How Many Atoms Are In 6.2 Moles Of Aluminum

Okay, buckle up, buttercups, because we're about to dive headfirst into a universe of teeny-tiny, invisible-to-the-naked-eye wonders! We're talking about atoms, the building blocks of everything around us. You know, like your trusty frying pan, that delicious snack you’re probably eyeing, or even the very air you’re breathing right now. Yep, all of it is just a zillion, bazillion, gazillion little atomic buddies hanging out together.
Now, imagine you’ve got a super-duper special measuring cup, but instead of measuring flour or sugar, it measures… well, atoms! Scientists have a fancy word for a big ol' bunch of atoms, and it’s called a mole. Think of a mole like a cosmic party pack. It's a specific, humongous number of things. And when we’re talking about atoms, this party pack is ridiculously, unbelievably, mind-bogglingly HUGE.
Let's talk about our star of the show today: Aluminum! You know, the stuff that makes those super-lightweight foil wraps that keep your sandwiches from going rogue in your lunchbox? Or maybe the sleek casing on your phone? That’s our buddy, Aluminum! So, the question on everyone’s lips (well, maybe not everyone's lips, but definitely yours now!) is: how many of these tiny aluminum atom party packs are we talking about if we have 6.2 moles of this amazing metal?
Must Read
To give you a tiny hint of the scale we're dealing with, one single mole of anything has a number of particles so big, it makes the national debt look like pocket change. It's a number so immense, if you tried to write it down without stopping, you’d probably run out of ink, paper, and probably the entire planet's trees! This magical, gargantuan number is called Avogadro’s Number, and it's approximately 6.022 x 1023. Let that sink in. That’s a 6 followed by 23 zeros! It's like counting every single grain of sand on all the beaches in the entire world, then multiplying that by a million, and then maybe adding a few extra galaxies for good measure.
So, if one mole of aluminum is already this outrageously, fantastically large number of atoms, what do you think happens when we have 6.2 of those giant party packs? It’s like saying, "Okay, we have the world's biggest birthday cake, and we're going to have 6.2 of them!" We're not just talking about a lot; we're talking about an astronomical, supernova-level, mind-bending abundance of aluminum atoms.
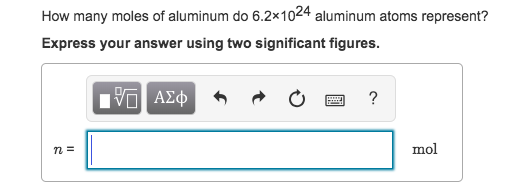
Let's break it down in a way that’s less like a science textbook and more like a whispered secret from the universe. If one mole is 6.022 x 1023 aluminum atoms, then 6.2 moles is simply 6.2 times that already colossal number. Imagine you have a giant box filled with 6.022 x 1023 little toy cars. Now, imagine you have 6.2 of those boxes! You wouldn't be able to fit them in your garage. You wouldn't be able to fit them in your entire neighborhood! You'd need a fleet of rocket ships just to store them!
So, how many atoms are in 6.2 moles of aluminum? Well, get ready for your jaw to hit the floor (or at least reach a respectable desk height). We take that magnificent 6.022 x 1023 and multiply it by our handy-dandy 6.2. And when we do that… drumroll please… we get a number that’s roughly 3.73 x 1024 aluminum atoms!
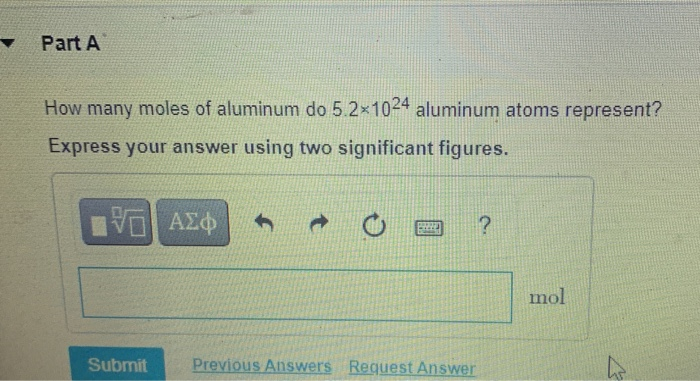
Let’s put that in perspective. That’s a 3 followed by a 7, then a 3, and then… wait for it… 24 zeros! That’s more zeros than you can shake a stick at. It’s more zeros than exist in the number of stars you can see on a clear night, multiplied by the number of hairs on all the heads of every single person on Earth, and then some! It's enough aluminum atoms to build a shimmering, metallic galaxy all its own! If each atom were the size of a tiny speck of glitter, you’d have enough glitter to cover every single surface in the known universe, and then some, with a dazzling, atomic sheen.
It’s a number so big, it makes the concept of “a lot” feel like an understatement of epic proportions. We’re talking about the sheer, unadulterated, atom-powered glory of aluminum on a scale that tickles the very edge of our imagination.
So, next time you’re wrapping up that sandwich or admiring your phone, remember the incredible, unseen army of aluminum atoms that are hard at work. And when you hear about moles, don't just think of a little furry creature; think of these epic, atomic party packs, each one holding a universe of tiny, essential building blocks. Isn’t science just the coolest, most mind-blowing adventure ever?