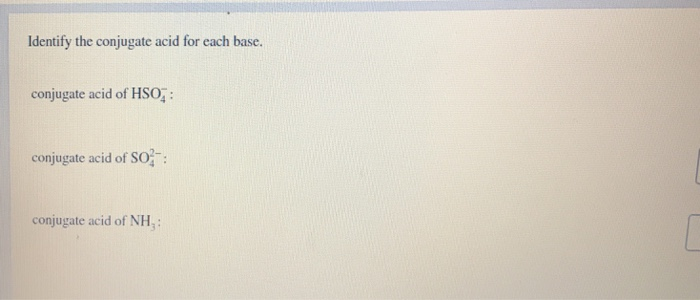
Identify The Conjugate Acid For Each Base

Hey there, science enthusiasts and curious minds! Ever feel like chemistry is all about complicated formulas and stuffy labs? Well, buckle up, buttercup, because we're about to inject a little fun into the world of acids and bases. Today, we're diving into a super cool concept: identifying the conjugate acid for each base. Don't let those fancy words intimidate you; it's actually more like a chemical "who's who" game, and trust me, it can make your brain do a little happy dance!
So, what exactly are we talking about here? Think of it this way: acids are like generous donors, always ready to hand over a proton (that's a positively charged hydrogen ion, H+). Bases, on the other hand, are like eager receivers, always looking for a proton to snatch up. And when a base grabs onto a proton, something awesome happens – it becomes a conjugate acid. It's like a transformation, a little chemical glow-up!
Why should you care about this proton-grabbing party? Because understanding these relationships unlocks a whole new level of appreciating how the world around you works. From the fizz in your soda to the delicate balance in your body, these acid-base shenanigans are happening all the time. And frankly, knowing this stuff just makes you sound way cooler at parties. 😉
Must Read
The Magic of the Proton Swap
Let's get down to the nitty-gritty, but in a way that’s more like a fun puzzle than a pop quiz. When we talk about acids and bases in this context, we're usually referring to the Brønsted-Lowry definition. This definition is super handy because it focuses on the transfer of protons.
A Brønsted-Lowry acid is a proton (H+) donor. It’s the one saying, "Here, take this proton, I've got plenty!"
A Brønsted-Lowry base is a proton (H+) acceptor. It’s the one saying, "Ooh, a proton! Mine!"

Now, here’s the kicker: when a base accepts a proton, it doesn't just disappear into the void. It forms something new. This new thing is its conjugate acid. It’s like the base got a proton-infused superpower!
Conversely, when an acid donates a proton, what’s left behind is its conjugate base. Think of it as the acid’s proton-depleted shadow. This entire process is a continuous cycle, a beautiful dance of proton giving and receiving.
Spotting the Conjugate Acid: Your Detective Skills
So, how do you become a master of identifying conjugate acids? It’s all about looking for that tell-tale sign: the presence of an extra hydrogen ion (H+). When you see a base, your mission, should you choose to accept it, is to imagine it grabbing a proton.
Let's take a common base, like the hydroxide ion, OH-. It's a pretty popular proton magnet, wouldn't you say? If OH- acts as a base, it needs to accept a proton. Where does this proton usually come from? From an acid, of course! Let's say it accepts a proton from water (H2O), which can act as both an acid and a base.

So, the base is OH-. It sees that H+ from H2O and wham! It grabs it. What does OH- + H+ become? You guessed it – H2O! So, in this scenario, the conjugate acid of the base OH- is H2O. Pretty neat, right? It's like finding out your favorite drink is also the secret ingredient to a super-powered potion!
Let's try another one. How about ammonia, NH3? Ammonia is a classic base. When it encounters an acid, it's like a kid spotting a dropped ice cream cone – it's going to scoop it up! If NH3 accepts a proton (H+), what does it become? It becomes NH4+. So, the conjugate acid of the base NH3 is the ammonium ion, NH4+. See? You're already a conjugate acid detective!
Why This Chemical CSI is Actually Fun
Okay, I know what you might be thinking: "This sounds like homework." But honestly, once you get the hang of it, it’s like unlocking a secret language. It helps you understand why certain reactions happen, why some solutions are acidic and others are basic. It’s the underpinning of so much of the chemistry we encounter daily.

Think about it: the buffering systems in your blood that keep you alive and kicking? They rely on these conjugate acid-base pairs. The tangy taste of your yogurt? That's partly due to lactic acid and its conjugate base. The effectiveness of cleaning products? Yep, acid-base chemistry is at play there too.
And here’s a fun thought experiment: imagine a world without proton exchange. It would be a very different, and probably much less interesting, place! This constant give-and-take is what drives so many vital processes. It's a fundamental aspect of the dynamic nature of the universe.
More Practice Makes Perfect (and More Fun!)
The more you practice identifying these conjugate acid-base pairs, the more intuitive it becomes. It's like learning to ride a bike – a little wobbly at first, but soon you're cruising!
Let's consider a base like the bicarbonate ion, HCO3-. What do you think its conjugate acid would be? If HCO3- acts as a base, it accepts a proton. So, HCO3- + H+ = H2CO3. Therefore, the conjugate acid of the base HCO3- is carbonic acid, H2CO3. Boom! Another one solved!
What about the carbonate ion, CO3^2-? If it acts as a base, it accepts a proton: CO3^2- + H+ = HCO3-. So, the conjugate acid of the base CO3^2- is the bicarbonate ion, HCO3-. See how these pairs are linked? It's a chemical family tree!
The key takeaway is to always ask yourself: "If this substance accepts a proton (H+), what would it become?" That will reveal its conjugate acid.
Your Inspiring Chemical Adventure Awaits!
So, there you have it! Identifying conjugate acids for bases isn't just a chemistry concept; it's a glimpse into the fundamental interactions that shape our world. It’s about understanding transformation, about the dynamic exchange of particles that makes life, well, life!
Don't stop here! The world of acids and bases is vast and fascinating. Explore further, ask more questions, and let your curiosity lead you down a path of scientific discovery. Every new concept you grasp is like adding another superpower to your brain’s utility belt. Go forth, be curious, and embrace the awesome power of chemistry! You’ve got this!





