Identify The Functional Groups In Maltose.

So, I was at this baking competition the other day – you know, the kind where people get way too serious about their sourdough. And there I was, trying to explain to Brenda from accounting why her brioche was a little… dense. I pointed to the sugar she’d added, a beautiful, crystalline mound, and said, “Brenda, it’s all about the chemistry, darling. Especially when you’re dealing with sugars like maltose!” She just blinked at me, bless her heart, and asked if I’d brought enough sprinkles. Clearly, a little molecular breakdown was in order. And that, my friends, is how we ended up talking about the functional groups in maltose!
Honestly, when you break it down, it’s not some intimidating, lab-coat-wearing kind of science. It’s the secret sauce behind so many things we love, from that slightly malty flavor in your favorite beer (oops, did I say beer? I meant… bread!) to the sweetness in your morning oatmeal. Maltose, this humble disaccharide, is a fascinating little molecule, and understanding its building blocks, its functional groups, is like unlocking a treasure chest of how it behaves.
Let’s dive in, shall we? Think of functional groups as the personality traits of a molecule. They’re specific arrangements of atoms that give a molecule its characteristic chemical properties and reactivity. They’re the bits that actually do things. Without them, our organic molecules would be… well, a lot less interesting. And maltose, with its two glucose units linked together, has a few particularly noteworthy ones.
Must Read
The Star of the Show: Hydroxyl Groups
If there’s one functional group that absolutely dominates maltose, it’s the hydroxyl group. Seriously, it’s everywhere! You’ve probably heard of them before; they’re the $-\text{OH}$ bits. You know, the oxygen atom bonded to a hydrogen atom?
In maltose, you’ve got a boatload of these hydroxyl groups. Each of the glucose units that make up maltose is festooned with them. Think of it like a Christmas tree, and the hydroxyl groups are the ornaments, sparkling and adding to the overall structure and, crucially, its properties.
Why are they so important? Well, these $-\text{OH}$ groups are super polar. That means the oxygen atom pulls the shared electrons closer to itself, giving it a slightly negative charge, while the hydrogen gets a slightly positive charge. This polarity is what makes maltose, and other sugars like it, so soluble in water. Remember how you dissolve sugar in your tea? That’s your hydroxyl groups working overtime, forming hydrogen bonds with the water molecules. It’s like a molecular party, where everyone’s holding hands!
Beyond solubility, these hydroxyl groups are the reason sugars can participate in reactions like esterification and etherification. These are fancy terms, I know, but they basically involve the $-\text{OH}$ group reacting with other molecules to form new bonds. This is how sugars get modified, how they link up, and how they can be converted into other things. It’s the fundamental basis for so much of carbohydrate chemistry. Pretty neat, huh?
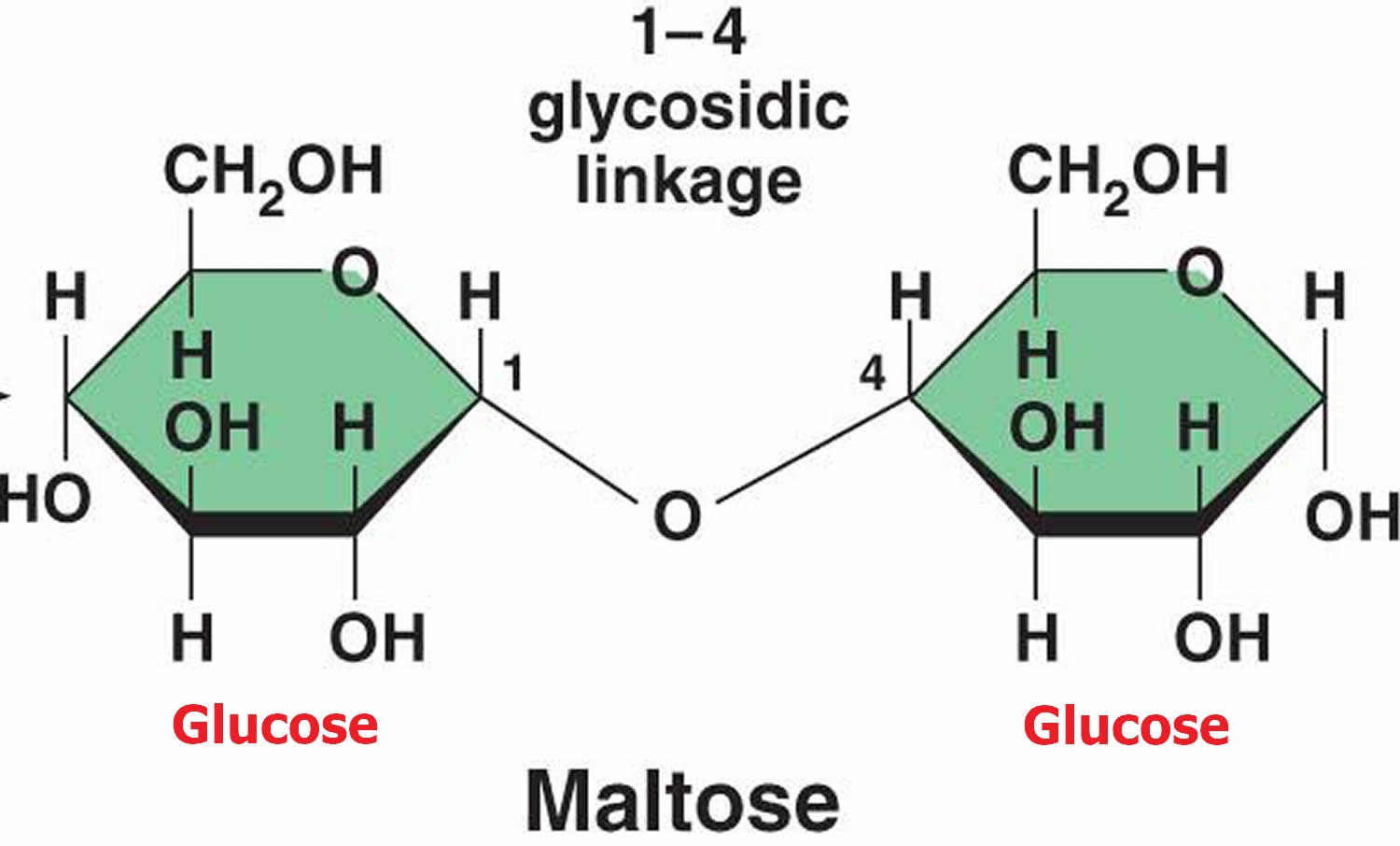
The Crucial Link: The Glycosidic Bond
Now, maltose isn’t just two loose glucose molecules hanging out. They’re joined together by a special type of bond called a glycosidic bond. And guess what functional group is essential for forming this bond? You guessed it – another hydroxyl group!
Specifically, a glycosidic bond is formed when the hydroxyl group on the anomeric carbon of one sugar molecule reacts with a hydroxyl group on another sugar molecule. The anomeric carbon is that super special carbon atom in a cyclic sugar that was originally part of the carbonyl group. It’s like the VIP section of the sugar molecule.
