How To Tell If Atoms Are In The Same Plane

So, you’re staring at some atoms, right? Maybe they’re all clustered together in a molecule, or perhaps they’re part of some larger structure. And you’re thinking, “Hey, are these little guys all hanging out on the same flat surface? Like, are they all on the same plane?” It’s a totally valid question, honestly. It’s not like atoms come with little sticky notes saying, “Yep, I’m on plane A!”
Think about it: we’re talking about stuff so tiny, it’s practically invisible. So how do we, with our relatively gigantic eyeballs, even begin to figure this out? It’s kind of like trying to tell if a bunch of gnats are all perfectly aligned on a dinner plate from across the room. Tricky, right?
Well, thankfully, we’ve got some tricks up our sleeves. Science, that is. And a little bit of geometry, which, let’s be real, can feel like a superpower sometimes. So, grab your imaginary coffee mug, settle in, and let’s dive into this whole “atoms in the same plane” mystery. It’s not as intimidating as it sounds, I promise. We’ll break it down, nice and easy.
Must Read
The Cosmic Dance of Atoms
Atoms, you know, are the fundamental building blocks of everything. They’re constantly buzzing around, bumping into each other, forming bonds. It's like a never-ending cosmic dance. And in this dance, sometimes they decide to get together and form a little group, a molecule. Or maybe they’re part of something even bigger, like a crystal lattice. Whatever the case, their arrangement matters. A LOT.
Why does it matter, you ask? Well, because the shape of a molecule, its 3D structure, totally dictates how it behaves. Think about it. A molecule that’s all bent out of shape is going to interact differently than one that’s perfectly straight, or, you guessed it, one where all its atoms are chilling on the same flat surface. It’s like trying to fit a square peg into a round hole. Sometimes it works, sometimes it’s a disaster. And for molecules, this can mean the difference between being a life-saving drug and… well, not being a life-saving drug. No biggie, right?
So, when we’re talking about atoms being in the same plane, we’re basically asking if they’re coplanar. That’s the fancy science word for it. Coplanar. Sounds important, doesn’t it? Like something you’d see on a really serious document.
The Geometric Basics: What Even IS a Plane?
Before we get too deep into the atomic ballet, let’s just quickly refresh ourselves on what a plane is. You know, in geometry class? Remember that? It’s like an infinitely large, perfectly flat surface. Think of a piece of paper, but it goes on forever in all directions. Or a perfectly still lake. Or, if you’re feeling a bit more adventurous, the surface of a perfectly flat yoga mat. You get the idea. It has no thickness. Zero. Nada.
Now, how do we define a plane? You need at least three non-collinear points. Collinear just means they all lie on the same straight line. So, if you have three points that aren’t on the same line, they’ll automatically define a unique plane. Like a tiny, perfect triangle. That triangle’s flat surface is part of an infinite plane. It’s the minimum requirement, the building blocks of flatness, if you will. Think of it as the cosmic equivalent of drawing three dots and connecting them. Boom, plane defined.
If you have more than three points, then things get interesting. They could all be on the same plane, or they could be all over the place. And that’s where our atom-checking skills come in!
Detecting Planarity: The Sherlock Holmes Approach
Okay, so how do we actually check if our atoms are playing nicely on the same flat surface? It’s not like we can just pull out a ruler and a spirit level. Atoms are a bit too… elusive for that. We need some smarter methods.
Method 1: The Vector Approach (Don’t Panic!)
Alright, I know what you’re thinking. “Vectors? Oh no, my brain is already foggy from that last coffee.” But seriously, it’s not that bad. Think of vectors as little arrows that represent direction and distance. We can use these arrows to describe the positions of our atoms relative to each other.
Let’s say we have four atoms: A, B, C, and D. If atoms A, B, and C are definitely on the same plane (because, hey, three points always define a plane, remember?), we can use them as our reference. We can then draw vectors from one of these reference atoms, say A, to the other atoms. So, we’d have vector AB and vector AC. These two vectors lie within our reference plane.
