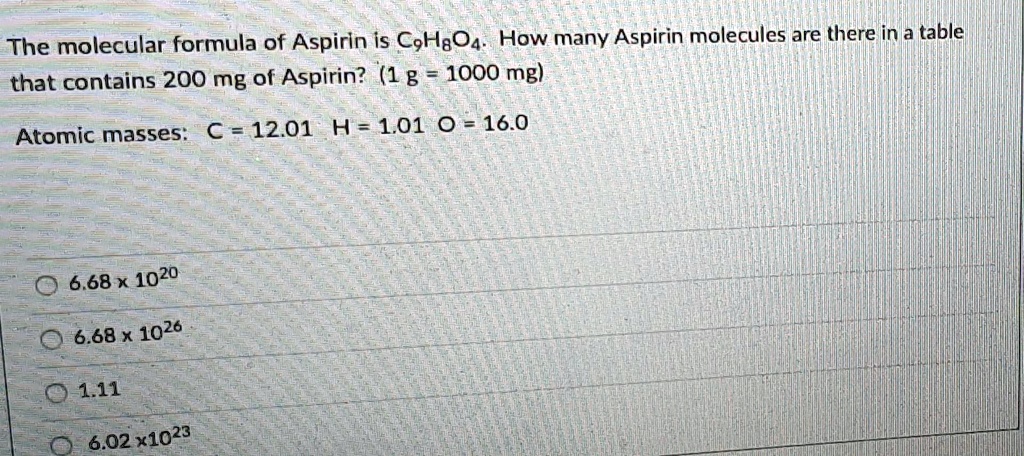
How Many Molecules Are In A Mole Of Aspirin
Ever have one of those days where your head feels like it’s hosting a tiny, aggressive marching band, complete with cymbals crashing and trumpets blaring? Yeah, me too. And what’s usually the go-to superhero in those moments? That little white tablet of aspirin, of course. You pop one, and after a bit of magic (or, you know, science), the band starts packing up their instruments, and blessed quiet descends.
But have you ever stopped to think, while you’re gratefully swallowing that pain-reliever, about what’s actually in there? We’re talking about the microscopic stuff, the building blocks that make aspirin… well, aspirin. And when scientists talk about these tiny bits, they often use a word that sounds a bit fancy: a mole.
Now, “mole” might conjure up images of furry little creatures digging tunnels in your garden, or maybe even that slightly awkward relative at family gatherings. But in the world of chemistry, a mole is a whole different beast. It’s not about furry critters or awkward silences; it’s about a ridiculously, mind-bogglingly huge number of things.
Must Read
Think of it like this: imagine you’re baking a cake. You need flour, sugar, eggs, and all sorts of good stuff. Now, if you were to measure out exactly the right amount of flour for a super-duper, party-sized cake, you wouldn’t just grab a handful, right? You’d use a measuring cup. A mole is kind of like a giant, universal measuring cup for molecules.
But instead of measuring out cups or grams, a mole measures out individual particles. And when we’re talking about aspirin, those particles are called molecules. So, the question is, how many of these tiny aspirin molecules are crammed into one single mole of aspirin? Get ready, because the answer is… well, it’s a number that makes your eyes water.
The Unveiling of Avogadro's Number
The number of molecules in a mole is so important, so fundamental, that it has its own fancy name: Avogadro’s Number. You can think of Avogadro as the guy who did the really hard counting work for us, so we don’t have to. He’s the unsung hero of microscopic quantities.
So, what is this magical number? Drumroll, please… It’s approximately 6.022 x 1023. That’s a 6 followed by 23 zeros! If you’re trying to visualize that, good luck. It’s like trying to count all the grains of sand on all the beaches in the world, and then multiplying that by a number so big it doesn’t even have a catchy nickname yet.
Let’s try to make that number a little less abstract, shall we? Imagine you had a dollar for every single molecule in one mole of aspirin. You’d be richer than every billionaire, every country, and probably every galaxy combined. You could buy the moon. Twice. And then have enough left over to buy a very nice ice cream sundae.
Or how about this: if you could count one molecule every second, without stopping, ever, it would take you longer than the entire age of the universe to count to Avogadro’s Number. You’d be counting through millennia, eons, and probably several geological eras. Your great-great-great-great-great-… (you get the idea) …grandchildren would still be counting. It's basically the universe’s way of saying, "Yep, things get really small."
Aspirin: The Humble Hero and Its Molecules
So, we’ve established that a mole is a colossal collection. Now, let’s bring it back to our friend, aspirin. The active ingredient in aspirin is a molecule called acetylsalicylic acid. It’s a mouthful, I know. You can just think of it as the “headache-busting secret sauce” of the tablet.
When you look at an aspirin tablet, it seems so small, so manageable. You can hold it between your thumb and forefinger. It feels like a single, simple thing. But inside that unassuming white disc are trillions upon trillions of these acetylsalicylic acid molecules, all chilling out, waiting for their moment to shine (or, more accurately, to get to work on that marching band in your head).
So, when a chemist says they have "one mole of aspirin," they aren’t talking about a pile of tablets. They are talking about a specific mass of aspirin that contains exactly 6.022 x 1023 molecules of acetylsalicylic acid. It’s like saying you have "a dozen eggs." You know exactly how many eggs you're getting, even though the eggs themselves can be different sizes.