In maltose, one glucose molecule is linked to another via an $\alpha(1 \to 4)$ glycosidic bond. This means the hydroxyl group on the first carbon (C1) of the first glucose molecule (in its alpha configuration) has bonded to the hydroxyl group on the fourth carbon (C4) of the second glucose molecule. This linkage is what makes maltose a disaccharide. If it were just single glucose units, it would be a monosaccharide, and a lot less interesting for our discussion.
This glycosidic bond is pretty stable, but it can be broken down by hydrolysis, especially with the help of enzymes like maltase. This is how our bodies break down maltose into its constituent glucose units for energy. It’s a controlled dismantling, like taking apart a LEGO set with the right instructions. And guess which functional group is involved in that process? Yup, the hydroxyl group, as water molecules come in to ‘undo’ the bond.
The Cyclic Nature: The Hemiacetal/Acetal Functionality
Okay, so sugars don’t always just hang around in a straight line. In solution, glucose (and thus maltose, which is made of glucose) exists primarily in a cyclic form. This is where things get a little more nuanced and oh-so-cool.
When glucose cyclizes, it forms a six-membered ring (called a pyranose ring). In this process, the aldehyde group of the open-chain form reacts with a hydroxyl group on the fifth carbon to form a hemiacetal. A hemiacetal has a structure where a single carbon atom is bonded to both an $-\text{OH}$ group and an $-\text{OR}$ group (where R is another carbon chain). Think of it as a partially formed acetal.
Now, the anomeric carbon we talked about earlier? That’s the carbon where the hemiacetal is located. This hemiacetal group is quite reactive. It can open and close, allowing the molecule to switch between its cyclic and open-chain forms. This interconversion is super important because it allows for the formation of different stereoisomers, like the alpha and beta anomers of glucose, which then affect how they link up via glycosidic bonds.
When two sugar units like glucose link up via a glycosidic bond, the anomeric carbon of one sugar (which was part of a hemiacetal) reacts with a hydroxyl group on another sugar. If the anomeric hydroxyl group of the first sugar reacts, and the resulting bond is formed with another sugar molecule, the hemiacetal is converted into an acetal. In the case of the glycosidic bond in maltose, the anomeric carbon of one glucose becomes an acetal carbon.
The presence of these cyclic structures, with their hemiacetal and acetal functionalities (even though one is converted to an acetal in the glycosidic bond), is key to the stability and reactivity of sugars. It’s what gives them their characteristic ring shapes and dictates their interactions with other molecules.
The 'Reducing Sugar' Bit: The Hemiacetal Again!
This is where things get really interesting for analytical chemists and anyone who likes their sweet treats to reveal their secrets. Maltose is classified as a reducing sugar. And guess which functional group is responsible for this property? You guessed it – the hemiacetal group!
A reducing sugar is a sugar that has a free hemiacetal (or hemiketal) group. This group can be oxidized, meaning it can donate electrons to another molecule. In the presence of a mild oxidizing agent, like Benedict’s reagent or Tollens’ reagent (think of these as fancy chemical tests), the hemiacetal group in maltose can be oxidized to a carboxylic acid. This oxidation causes a visible change, like the formation of a red precipitate in Benedict’s test. That’s how you know you’ve got a reducing sugar on your hands!