Now, we look at atom D. We draw a vector from A to D, making vector AD. The big question is: does vector AD lie in the same plane as vectors AB and AC? If it does, then atom D is coplanar with A, B, and C. If it doesn’t, then D is off on a tangent, and our atoms are not all on the same plane.

How do we mathematically check if AD lies in the plane defined by AB and AC? This is where a cool mathematical operation called the scalar triple product comes in. It sounds fancy, but it's basically a way of checking if three vectors are coplanar. If the scalar triple product of AB, AC, and AD is zero, then they are all coplanar. If it’s not zero, then they are not. Easy peasy, lemon squeezy. Or, in this case, zero equals coplanar, non-zero equals… not coplanar. Your new superpower!
Think of it this way: if you have two vectors on a flat surface, and you try to add a third vector to them that also stays on that surface, you can do it. It’s like drawing on a piece of paper. But if you try to add a third vector that sticks straight up from the paper, it’s not going to be in the same plane anymore, is it? The scalar triple product is the math way of saying, “Nope, that third vector is sticking up!”
Method 2: Looking at Bond Angles and Distances (The Molecular Detective)
Sometimes, we don’t need all the fancy vector math. We can get a lot of information just by looking at how atoms are connected and the angles between those connections. This is especially true for organic chemistry, where molecules are made up of carbon, hydrogen, oxygen, and so on. These atoms have specific bonding preferences.
For instance, a carbon atom that is bonded to four other atoms (like in methane, CH4) will usually have a tetrahedral arrangement. That means the four atoms aren't going to be in the same plane. It’s like a little pyramid. So, if you see a carbon atom with four single bonds, and all the atoms attached to it are relatively small, chances are they’re not all coplanar. It’s like looking at a four-leaf clover – it’s got a bit of a 3D vibe.
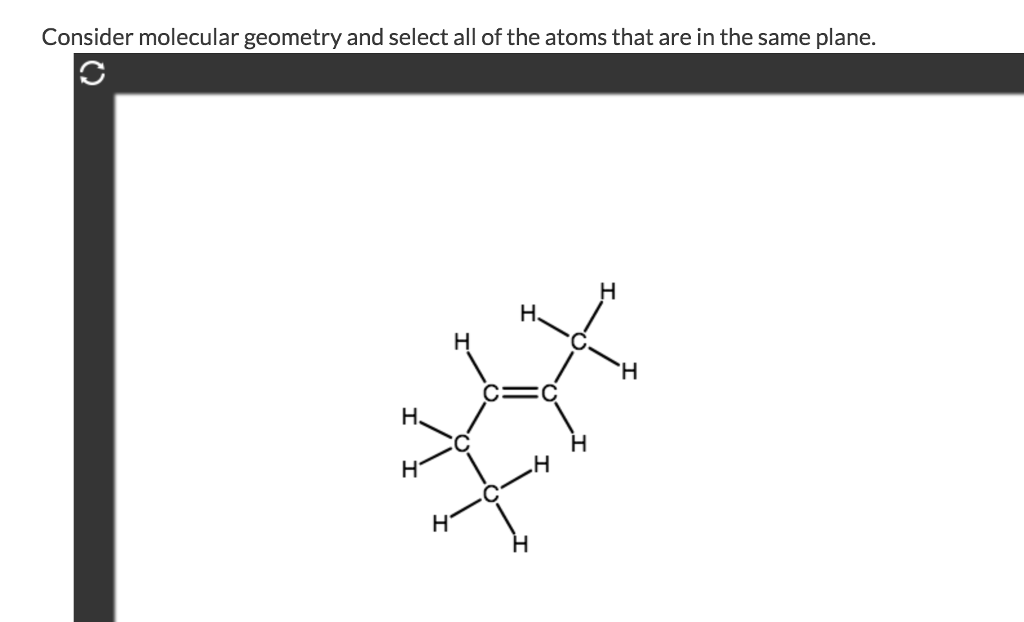
But then you have atoms that like to be flat. Take a carbon atom that’s involved in a double bond. For example, in ethylene (C2H4), the two carbon atoms are double-bonded, and each carbon is also single-bonded to two hydrogen atoms. All six atoms in ethylene – the two carbons and the four hydrogens – are in the same plane! How neat is that?
