and to each other.
It's a fundamental idea that unlocks so much potential. It's like finding a secret key that opens doors you never knew existed. You start seeing the world differently, noticing how things are designed to interact.
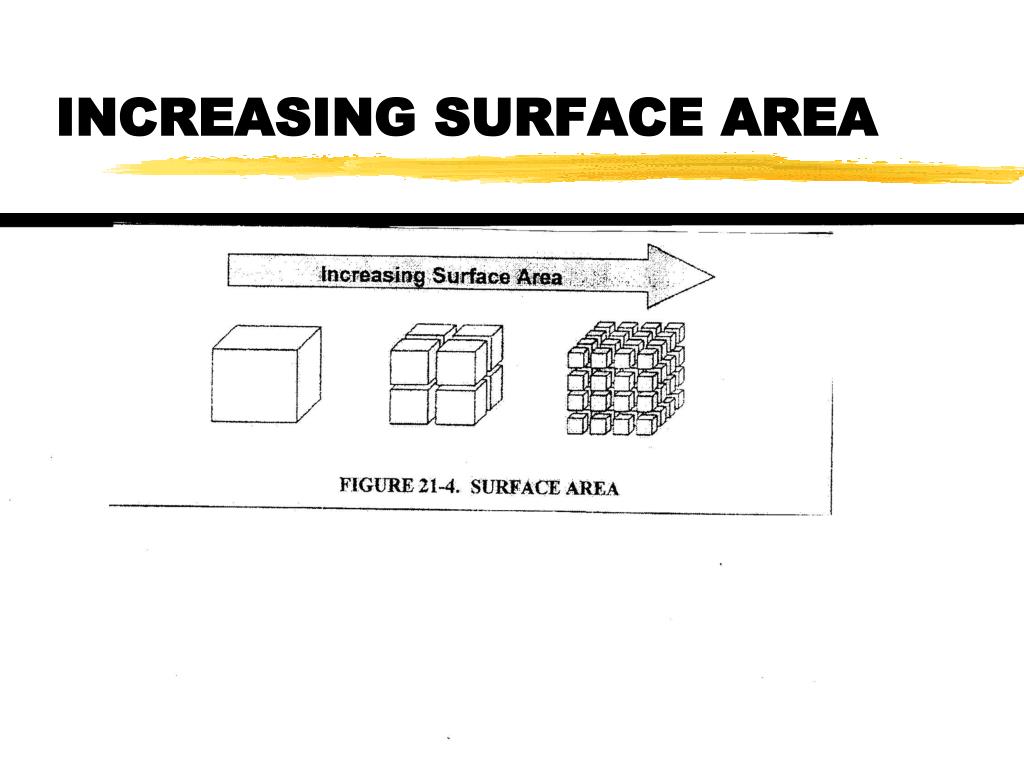
Think about catalysts in industrial processes. Many chemical reactions that are important for making plastics or fuels would be too slow to be useful without catalysts. These catalysts often have very high surface areas, allowing them to efficiently speed up those reactions.
They provide a platform for the molecules to meet and react. It’s like setting up a super-efficient dating service for chemicals. The more surface area available, the more matches can be made.
So, the next time you see something dissolve quickly, or a powder burst into flame, or even just look at a sponge, remember the hidden superpower at play. It’s the wonder of increasing the surface area of a solid.
It’s a simple idea, but it leads to some truly spectacular results. It's a reminder that sometimes, the most amazing things happen when we break things down and give them more room to shine. It’s a concept that’s both elegant and incredibly powerful.
It’s a journey into understanding why things behave the way they do. And that curiosity, that desire to know more, is what makes it so special and entertaining. You might just find yourself looking at everyday objects with new eyes, wondering about their hidden surfaces and the magic they hold.
The more you have on the outside, the more you can do on the inside.
It’s this constant interplay between the surface and what’s within that makes the world so dynamic. And for solids, increasing that surface is like giving them a secret ingredient for success. It’s a concept that’s truly worth exploring.