Because maltose is made of two glucose units linked by an $\alpha(1 \to 4)$ bond, the anomeric carbon of one of the glucose units is free and exists as a hemiacetal. The other glucose unit’s anomeric carbon is involved in the glycosidic bond and is therefore an acetal, not a hemiacetal. So, even though it’s a disaccharide, it still has that crucial free hemiacetal group, making it a reducing sugar. This is a really important characteristic for identifying and quantifying sugars in various samples. It’s like a built-in barcode for identifying its chemical identity.
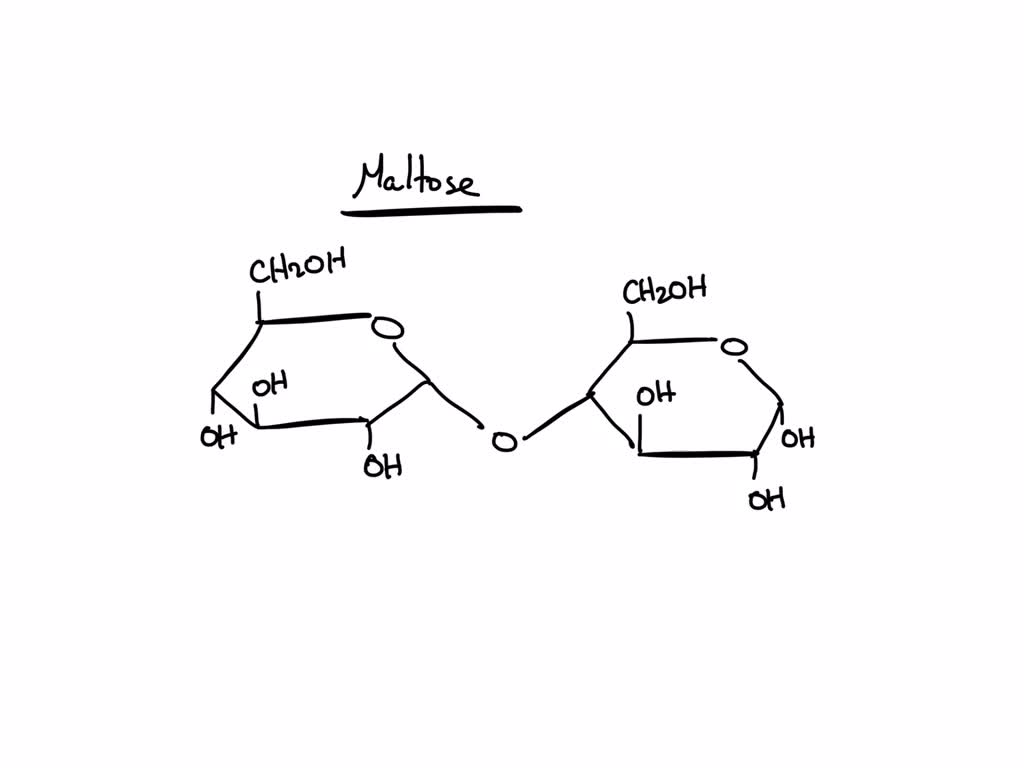
Putting it All Together: Maltose in Action
So, let’s recap what we’ve found lurking within the structure of maltose. We have an abundance of hydroxyl groups ($\text{-OH}$) that contribute to its water solubility and its ability to form hydrogen bonds. We have the pivotal glycosidic bond, formed between hydroxyl groups, which links the two glucose units together. And we have the cyclic structures with their hemiacetal and acetal functionalities, with the crucial free hemiacetal group on one of the glucose units giving maltose its ‘reducing sugar’ status.
These functional groups aren’t just abstract chemical concepts. They dictate everything about maltose. Their arrangement is what gives it its specific sweetness, its ability to be digested by enzymes, and its role in the brewing process. For instance, in brewing, yeast enzymes break down maltose into glucose, which the yeast then ferments to produce ethanol and carbon dioxide. Without those functional groups, that glorious pint would never come to be!
When I was chatting with Brenda (before the sprinkle intervention), I tried to explain this. I said, “Brenda, that sweetness you taste? That’s those polar hydroxyl groups interacting with your taste buds. And the fact that your yeast can eat it to make bread rise? That’s the glycosidic bond and the hemiacetal group that the yeast enzymes can get to work on.” She just nodded slowly, still eyeing the sprinkle jar. Some people are just more visually stimulated, I guess.
The beauty of organic chemistry, and biochemistry in particular, is that it’s all about these little structural details. A single atom, a slight rearrangement, and you have a molecule with entirely different properties. Maltose, with its combination of hydroxyls, glycosidic bonds, and hemiacetal/acetal groups, is a perfect example of how these functional groups define a molecule’s identity and its role in the world. So next time you enjoy something with a malty flavor, take a moment to appreciate the intricate dance of atoms and functional groups that made it all possible. It’s more than just sugar; it’s a testament to the elegance of molecular design!