Why? Because double bonds force a specific geometry called trigonal planar. Think of a three-pronged fork, where the tines are spread out as much as possible in a flat way. The carbon atom is at the center, and the atoms it’s bonded to are at the ends of those prongs. So, if you see a central atom with a double bond, and the atoms attached are arranged in a trigonal planar fashion, you’re probably looking at a planar molecule. It’s like the universe is saying, “Let’s keep this nice and flat!”
The same applies to aromatic rings, like benzene. Benzene has a six-carbon ring with alternating double bonds. Every single atom in that ring, plus the hydrogens attached to each carbon, lies perfectly flat. It's super stable because of this planarity. It’s like the atoms are holding hands in a perfect circle, all on the same dance floor.
So, if you're looking at a molecule and you see a lot of double bonds or rings, especially with certain elements like carbon, there's a good chance a significant portion of it, or even the whole thing, is planar. It’s a good indicator, a strong clue.
Method 3: Using Software (The High-Tech Approach)
Let’s be honest, most of us aren’t going to be calculating scalar triple products by hand for every molecule we encounter. That’s where the magic of modern computing comes in. There are tons of awesome software programs out there that can model molecules in 3D. You might have seen them in textbooks or online. They let you rotate and zoom in on molecules, seeing their exact spatial arrangement.
These programs are your best friends when it comes to figuring out planarity. You can often just click on a group of atoms, and the software will tell you if they are coplanar, or calculate the angles and distances that tell you if they should be coplanar.
Some programs will even highlight planar sections of a molecule for you. It’s like having a built-in planarity detector! You can also use these programs to visualize the electron density around atoms, which can give you hints about bonding types and expected geometries. It's like having x-ray vision for molecular structures.

If you’re really getting into it, you might even use computational chemistry software. These programs can perform calculations that predict molecular structures and properties. They’ll tell you the precise coordinates of every atom in 3D space. From those coordinates, you can then easily determine if a set of atoms falls on the same plane. It’s the most accurate way, hands down. You’re basically asking a super-smart computer, “Are these atoms on the same page?” and it’ll give you a definitive answer.
Why Should You Even Care About Planarity?
Okay, so we’ve talked about how to tell if atoms are in the same plane. But why should you bother? Is it just some abstract geometry problem, or does it have real-world implications? Spoiler alert: it has HUGE implications!
Remember that whole drug example? Many drugs work by fitting into specific protein pockets in our bodies. If a drug molecule isn't the right shape, it just won't fit, and it won't do its job. The planarity (or lack thereof) of its atoms is a crucial part of that shape. It affects how the drug interacts with its target. A flat molecule might slide into a slot easily, while a lumpy one might get stuck or miss it altogether.
Planarity also affects things like light absorption and emission. Molecules that are planar often have conjugated systems of double bonds (like in those aromatic rings we talked about). These systems allow electrons to move more freely, which is key for absorbing and emitting light. This is how pigments get their colors, how solar cells work, and how our eyes detect light. So, the flatness of atoms can literally determine the colors we see!
In materials science, the arrangement of atoms in a solid is critical. Whether those atoms form flat layers or a more complex 3D network determines the material’s properties – its strength, conductivity, flexibility, you name it. Think about graphene, that amazing one-atom-thick sheet of carbon atoms. Its incredible strength and conductivity are all thanks to its perfect planarity. It's literally a 2D material!
So, next time you’re looking at atoms, don’t just think of them as tiny dots. Think about their spatial relationships. Are they holding hands in a straight line? Are they forming a little flat triangle? Or are they scattered like confetti at a party? The answer to that question can unlock a whole universe of understanding about how molecules behave and how the world around us works. Pretty cool, right? It’s like having a secret code to understanding matter. And now, you’ve got a few more keys to that code!