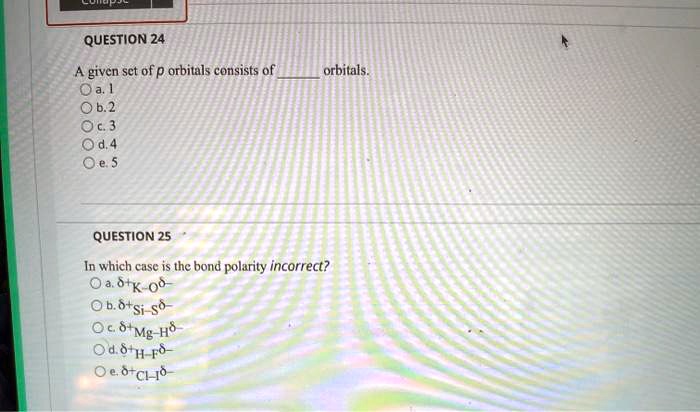
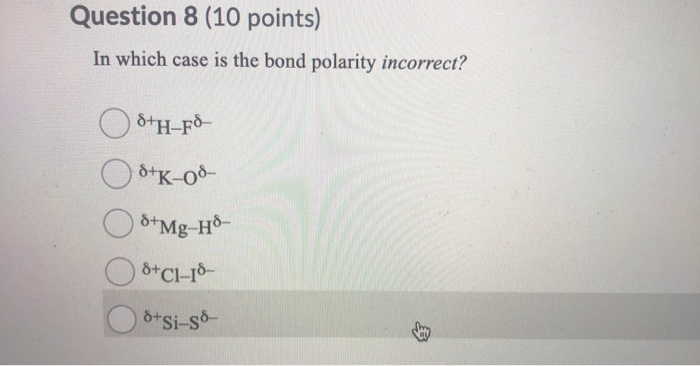
In Which Case Is The Bond Polarity Incorrect
Ah,
Understanding concepts like bond polarity isn't just for lab coats and textbooks; it has surprising and practical benefits for our everyday lives. At its core, bond polarity helps us predict how substances will behave. It tells us whether a molecule will be
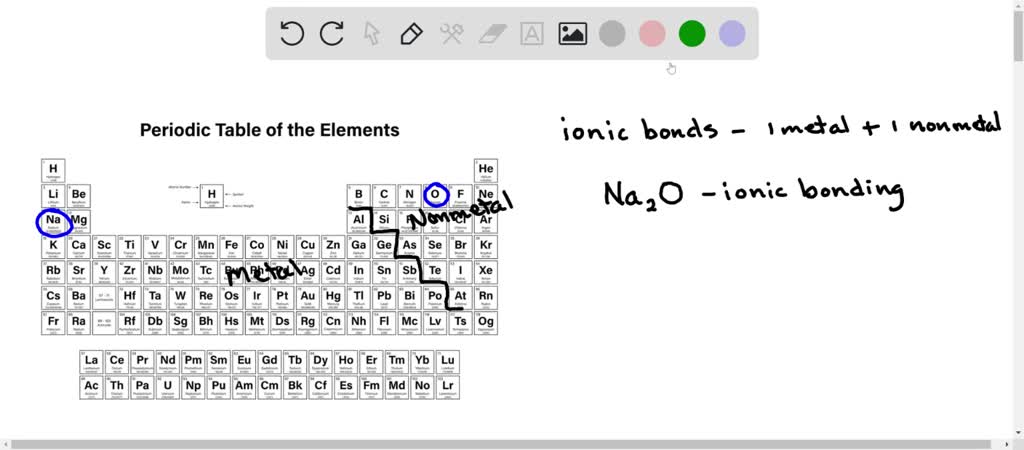
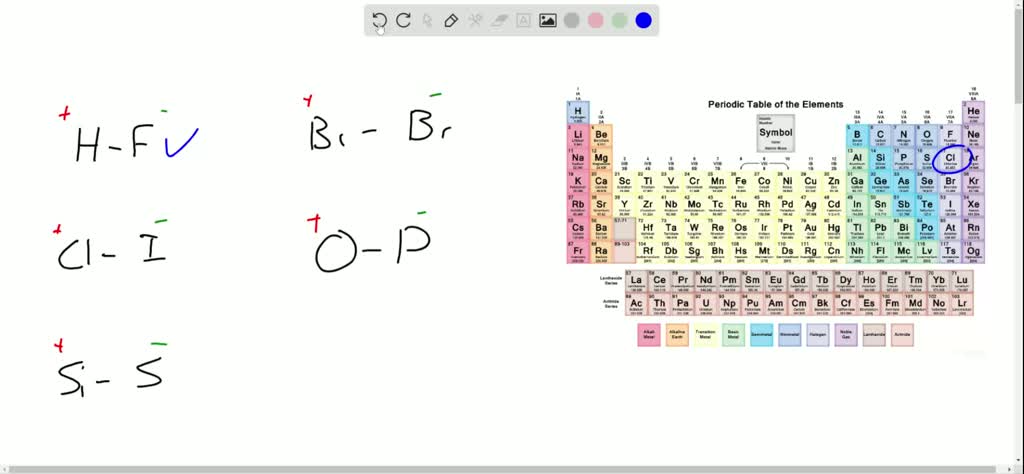
Consider common examples: the water molecule itself, H₂O, is a classic case of polar bonds. The oxygen atom pulls the shared electrons closer, giving it a partial negative charge and the hydrogen atoms a partial positive charge. This polarity is why water can dissolve so many different substances, acting as the universal solvent we rely on daily. Conversely, oils and fats, with their largely nonpolar bonds, don't mix with water – a principle you see every time you try to wash a greasy pan with just water. Even the way paints adhere to surfaces or how certain plastics are manufactured involves understanding these subtle electrical differences within molecules.
Must Read
So, how can you get a better handle on, or simply appreciate, the nuances of bond polarity, especially when things get a bit tricky? One of the most effective ways is to visualize. Many online resources and apps offer 3D models of molecules that clearly illustrate the charge distribution. Seeing the electron cloud shift can make a world of difference. Secondly, practice! Work through examples, perhaps starting with simple diatomic molecules and moving to more complex ones. Don't be afraid to encounter cases where the initial assumption about bond polarity might be
Finally, connect it back to the real world. Next time you're cooking, notice how oil and vinegar separate, or how salt dissolves in water. These are all demonstrations of polarity in action. The more you see these principles at play, the more intuitive and