The reason this concept of the mole is so important is that it allows chemists to work with incredibly tiny things in a way that’s manageable. Imagine trying to measure out individual molecules for an experiment. It would be like trying to catch a specific ant in a stadium full of ants using tweezers. Impossible!
But with the mole, we have a way to scale up. We can talk about macroscopic amounts (like grams or kilograms) that correspond to a very precise number of microscopic particles. This is crucial for everything from developing new medicines to understanding how our bodies work.
Why So Many? The Nature of the Microscopic World
You might be wondering, "Why do we need so many molecules for aspirin to actually do anything?" It’s a fair question! Think about it like building with LEGOs. If you want to build a small toy car, you only need a few LEGO bricks. But if you want to build a life-sized LEGO replica of your house, you’re going to need a mountain of bricks.
Molecules are the LEGO bricks of our universe. And for a drug like aspirin to have a noticeable effect on your body, it needs to interact with a vast number of cells and receptors. Each molecule of aspirin does its small job, and when you have billions upon billions of them working together, they can achieve a significant outcome – like silencing that internal marching band.

It’s a bit like a flash mob. One person dancing by themselves is interesting, maybe a little quirky. But a thousand people dancing the same routine in a public square? That’s a spectacle! Each molecule is an individual dancer, and the mole represents the entire, coordinated flash mob of aspirin molecules ready to get to work.
The strength of aspirin, or any medication, is determined by how much of that active ingredient is present. And that amount is often talked about in terms of moles, or related to it, because it gives scientists a consistent way to compare different samples and ensure they are using the right "dose" of molecules.
The Practical Side of Gigantic Numbers
So, how does this translate to your everyday life, besides just understanding why that little white pill works? Well, it’s the invisible foundation of so much of what we rely on.
When you buy a bottle of aspirin, the label will tell you the strength in milligrams (mg). This milligram amount is directly related to the number of moles, and therefore the number of molecules, of acetylsalicylic acid present. A 500mg aspirin tablet has a certain number of molecules. A 100mg tablet has fewer.
The pharmaceutical industry relies heavily on these precise measurements. They need to know exactly how many molecules of a drug are in each dose to ensure it’s safe and effective. Too few, and it won’t work. Too many, and it could be dangerous.

Think about cooking. If a recipe calls for 2 cups of flour, and you accidentally use 2 tablespoons, your cake will be… well, let’s just say it won’t be a cake. It’ll be more like a slightly sweet, crumbly disappointment. The mole concept is the chemistry equivalent of accurate recipe measurements, just on an unfathomably grander scale.
It also helps in understanding chemical reactions. When two substances react, they do so in specific ratios of molecules. Knowing how many molecules are in a mole allows chemists to predict how much of one substance is needed to react with another, much like a chef knows how many eggs to add to a certain amount of flour.
A Moment of Scientific Wonder
The next time you reach for that aspirin, take a moment. Consider the incredible, invisible world contained within that small tablet. Think of the 6.022 x 1023 tiny warriors of relief, all meticulously counted and packaged by the laws of nature and the ingenuity of science.
It’s a testament to the fact that even the most mundane objects are brimming with incredible complexity. The world around us, from the air we breathe to the medicines we take, is built upon foundations that are almost too vast to comprehend. And the mole, with its astronomical number, is our gateway to appreciating that microscopic magnificence.
So, while you’re enjoying the sweet relief from your headache, give a silent nod to Avogadro and his number. It's the silent, unseen force that makes your pain relief possible, one impossibly tiny molecule at a time.